Abstract
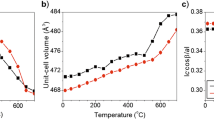
Kinetic studies of isothermal heating experiments (600–800° C) on aluminous pyroxenes (Mg0.942Fe 2+0.880 Fe 3+0.068 Mn0.016Ca0.010Al0.084) (Si1.848Al0.152) permit the determination of rate constant of isothermal disordering as 2.5457 E13(±1.4 E13) min−1. The activation energy is determined as 278 (±23) kJ/mol. Data on two other aluminous pyroxenes at 700° C indicate that the rate constant decreases significantly with increasing amount of trivalent cations. There is a similar but reverse correlation between the concentration of trivalent cations and the Fe2+-Mg equilibrium distribution between sites. The site distribution coefficient increases with increasing concentration of trivalent cations at constant temperature.
Similar content being viewed by others
References
Besancon JR (1981a) Rate of cation ordering in orthopyroxenes. Am Mineral 66:965–973
Dienes GJ (1955) Kinetics of order-disorder transformation. Acta Metall 3:549–557
Ganguly J (1982) Mg-Fe order-disorder in ferromagnesian silicates II. Thermodynamics, kinetics and geological applications. In: Advances in physical geochemistry, Vol. 2, Springer, New York, pp 58–99
Mueller RF (1967) Model for order-disorder kinetics in certain quasi-binary crystals of continuously variable composition. J Phys Chem Solids 28:2239–2243
Mueller RF (1969) Kinetics and thermodynamics of intracrystalline distribution. Mineral Soc Amer Spec Pap 2:83–93
Seifert F, Virgo D (1975) Kinetics of Fe2+-Mg order-disorder reaction in anthophillites: quantitative cooling rates. Science 188:1107–1109
Seifert F (1983) Mössbauer line broadening in aluminous orthopyroxenes: evidence for next nearest neighbors interactions and short-range order. N Jahrb Mineral Abh 148:141–162
Tazzoli V, Domeneghetti MC (1987) Crystal-chemistry of natural and heated aluminous orthopyroxenes. Phys Chem Minerals 15:131–139
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Saxena, S.K., Tazzoli, V. & Domeneghetti, M.C. Kinetics of Fe2+-Mg distribution in aluminous orthopyroxenes. Phys Chem Minerals 15, 140–147 (1987). https://doi.org/10.1007/BF00308776
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00308776