Summary
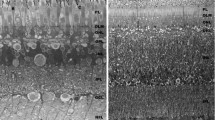
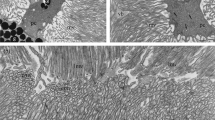
(1) The distribution of the extracellular space (ECS) in the outer part of the locust compound eye has been mapped with lanthanum and ruthenium red, applied to the retina. (2) In the photoreceptor zone, about 2.4% of the volume is ECS, in agreement with radiotracer and electrical estimates. Of this ECS, about 70% lies in lacunae between ommatidia, but only 1–2% adjacent to the photosensitive rhabdom. The lacunae are filled with material which binds applied tracers, and are thought to be structural spaces. (3) It has been suggested several times that such a small cation pool is insufficient to sustain more than a few large photoresponses, but this is shown to be incorrect. Enough Na+ lies within the rhabdomal ECS and within rapid diffusional access to it, to impose no immediate limitation. (4) The palisade vacuoles surrounding the rhabdom are intracellular, and are typical of light as well as dark-adapted eyes. (5) Tracers fail to penetrate more than about 30 μm into the axon zone, in agreement with electrical, dye and radiotracer indications of a blood-eye barrier near this point. Septate and gap junctions between glial membranes proliferate at this level, the lacunae disappear, and the axonal clefts narrow, but no tight junctions were seen. Comparison is made with the barrier around the nerve cord. (6) The secondary pigment cells in the retina may function as osmotic/ionic buffers, in conjunction with the blood-eye barrier.
Similar content being viewed by others
References
Adamson, A.W.: The diffusion and self-diffusion of electrolytes and hydration effects. J. Phys. Chem. 58, 514–523 (1954)
Bader, C.R., Baumann, F., Bertrand, D.: Role of intracellular calcium and sodium in light adaptation in the retina of the honey bee drone (Apis mellifera, L.). J. gen. Physiol. 67, 475–491 (1976)
Baskin, D.G.: The fine structure of polychaete septate junctions. Cell Tiss. Res. 174, 55–67 (1976)
Baumann, F., Hadjilazaro, B.: A depolarizing aftereffect of intense light in the drone visual receptor. Vision Res. 12, 17–31 (1976)
Bernard, F.: Recherches sur la morphogènese des yeux composés d'arthropodes. Bull. biol. Fr.-Belg. 23, Suppl., 1–162 (1937)
Bernays, E.A., Chapman, R.F.: Changes in haemolymph osmotic pressure in Locusta migratoria (L.) nymphs in relation to feeding. J. Ent. A 48, 149–155 (1974a)
Bernays, E.A., Chapman, R.F.: The effect of haemolymph osmotic pressure on the meal size of nymphs of Locusta migratoria (L.). J. exp. Biol. 61, 473–480 (1974b)
Brown, H.M., Hagiwara, S., Koike, H., Meech, R.W.: Membrane properties of a barnacle photoreceptor examined by the voltage clamp technique. J. Physiol. (Lond.) 208, 385–413 (1970)
Brown, J.E., Mote, M.I.: Ionic dependence of reversal voltage of the light response in Limulus ventral photoreceptors. J. gen. Physiol. 63, 337–350 (1974)
Eley, S., Shelton, P.M.J.: (1976) Cell junctions in the developing compound eye of the desert locust Schistocerca gregaria. J. Embryol. exp. Morphol. 36, 409–423 (1976)
Franceschini, N.: Sampling of the visual environment by the compound eye of the fly: fundamentals and applications. In: Photoreceptor optics, pp. 98–125 (A.W. Snyder and R. Menzel, eds.). Berlin-Heidelberg-New York: Springer 1975
Fulpius, B., Baumann, F.: Effects of sodium, potassium and calcium ions on slow and spike potentials in single photoreceptor cells. J. gen. Physiol. 53, 541–561 (1969)
Gemne, G.: Crystalline regions on the axon membrane surface of insect photoreceptor cells. Life Sci. 7, 1239–1243 (1968)
Gilula, N.B.: Junctions between cells, Chap. I of Cell communication, pp. 1–29 (R. Cox, ed.). New York: Wiley 1974
Glauert, A.M.: Fixation, dehydration and embedding of biological specimens. Practical methods in electron microscopy. Amsterdam: North Holland/American Elsevier 1975
Goldsmith, T.H.: Fine structure of the retinulae in the compound eye of the honeybee. J. Cell Biol. 14, 489–494 (1962)
Grossman, R.G., Seregin, A.: Glial-neural interaction demonstrated by the injection of Na+ and Li+ into cortical glia. Science 195, 196–198 (1977)
