Abstract
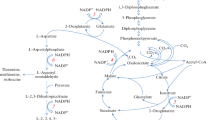
Diauxic growth of Pseudomonas oxalaticus was observed on a mixture of formate and oxalate in batch cultures. In the first phase of growth only formate was used. The capacity to oxidize oxalate appeared during the lag phase of 2–4 h after the exhaustion of formate and was followed by a second phase of growth on oxalate. The rate of autotrophic 14CO2 fixation measured in washed cell suspensions decreased markedly in this second growth phase on the addition of oxalate. In mixtures of formate with acetate, glyoxylate or glycollate, simultaneous utilization of both substrates was observed. During growth on acetate plus formate formate-oxidizing capacity remained low. With low acetate concentrations, sufficient formate remained after the exhaustion of acetate to support a second growth phase on formate. This phase followed a 1.5–2 h lag, during which formate-oxidizing capacity increased and the Calvin cycle enzymes were synthesized. In mixtures of formate with glyoxylate or glycollate, the formate-oxidizing capacity was high, formate was oxidized rapidly, and no second growth phase was seen. In these latter mixtures high activities of a membrane-bound, phenazine methosulphate/2,6-dichlorophenolindophenollinked formate dehydrogenase and low activities of the soluble NAD-linked formate dehydrogenase were detected. The synthesis of ribulose-1,5-diphosphate carboxylase was totally repressed during growth on formate plus glycollate and partially repressed on formate plus glyoxylate. The regulation of Calvin cyclus enzymes in Pseudomonas oxalaticus is discussed.
Similar content being viewed by others
Abbreviations
- PMS:
-
phenazine methosulphate
- DCPIP:
-
2,6-dichlorophenolindophenol
- RuDP:
-
ribulose-1,5-diphosphate
- EDTA:
-
ethylenediamine tetraacetate
- GSH:
-
reduced glutathione
References
Blackmore, M. A., Quayle, J. R.: Choice between autotrophy and heterotrophy in Pseudomonas oxalaticus. Growth in mixed substrates. Biochem. J. 107, 705–713 (1968)
Blackmore, M. A., Quayle, J. R.: Microbial growth on oxalate by a route not involving glyoxylate carboligase. Biochem. J. 118, 53–59 (1970)
Calkins, V. P.: Microdetermination of glycolic and oxalic acids. Industr. Engin. Chem. 15, 762–763 (1943)
Dijkhuizen, L., Harder, W.: Substrate inhibition in Pseudomonas oxalaticus OX1: a kinetic study of growth inhibition by oxalate and formate using extended cultures. Antonie van Leeuwenhoek 41, 135–146 (1975)
Dijkhuizen, L., Groen, L., Harder, W., Konings, W. N.: Active transport of oxalate by Pseudomonas oxalaticus OX1. Arch. Microbiol. 115, 223–227 (1977a)
Dijkhuizen, L., Wiersma, M., Harder, W.: Energy production and growth of Pseudomonas oxalaticus OX1 on oxalate and formate. Arch. Microbiol. 115, 229–236 (1977b)
Dixon, G. H., Kornberg, H. L.: Assay methods for key enzymes of the glyoxylate cycle. Biochem. J. 72, 3p (1959)
Harder, W., Dijkhuizen, L.: Mixed substrate utilization in microorganisms. In: Continuous culture 6: Applications and new fields (A. C. R. Dean, D. C. Ellwood, C. G. T. Evans, J. Melling, eds.), pp. 297–314. Chichester-Oxford: Ellis Harwood 1976
Harder, W., Visser, K., Kuenen, J. G.: Laboratory fermenter with an improved magnetic drive. Lab. Practice 23, 644–645 (1974)
Hart, B. A., Gibson, J.: Ribulose-5-phosphate kinase from Chromatium. In: Methods in enzymology (W. B. Jakoby, ed.), Vol. XLII, pp. 115–119. New York: Academic Press 1975
Johnson, P. A., Jones-Mortimer, M. C., Quayle, J. R.: Use of a purified bacterial formate dehydrogenase for the micro-estimation of formate. Biochim. Biophys. Acta 89, 351–353 (1964)
Kaltwasser, H.: Harnsäureabbau und Biosynthese der Enzyme Uricase, Glyoxycarboligase und Urease bei Hydroxygenomonas H16. 1. Bildung von Glyoxylatcarboligase und d-Glycerat-3-Dehydrogenase. Arch. Mikrobiol. 64, 71–84 (1968)
Khambata, S. R., Bhat, J. V.: Studies on a new oxalate decomposing bacterium, Pseudomonas oxalaticus. J. Bacteriol. 66, 505–507 (1953)
Knight, M., Dijkhuizen, L., Harder, W.: Diauxic growth of Pseudomonas oxalaticus OX1 on mixtures of formate and oxalate. Proc. Soc. Gen. Microbiol. 3, 7 (1975)
Lang, E., Lang, H.: Spezifische Farbreaktion zum direkten Nachweis der Ameisensäure. Z. Anal. Chem. 260, 8–10 (1972)
Lowry, O. H., Rosebrough, N. J., Farr, A. L., Randall, R. J.: Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193, 265–275 (1951)
Quayle, J. R.: Metabolism of C1 compounds in autotrophic and heterotrophic microorganisms. Ann. Rev. Microbiol. 15, 119–152 (1961)
Quayle, J. R., Keech, D. B.: Carbon assimilation by Pseudomonas oxalaticus (OX1). 3. Oxalate utilization during growth on oxalate. Biochem. J. 75, 515–523 (1960)
Quayle, J. R., Taylor, G. A.: Carbon assimilation by Pseudomonas oxalaticus (OX1). 5. Purification and properties of glyoxylic dehydrogenase. Biochem. J. 78, 611–615 (1961)
Schlegel, H. G.: The physiology of hydrogen bacteria. Antonie van Leeuwenhoek 42, 181–201 (1976)
Vogel, A. I.: A textbook of quantitative inorganic analysis, 2nd ed., p. 273. London-New York-Toronto: Longmans, Green & Co. 1955
Weissbach, A., Horecker, B. L., Hurwitz, J.: The enzymatic formation of phosphoglyceric acid from ribulose diphosphate and carbon dioxide. J. Biol. Chem. 218, 795–810 (1956)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dijkhuizen, L., Knight, M. & Harder, W. Metabolic regulation in Pseudomonas oxalaticus OX1. Arch. Microbiol. 116, 77–83 (1978). https://doi.org/10.1007/BF00408736
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00408736