Summary
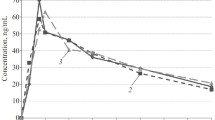
The steady-state intravenous pharmacokinetics of pirenzepine has been investigated in 57 subjects whose renal function ranged from normal to chronic failure requiring regular haemodialysis.
Pirenzepine renal clearance, total clearance and terminal (dominant) half-life were found to be correlated with the creatinine clearance (CLCR), but this was not the case for the volume of distribution and the nonrenal clearance. The therapeutic regimen was well tolerated by all subjects. Haemodialysis did not significantly contribute to the elimination of pirenzepine. Dosage adjustment need only be considered in patients with CLCR<25 ml/min in order to reduce the frequency of minor side-effects.
Similar content being viewed by others

References
Carmine AA, Brogden RN (1985) Pirenzepine: A review of its pharmacodynamic and pharmacokinetic properties and therapeutic efficacy in peptic ulcer disease and other allied diseases. Drugs 30: 85–126
Ventkateswaran PS, Jeffers A, Hocken AG (1972) Gastric acid secretion in chronic renal failure. Br Med J 4: 22–23
Shepherd AMN, Stewart WK, Wormsley KG (1973) Peptic ulceration in chronic renal failure. Lancet 1: 1357–1359
Hammer R, Bozler G, Zimmer A, Koss FW (1977) Pharmakokinetik und Metabolismus von L-S 519 CL2 (Pirenzepin) beim Menschen. Therapiewoche 27: 1575–1593
Tanswell P, Kasper W, Zahn G (1986) Automated monoclonal radioimmunoassays for pirenzepine, a selective muscarinic receptor antagonist, in plasma and urine. J Immunol Methods 93: 247–258
Heinzel G (1982). In: Bozler G, van Rossum SM (eds) Pharmacokinetics during drug development. Springer, New York, pp 207–208
Londong W, Londong V, Tanswell P, Voderholzer U (1985) A pharmacokinetic study of the efficacy of pirenzepine after several days of treatment. Klin Wochenschr 63 [Suppl 4]: 168
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Krakamp, B., Tanswell, P., Vogel, H. et al. Steady-state intravenous pharmacokinetics of pirenzepine in patients with differing degrees of renal dysfunction. Eur J Clin Pharmacol 36, 75–78 (1989). https://doi.org/10.1007/BF00561028
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00561028