Summary
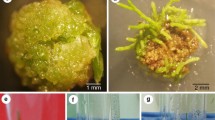
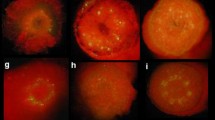
The Agrobacterium tumefaciens octopine strain B6S3 and the nopaline strain C58 were compared for their ability to induce opine synthesis on Kalanchoe daigremontiana stem fragments. Whereas B6S3 induced high levels of octopine synthesis, C58 induced only low levels of nopaline synthesis. However, C58-induced nopaline synthesis was greatly increased by mixed infection with B6S3. This effect (called “helper-effect”) was shown to be due to the activity of a 5 kb fragment from the virulence region of the B6S3 Ti plasmid, since incorporation of this fragment into the C58 plasmid enabled C58 to induce high levels of nopaline synthesis in the absence of a helper strain. The 5kb region contains the vir F locus, as defined earlier (Hooykaas et al. 1984b). A possible correlation between the helper function and vir F is discussed. Our results show that large differences in virulence on particular host plants exist between natural Agrobacterium strains and can be overcome by mixed infections.
Similar content being viewed by others
References
Akiyoshi DE, Morris RO, Hinz R, Mishke BS, Kosuge T, Garfinkel DJ, Gordon MP, Nester EW (1983) Cytokinin balance in crown gall tumors is regulated by specific loci in the T-DNA. Proc Natl Acad Sci 80:407–411
Barry GF, Rogers SG, Fraley RT, Brand L (1984) Identification of a cloned cytokinin gene. Proc Natl Acad Sci 81:4776–4780
Bolivar F (1978) Construction and characterization of new cloning vehicles III. Derivatives of plasmid pBR322 carrying unique EcoRI sites for selection of EcoRI generated recombinant DNA molecules. Gene 4:121–126
Caplan A, Herrera-Estrella L, Inzé D, Van Haute E, Van Montagu M, Schell J, Zambryski P (1983) Introduction of genetic material into plant cells. Science 222:815–821
De Greve H, Decraemer H, Seurinck J, Van Montagu M, Schell J (1981) The functional organization of the octopine Agrobacterium tumefaciens plasmid pTiB6S3. Plasmid 6:235–248
Depicker A, De Wilde M, De Vos G, Van Montagu M, Schell J (1980) Molecular cloning of overlapping segments of the nopaline Ti plasmid pTiC58 as a means to restriction endonuclease mapping. Plasmid 3:193–211
De Vos G, De Beuckeleer M, Van Montagu M, Schell J (1981) Restriction endonuclease mapping of the octopine tumor-inducing plasmid pTiAch5 of Agrobacterium tumefaciens. Plasmid 6:249–253
Dhaese P, De Greve H, Decraemer H, Schell J, Van Montagu M (1979) Rapid mapping of transposon insertion and deletion mutations in the large Ti-plasmids of Agrobacterium tumefaciens. Nucl Acids Res 7:1837–1849
Engler G, Depicker A, Maenhout R, Villarroel R, Van Montagu M, Schell J (1981) Physical mapping of DNA base sequence homologies between an octopine and a nopaline Ti plasmid of Agrobacterium tumefaciens. J Mol Biol 152:183–208
Garfinkel DJ, Nester EW (1980) Agrobacterium tumefaciens mutants affected in crown gall tumorigenesis and octopine catabolism. J Bacteriol 144:732–743
Guyon P, Chilton MD, Petit A, Tempé J (1980) Agropine in “nulltype” crown gall tumors: evidence for generality of the opine concept. Proc Natl Acad Sci 77:2693–2697
Hernalsteens JP, De Greve H, Van Montagu M, Schell J (1978) Mutagenesis by insertion of the drug resistance transposon Tn7 applied to the Ti plasmid of Agrobacterium tumefaciens. Plasmid 1:218–225
Hille J, Klasen I, Schilperoort RA (1982) Construction and application of R prime plasmids, carrying different segments of an octopine Ti plasmid from Agrobacterium tumefaciens, for complementation of vir genes. Plasmid 7:107–118
Hille J, Van Kan J, Schilperoort RA (1984) trans-acting virulence functions of the octopine Ti plasmid from Agrobacterium tumefaciens. J Bacteriol 158:754–756
Hoekema A, Hooykaas PJJ, Schilperoort RA (1984) Transfer of the octopine T-DNA segment to plant cells mediated by different types of Agrobacterium tumor- or root-inducing plasmids: generality of virulence systems. J Bacteriol 158:383–385
