Abstract
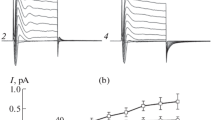
Hypotonically induced changes in whole-cell currents and in cell volume were studied in the HSG cloned cell line using the whole-cell, patch clamp and Coulter counter techniques, respectively. Exposures to 10 to 50% hypotonic solutions induced dose-dependent increases in whole-cell conductances when measured using K+ and Cl− containing solutions. An outward current detected at 0 mV, corresponded to a K+ current which was transiently activated, (usually preceding activation of an inward current and had several characteristics in common with a Ca2+-activated K+ current we previously described in these cells. The hypotonically induced inward current had characteristics of a Cl− current. This current was inhibited by NPPB (5-nitro-2-(3-phenyl-propylamino)-benzoate) and SITS (4-acetamido-4′-isothiocyanostilbene), and its reversal potentials corresponded to the Cl− equilibrium potentials at high and low external Cl− concentrations. The induced current inactivated at voltages greater than +80 mV, and the I-V curve was outwardly rectifying. The current was unaffected by addition of BAPTA or removal of GTP from the patch pipette, but was inhibited by removal of ATP or by the presence of extracellular arachidonic acid, quinacrine, nordihydroguairetic acid, and cytochalasin D. Moreover, exposure of HSG cells to hypotonic media caused them to swell and then to undergo a regulatory volume decrease (RVD) response. Neither NPPB, SITS or quinine acting alone could inhibit RVD, but NPPB and quinine together totally inhibited RVD. These properties, plus the magnitudes of the induced currents, indicate that the hypotonically induced K+ and Cl− currents may underlie the RVD response. Cytochalasin D also blocked the RVD response, indicating that intact cytoskeletal F-actin may be required for activation of the present currents. Hence, our results indicate that hypotonic stress activates K+ and Cl− conductances in these cells, and that the activation pathway for the K+ conductance apparently involves [Ca2+], while the activation pathway for the Cl− conductance does not involve [Ca2+] nor lipoxygenase metabolism, but does require intact cytoskeletal F-actin.
Similar content being viewed by others
References
Ackerman, M.J., Wickman, K.D., Clapham, D.E. 1994. Hypotonicity activates a native chloride current in Xenopus oocytes. J. Gen. Physiol. 103:153–179
Altenberg, G.A., Deitmer, J.W., Glass, D.C., Reuss, L. 1994. P-glycoprotein-associated Cl− currents are activated by cell swelling but do not contribute to cell volume regulation. Cancer Res 54:618–622
Chan, H.C., Fu, W.O., Chung, Y.W., Huang, S.J., Zhou, T.S., Wong, P.Y.D. 1993. Characterization of a swelling-induced chloride conductance in cultured rat epididymal cells. Am. J. Physiol. 265:C997-C1004
Cantiello, H.F., Prat, A.G., Bonventre, J.V., Cunningham, C.C., Hartwig, J.H., Ausiello, D.A. 1993. Actin-binding protein contributes to cell volume regulatory ion channel activation in melanoma cells. J. Biol. Chem. 268:4596–4599
Christensen, O., Hoffmann, E.K. 1992. Cell swelling activates K+ and Cl− channels as well as nonselective, stretch-activated cation channels in Ehrlich ascites tumor cells. J. Membrane Biol. 129:13–36
Cornet, M., Lambert, I.H., Hoffmann, E.K. 1993. Relation between cytoskeleton, hypo-osmotic treatment and volume regulation in Ehrlich ascites tumor cells. J. Membrane Biol. 131:55–66
Diaz, M., Valverde, M.A., Higgins, C.F., Rucareanu, C., Sepúlveda, F.V. 1993. Volume-activated chloride channels in HeLa cells are blocked by verapamil and dideoxyforskolin. Pfluegers Arch 422:347–353
Diener, M., Nobles, M., Rummel, W. 1992. Activation of basolateral Cl− channels in the rat colonic epithelium during regulatory volume decrease. Pfluegers Arch. 421:530–538
Doroshenko, P., Neher, E. 1992. Volume-sensitive chloride conductance in bovine chromaffin cell membrane. J. Physiol. 449:197–218
Dreinhofer, J., Gogelein, H.M., Greger, R. 1988. Blocking kinetics of Cl− channels in colonic carcinoma cells (HT29) as revealed by 5-nitro-2-(3-phenylpropylamino) benzoic acid (NPPB). Biochim. Biophys. Acta 946:135–142
Foskett, J.K. 1990. [Ca2+]i modulation of Cl− content controls cell volume in single salivary acinar cells during fluid secretion. Am. J. Physiol 259:C998-C1004
Foskett, J.K., Melvin, J.E. 1990. Activation of salivary secretion coupling of cell volume and [Ca2+] i in single cells. Science 224:1582–1585
Foskett, J.K., Spring, K.R. 1985. Involvement of calcium and cytoskeleton in gallbladder epithelial cell volume regulation. Am. J. Physiol. 248:C27-C36
Grynkiewicz, G., Poenie, M., Tsien, R.Y. 1985. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J. Biol. Chem. 260:3440–3450
Hazama, A., Okada, Y. 1988. Ca2+ sensitivity of volume-regulatory K+ and Cl− channels in cultured human epithelial cells. J. Physiol. 402:687–702
Hoffmann, E.K., Simonsen, L.O. 1989. Membrane mechanisms in volume and pH regulation in vertebrate cells. Physiol. Reviews 69:315–382
Hyun, C.S., Binder, H.J. 1993. Mechanism of leukotriene D4 stimulation of Cl− secretion in rat distal colon in vitro. Am. J. Physiol. 265:G467-G473
Ishikawa, T., Cook, D.I.. 1994. Characterization of an outwardly rectifying chloride channel in a human submandibular gland duct cell line (HSG) Pfluegers Arch. 427:203–209
Ishikawa, T., Cook, D.I., Young, J.A. 1991. Ionic channels in a human salivary duct cell line (HSG). In: Ionic Basis and Energy Metabolism of Epithelial Transport. M. Murakami, Y. Seo, A, Kuwahara, and H. Watari, editors. Nat. Inst. Physiol. Sci., pp. 185–186. Okazaki, Japan.
