Abstract
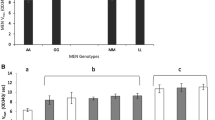
α-Glycerophosphate dehydrogenase (GPDH) occurs in Drosophila melanogaster in three isozymic forms. These are separable by starch gel electrophoresis and have been tentatively numbered 1, 2, and 3. GPDH-1 is most concentrated in the adult thorax and GPDH-3 in the abdomen; 1 and 3 are in approximately equal amounts in the head. GPDH-2 is relatively weak in all preparations. In larvae, only GPDH-3 is present. Purified GPDH-1 has optimal activity at pH 6.7–7.0. GPDH-3 at pH 7.5, and GPDH-2 is intermediate. Changes in total GPDH activity parallel larval growth, pupal histolysis, and differentiation of adult tissues. In the latter period the ratio of activity at pH 6.7 to pH 7.6 increases, reflecting the shift from GPDH-3 to GPDH-1. Two types of homozygous GPDH patterns which differ in the electrophoretic mobilities of all three isozymes have been found in inbred strains. In heterozygous adults six bands, the parental forms of GPDH-1 and GPDH-3 and “hybrid” forms of each, can be resolved. Analysis of F2 and backcross progeny suggests that a single genetic locus affects all three isozymes. Heterozygous embryos have only the maternal form of GPDH-3 until just before they hatch as first instar larvae. At this stage they have maternal and paternal GPDH-3 plus an intermediate band.
Similar content being viewed by others
References
Baranowski, T. (1963). α-Glycerophosphate dehydrogenase. In Boyer, P. D., Lardy, H., and Myrbäck, K. (eds.), The Enzymes, Vol. 7, Academic Press, New York, pp. 85–96.
Brosemer, R. W. (1965a). Changes in glycerophosphate dehydrogenase activity during development of the grasshopper Schistocerca vaga. Biochim. Biophys. Acta 96 61.
Brosemer, R. W. (1965b). The effect of puromycin and actinomycin D on the development of grasshopper flight muscle glycerophosphate dehydrogenase. Biochim. Biophys. Acta 99 388.
Brosemer, R. W., and Marquard, R. R. (1966). Insect extramitochondrial glycerophosphate dehydrogenase: II. Enzymatic properties and amino acid composition of the enzyme from honeybee (Apis mellifera) thoraxes. Biochim. Biophys. Acta 128 464.
Bücher, T. (1965). Formation of the specific structural and enzymic pattern of insect flight muscle. In Goodwin, T. W. (ed.), Aspects of Insect Biochemistry, Biochemical Society Symposium 25, Academic Press, New York, pp. 15–28.
Courtright, J. B. (1967). Polygenic control of aldehyde oxidase in Drosophila. Genetics 57 25.
Dickinson, W. J. (1968). Genetics and developmental regulation of aldehyde oxidase in Drosophila melanogaster. Genetics 60 173 (Abstr.).
Grell, E. H. (1967). Electrophoretic variants of α-glycerophosphate dehydrogenase in Drosophila melanogaster. Science 158 1319.
Grell, E. H., Jacobson, K. B., and Murphy, T. B. (1965). Alcohol dehydrogenase in Drosophila melanogaster. Isozymes and genetic variants. Science 149 80.
Hitzeroth, H., Klose, J., Ohno, S., and Wolf, U. (1968). Asynchronous activation of parental alleles at the tissue-specific gene loci observed on hybrid trout during early development. Biochem. Genet. 1 287.
Hubby, J. D., and Lewontin, R. C. (1966). A molecular approach to the study of genic heterozygosity in natural populations: I. The number of alleles at different loci in Drosophila pseudoobscura. Genetics 54 577.
Ingram, V. M. (1963). The Hemoglobins in Genetics and Evolution. Columbia University Press, New York, 165 pp.
Koen, A. L., and Shaw, C. R. (1964). A preparative method employing starch gel electrophoresis and electrodialysis. Anal. Biochem. 9 495.
O'Brian, S. J., and MacIntyre, R. J. (1968). Reports of new mutants. Drosophila Information Service 43 60.
Ohno, S., Stenius, C., Christian, L. C., and Harris, C. (1968). Synchronous activation of both parental alleles at the 6PGD locus of Japanese quail embryos. Biochem. Genet. 2 197.
Rechsteiner, M. C. (1968). Lactate dehydrogenase (LDH) and alpha-glycerophosphate dehydrogenase (αGPDH) in Drosophila melanogaster. Am. Zool. 8 821 (Abst.)
Sactor, B., and Dick, A. (1962). Pathways of hydrogen transport in the oxidation of extramitochondrial reduced diphosphopyridine nucleotide in flight muscle. J. Biol. Chem. 237 3259.
Wright, D. A., and Moyer, F. H. (1966). Parental influences on lactate dehydrogenase in the early development of hybrid frogs in the genus Rana. Exptl. Zool. 163 215.
Wright, D. A., and Moyer, F. H. (1968). Inheritance of frog lactate dehydrogenase patterns and the persistence of maternal isozymes during development. J. Exptl. Zool. 167 197.
Wright, D. A., and Shaw, C. R. (1969). Gene expression and enzyme changes during development in Drosophila (in preparation).
Young, H. L., and Pace, N. (1958). Some physical and chemical properties of crystalline α-glycerophosphate dehydrogenase. Arch. Biochem. Biophys. 75 125.
Zebe, E., Delbruck, A., and Bücher, T. (1959). Über den Glycerin-I-P-Cyclus im Flugmuskel von Locusta migratoria. Biochem. Z. 331 254.
Author information
Authors and Affiliations
Additional information
This project was supported in part by National Institutes of Health research grant GM-15597.
Rights and permissions
About this article
Cite this article
Wright, D.A., Shaw, C.R. Genetics and ontogeny of α-glycerophosphate dehydrogenase isozymes in Drosophila melanogaster . Biochem Genet 3, 343–353 (1969). https://doi.org/10.1007/BF00485718
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00485718