Abstract
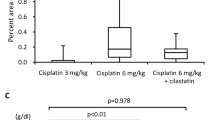
Adriamycin (ADR) has a broad spectrum of antitumoral activity but is ineffective against human brain tumors. However, such tumors can be sensitive to a combination of adriamycin and lipophilic antineoplastic agents such as the nitrosoureas. CCNU, a nitrosourea, induces cholestasis in the rat and ADR is predominantly excreted via the biliary route. We decided to investigate the effect of CCNU on the nephrotic syndrome induced by ADR. Female Wistar rats were injected with a single dose of 10 mg/kg ADR and 24 h later were force fed 20 mg/kg CCNU in a single dose. Animals were sacrificed 4, 8, 15, 21, 28 or 60 days after the injection of ADR. A high rate of fatality (60%) occurred after the 21st day of treatment. Biological changes (alkaline phosphatase, SGPT, bilirubin) and ultrastructural studies showed that CCNU and CCNU+ADR induced the same degree of cholestasis. With the administered dose, CCNU is not nephrotoxic, ADR induces a nephrotic syndrome and ADR+CCNU appeared more nephrotoxic. With ADR, visceral epithelial foot process fusion was seen on day 15 and tubulo-interstitial lesions and glomerulosclerosis on day 60. With ADR+CCNU fusion of the foot process was seen on day 4, glomerular vacuolation on day 8, tubulo-interstitial alterations on day 15 and glomerulosclerosis on day 60. For both ADR and ADR+CCNU wrinkling and thickening of the basement membrane of proximal tubular cells were seen on day 60. Lipid mesangial overload was seen with ADR and was more intense with ADR+CCNU on day 60. CCNU hepatoxicity modifies the excretion of ADR and the predominantly renal excretion of ADR seems to induce earlier renal alterations in ADR+CCNU-treated rats. This study supports the concept that lipid mesangial overload may play an important role in chronic progressive glomerulosclerosis and thus the ADR+CCNU combination appears to be an interesting model in which to study these relationships.
Similar content being viewed by others

References
Bachur NR, Egorin MJ, Hildebrand RC, Takanashi S (1973) Human biliary metabolites of adriamycin and daunorubicin (abstract). Proc Am Assoc Cancer Res 14: 14
Benjamin RS, Riggs CE, Bachur NR (1973) Pharmacokinetics and metabolism of adriamycin in man. Clin Pharmacol Ther 14: 592–600
Benjamin RS, Riggs CE Jr, Serpick AA (1974) Biliary excretion of adriamycin (A) in man (abstract). Clin Res 22: 483
Benjamin RS, Riggs CE, Bachur NR (1977) Plasma pharmacokinetics of adriamycin and its metabolites in humans with normal hepatic and renal function. Cancer Res 37: 1416–1420
Bertani T, Poggi A, Pozzoni R, Delaini F, Sacchi G, Thoua Y, Mecca G, Remuzzi G, Donati MB (1982) Adriamycin induced nephrotic syndrome in rats — sequence of pathologic events. Lab Invest 46: 16–23
Bertani T, Rocchi G, Mecca G, Sacchi G, Remuzzi G (1983) Adriamycin induced chronic proteinuria: a new model of glomerular focal sclerosis (abstract). Kidney Int 23: 192
Bessey OA, Lowry OH, Brock MJ (1946) A method for the rapid determination of alkaline phosphatase with five cubic millimeters of serum. J Biol Chem 164: 321–325
Blum RH (1975) An overview of studies with adriamycin (NSC 123127) in the United States. Cancer Chemother Rep 6: 247–253
Blum RH, Carter SK (1974) Adriamycin: a new anticancer drug with significant clinical activity. Ann Int Med 80: 249–259
Carter SK (1975) Adriamycin — a review. J Natl Cancer Inst 55: 1265–1274
Chang P, Riggs CE Jr, Scheerer MT, Wiernik PH, Bachur NR (1976) Combination chemotherapy with adriamycin and streptozotocin: II Clinicopharmacologic correlation of augmented adriamycin toxicity caused by streptozotocin. Clin Pharmacol Ther 20: 611–616
Difronzo G, Gambretta RA, Lenaz L (1971) Distribution and metabolism of adriamycin in mice comparison with daunomycin. Rev Eur Etud Clin Biol 16: 572–576
Einhorn LH, Livingston RB, Gottlieb JA (1973) Combination chemotherapy with adriamycin (NSC-123127) and 1-(2-chloroethyl)-3-cyclohexyl-1-nitrosourea (CCNU; 79037). Cancer Chemother Rep 57: 437–445
Fajardo LF, Eltringham JR, Stewart JR, Klauber MR (1980) Adriamycin nephrotoxicity. Lab Invest 43: 242–253
Fewer D, Wilson CB, Boldrey EB (1972) Phase II study of 1-(2-chloroethyl)-3-cyclohexyl-1-nitrosourea (CCNU, NSC-79037) in the treatment of brain tumors. Cancer Chemother Rep 56: 421–427
Friedman MA, Carter SB (1978) Serious toxicities associated with chemotherapy. Semin Oncol 5: 193–202
Grond J, Weening JJ, Elema JD (1984) Glomerular sclerosis in nephrotic rats comparison of the longterm effects of adriamycin and aminonucleoside. Lab Invest 51: 277–285
Pugh D, Leaback H, Walker PG (1957) Studies on glucosaminidases: N-acetyl-B-d-glucosaminidase in rat kidney. Biochem J 65: 464–469
Riggs CE, Benjamin RS, Serpick AA, Bachur NR (1977) Biliary disposition of adriamycin. Clin Pharmacol Ther 22: 234–241
Selawry OS, Hansen HH (1972) Superiority of CCNU 1-(2-chloroethyl)-3-cyclohexyl-1-nitrosourea, NSC-79037 over BCNU (1,3-bis 2-chloroethyl-1-nitrosourea); NSC 409962 in treatment of advanced Hodgkin's disease (abstract). Proc Am Assoc Cancer Res 13: 46
Takita H, Brugarola SA (1973) Effect of CCNU (NSC-79037) on bronchogenic carcinoma. J Natl Cancer Inst (USA) 50: 49–53
Tavoloni N, Guarino AM (1980) Biliary and urinary excretion of adriamycin in anesthetized rats. Pharmacology 20: 256–267
Viotte G, Lahouel M, Ducastelle TH, Sumereau E, Morin JP, Hemet J, Fillastre JP (1986) Hepatotoxicité du (chloro-2-ethyl)-1-cyclohexyl-3-nitroso-1-urea (CCNU) chez la rat. Pathol Biol 34: 40–47
Walker MD (1973) Nitrosoureas in central nervous system tumors. Cancer Chemother Rep 4: 21–26
Woolley PV (1983) Hepatic and pancreatic damage produced by cytotoxic. Cancer Treat Rev 10: 117–137
Young DM (1975) Pathologic effects of adriamycin in experimental system. Cancer Chemother Rep 6: 159–165
Author information
Authors and Affiliations
Additional information
This work was supported by INSERM-CNAMTS and ROGER BELLON Company grants
Rights and permissions
About this article
Cite this article
Raguenez-Viotte, G., Lahouel, M., Ducastelle, T. et al. CCNU-Adriamycin association induces earlier and more severe nephropathy in rats. Arch Toxicol 61, 282–291 (1988). https://doi.org/10.1007/BF00364851
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00364851