Izutsu, K.T., Fatherazi, S., Wellner, R.B., Herrington, J., Belton, C.M., Oda, D. 1994. Characterization and regulation of a muscarinically activated current in HSG cells. Am. J. Physiol. 266:C58-C66
Krapivinsky, G.B., Ackerman, M.J., Gordon, E.A., Krapivinsky, L.D., Clapham, D.E. 1994. Molecular characterization of a swelling-induced chloride conductance regulatory protein. Cell 76:439–448
Kubo, M., Okada, Y. 1992. Volume-regulatory Cl− channel currents in cultured human epithelial cells. J. Physiol. 456:351–371
Lambert, I.H. 1987. Effect of arachidonic acid, fatty acids, prostaglandins, and leukotrienes on volume regulation in Ehrlich ascites tumor cells. J. Membrane Biol. 98:207–221
Lewis, S.E.A., Donaldson, P. 1990. Ion channels and cell volume regulation: Chaos in an organized system. News Physiol. Sci. 5:112–119
Lewis, R.S., Ross, P.E., Cahalan, M.D. 1993. Chloride channels activated by osmotic stress in T lymphocyte. J. Gen. Physiol. 101:801–826
Mastrocola, T., Lambert, I.H., Kramhøft, B., Rugolo, M., Hoffmann, E.K. 1993. Volume regulation in human fibroblasts: Role of Ca2+ and 5-lipoxygenase products in the activation of the Cl− efflux. J. Membrane. Biol. 136:55–62
Moran, A., Turner, R.J. 1993. Secretagogue-induced RVD in HSY cells is due to K+ channels activated by Ca2+ and protein kinase C. Am. J. Physiol. 265:C1405-C1411
Patton, L.L., Wellner, R.B. 1993. Established salivary cell lines. In: Biology of the Salivary Glands. K. Dobrosielski-Vergona, editor. CRC Press, Boca Raton, FL
Paulmichi, M., Li, Y., Wickman, K., Ackerman, M., Peralta, E., Clapham, D. 1992. New mammalian chloride channel identified by expression cloning. Nature 356:238–241
Pollard, C.E. 1993. A volume-sensitive Cl− conductance in mouse neuroblastoma x rat dorsal root ganglion cell line (F 11). Brain Res 614:178–184
Rugolo, M., Mastrocola, T., De Luca, M., Romeo, G., Galietta, L.J.V. 1992. A volume-sensitive chloride conductance revealed in cultured human keratinocytes by 36Cl− efflux and whole-cell patch clamp recording. Biochim. Biophys. Acta 1112:39–44
Samman, G., Ohtsuyama, M., Sato, F., Sato, K. 1993. Volumeactivated K+ and Cl− pathways of dissociated eccrine clear cells. Am. J. Physiol. 265:R990-R1000
Sato, K., Ohtsuyama, M., Sato, F. 1993. Whole-cell K and Cl currents in dissociated eccrine secretory coil cells during stimulation. J. Membrane Biol. 134:93–106
Shirasuna, K., Sato, M., Miyazaki, T. 1981. A neoplastic epithelial duct cell line established from an irradiated human salivary gland. Cancer 48:745–752
Shliwa, M. 1982. Action of cytochalasin D on cytoskeletal networks. J. Cell Biol. 92:79–91
Stoddard, J.S., Steinbach, J.H., Simchowitz, L. 1993. Whole cell Cl− currents in human neutrophils induced by cell swelling. Am. J. Physiol. 265:C156-C165
Suzuki, M., Miyazaki, K., Ikeda, M., Kawaguchi, Y., Sakai, O. 1993. F-Actin network may regulate a Cl− channel in renal proximal tubule cells. J. Membrane Biol. 134:31–39
Valverde, M.A., Diaz, M., Sepúlveda, F.V., Gill, D.R., Hyde, S.C., Higgins, C.F. 1992. Volume-regulated chloride channels associated with the human multidrug-resistance p-glycoprotein. Nature 355:830–833
Worrell, R.T., Butt, A.G., Cliff, W.H., Frizzell, R.A. 1989. A volume-sensitive chloride conductance in human colonic cell line T84. Am. J. Physiol. 256:C1111-C1119
Young, J.A., van Lennep, E.W. 1979. Transport in salivary and salt glands. In: Membrane Transport in Biology, Vol. IVB, Chapter 12, pp. 563–692
Author information
Authors and Affiliations
Additional information
We thank Mr. Louis Stamps for excellent technical support. Thanks also to Dr. Mitsunobu Sato from the Second Department of Oral and Maxillofacial Surgery, Tokushima University, Japan for sending us the HSG-PA cells, and to Dr. Englert from Hoechst company for providing us with NPPB. This work was supported by National Institute of Dental Research grants R01 DE09812 and R03 DE10535.
Rights and permissions
About this article
Cite this article
Fatherazi, S., Izutsu, K.T., Wellner, R.B. et al. Hypotonically activated chloride current in HSG cells. J. Membarin Biol. 142, 181–193 (1994). https://doi.org/10.1007/BF00234940
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00234940