Holsters M, Silva B, Van Vliet F, Genetello C, De Block M, Dhaese P, Depicker A, Inzé D, Engler G, Villarroel R, Van Montagu M, Schell J (1980) The functional organization of the nopaline A. tumefaciens plasmid pTiC58. Plasmid 3:212–230
Hooykaas PJJ, Hofker M, Den Dulk-Ras H, Schilperoort RA (1984b) A comparison of virulence determinants in an octopine Ti plasmid, a nopaline Ti plasmid and a Ri plasmid by complementation analysis of Agrobacterium tumefaciens mutants. Plasmid 11:195–205
Hooykaas PJJ, Schilperoort RA (1984a) The molecular genetics of Crown gall tumorigenesis. Adv Genet 22:209–283
Iyer VN, Klee HJ, Nester EW (1982) Units of genetic expression in the virulence region of a plant tumor-inducing plasmid of Agrobacterium tumefaciens. Mol Gen Genet 188:418–424
Kao JC, Perry KL, Kado CI (1981) Indoleacetic acid complementation and its relation to host range specifying genes on the Ti plasmid of Agrobacterium tumefaciens. Mol Gen Genet 188:425–432
Klee HJ, White FF, Iyer VN, Gordon MP, Nester EW (1983) Mutational analysis of the virulence region of an Agrobacterium tumefaciens Ti plasmid. J Bacteriol 153:878–883
Knauf VC, Panagopoulos CG, Nester EW (1982) Genetic factors controlling the host range of Agrobacterium tumefaciens. Phytopathology 72:1545–1549
Kanuf VC, Yanofsky M, Montoya A, Nester EW (1984) Physical and functional map of an Agrobacterium tumefaciens tumor-inducing plasmid that confers a narrow host range. J Bacteriol 160:564–568
Leemans J, Deblaere R, Willmitzer L, De Greve H, Hernalsteens JP, Van Montagu M, Schell J (1982) Genetic identification of functions of TL-DNA transcripts in octopine Crown gall cells. EMBO J 1:147–152
Leemans J, Shaw C, Deblaere R, De Greve H, Hernalsteens JP, Maes M, Van Montagu M, Schell J (1981) Site-specific mutagenesis of Agrobacterium Ti plasmids and transfer of genes to plant cells. J Mol Appl Genet 1:149–164
Nester EW, Gordon MP, Amasino RM, Yanofski MF (1984) Crown gall: a molecular and physiological analysis. Ann Rev Plant Physiol 35:387–413
Ooms G, Klapwijk PM, Poulis JA, Schilperoort RA (1980) Characterization of Tn904 insertions in octopine Ti plasmid mutants of Agrobacterium tumefaciens. J Bacteriol 144:82–91
Otten L (1982) Lysopine dehydrogenase as an early marker in Crown gall transformation. Plant Sci Lett 25:15–27
Otten L, De Greve H, Leemans J, Hain R, Hooykaas PJJ, Schell J (1984) Restoration of virulence of vir region mutants of Agrobacterium tumefaciens strain B6S3 by coinfection with normal and mutant Agrobacterium strains. Mol Gen Genet 195:159–163
Rao RN, Rogers SG (1979) Plasmid pKC7: a vector containing ten restriction endonuclease sites suitable for cloning DNA segments. Gene 7:79–82
Schröder G, Waffenschmidt S, Weiler EW, Schröder J (1984) The T-region of Ti plasmids codes for an enzyme synthesizing indole-3-acetic Eur J Biochem 138:387–391
Thomashow MF, Panagopoulos CG, Gordon MP, Nester EW (1980) Host range of Agrobacterium tumefaciens is determined by the Ti-plasmid. Nature 283:794–796
Van Haute E, Joos H, Maes M, Warren E, Van Montagu M, Schell J (1983) Intergeneric transfer and exchange recombination of restriction fragments cloned in pBR322: a novel strategy for the reversed genetics of Ti plasmids of Agrobacterium tumefaciens. EMBO J 2:411–417
Zambryski P, Joos H, Genetello C, Leemans J, Van Montagu M, Schell J (1983) Ti plasmid vector for the introduction of DNA into plant cells without alteration of their normal regeneration capacity. EMBO J 2:2143–2150
Author information
Authors and Affiliations
Additional information
Communicated by H. Saedler
Dedicated to Professor Georg Melchers to celebrate his 50-year association with the journal
Rights and permissions
About this article
Cite this article
Otten, L., Piotrowiak, G., Hooykaas, P. et al. Identification of an Agrobacterium tumefaciens pTiB6S3 vir region fragment that enhances the virulence of pTiC58. Molec Gen Genet 199, 189–193 (1985). https://doi.org/10.1007/BF00330258
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00330258