Hamdorf, K., Schwemer, J.: Photoregeneration and the adaptation process in insect photoreceptors. In: Photoreceptor optics, pp. 263–289 (A.W. Snyder and R. Menzel, eds.). Berlin-Heidelberg-New York: Springer (1975)
Heisenberg, M.: Separation of receptor and lamina potentials in the electroretinogram of normal and mutant Drosophila. J. exp. Biol. 55, 85–100 (1971)
Horridge, G.A.: The retina of the locust. In: The functional organization of the compound eye, pp. 513–541 (C.G. Bernhard, ed.). New York-London: Pergamon Press 1966
Horridge, G.A., Barnard, P.B.T.: Movement of palisade in locust retinula cells when illuminated. Quart. J. micr. Sci. 106, 131–135 (1965)
Hoyle, G.: Potassium ions and insect nerve muscle. J. exp. Biol. 30, 121–135 (1953)
Jacobs, M.H.: Diffusion processes. Ergebn. Biol. 12, 1–160 (1935)
Krebs, W.: Area and volume relationship in the crayfish retina. Vision Res. 14, 441–442 (1974)
Krebs, W., Helrich, C.S., Wulff, V.J.: The role of restricted extracellular compartments in vision. Vision Res. 15, 767–770 (1975)
Lane, N.J., Treherne, J.E.: Studies on perineurial junctional complexes and the sites of uptake of microperoxidase and lanthanum in the cockroach central nervous system. Tissue & Cell 4, 427–436 (1972)
Lasansky, A., Fuortes, M.G.F.: The site of origin of electrical responses in visual cells of the leech, Hirudo medicinalis. J. Cell Biol. 42, 241–252 (1969)
Leslie, R.A.: The effects of ionic lanthanum and hypertonic physiological salines on the nervous systems of larval and adult stick insects. J. Cell Sci. 18, 271–286 (1975)
Luft, J.H.: Ruthenium red and violet. I. Chemistry, purification methods of use for electron microscopy and mechanism of action. Anat. Rec. 171, 347–369 (1971)
McLaughlin, B.J.: The accessibility of a developing lepidopteran nervous system to lanthanum and peroxidase. J. Cell Sci. 14, 389–409 (1974)
Meinertzhagen, I.A.: The organization of perpendicular fibre pathways in the insect optic lobe. Phil. Trans. B 274, 555–596 (1976)
Millechia, R., Mauro, A.: The ventral photoreceptor cells of Limulus. II. The basic photoresponse. J. gen. Physiol. 54, 310–330 (1969)
Mote, M.I.: Electrical correlates of neural superposition in the eye of the fly Sarcophaga bullata. J. exp. Zool. 175, 159–168 (1970)
Perrelet, A., Baumann, F.: Evidence for extracellular space in the rhabdome of the honeybee drone eye. J. Cell Biol. 40, 825–830 (1969)
Ribi, W.A.: Fine structure of the first optic ganglion (lamina) of the cockroach, Periplaneta americana. Tissue & Cell 9, 57–72 (1977)
Ruck, P.: Retinal structures and photoreception. Ann. Rev. Entomol. 9, 83–102 (1964)
Satir, P., Gilula, N.B.: The fine structure of membranes and intercellular communication in insects. Ann. Rev. Entomol. 18, 143–166 (1973)
Schiff, H., Gervasio, A.: Functional morphology of the Squilla retina. Pubbl. Staz. zool. Napoli 37, 610–629 (1969)
Shaw, S.R.: Interreceptor coupling in ommatidia of drone honeybee and locust compound eyes. Vision Res. 9, 999–1029 (1969)
Shaw, S.R.: Retinal resistance barriers and electrical lateral inhibition. Nature (Lond.) 255, 480–483 (1975)
Shaw, S.R.: Neural effects of extracellular fields in insect eyes. Association for Research in Vision and Ophthalmology, Sarasota Meeting Abstracts, p. 17 (1976)
Shaw, S.R.: Restricted diffusion and extracellular space in the insect retina. J. comp. Physiol. 113, 257–282 (1977a)
Shaw, S.R.: Blood-brain barrier and extracellular space in the insect eye. Proc. Austral. physiol. pharmacol. Soc. 8, 90P (1977b)
Shivers, R.R.: “Tight” junctions in the sheath of normal and regenerating motor nerves of the crayfish, Orconectes virilis. Cell Tiss. Res. 177, 475–480 (1977)
Skaer, H.le B., Lane, N.J.: Junctional complexes, perineurial and glial-axonal relationships and the ensheathing structures of the insect nervous system; a comparative study using conventional and freeze-cleaving techniques. Tissue & Cell 6, 695–718 (1974)
Treherne, J.E.: The environment and function of insect nerve cells. In: Insect neurobiology (J.E. Treherne, ed.), pp. 187–244. Amsterdam: North Holland Publishing Co. (1974)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shaw, S.R. The extracellular space and blood-eye barrier in an insect retina: An ultrastructural study. Cell Tissue Res. 188, 35–61 (1978). https://doi.org/10.1007/BF00220513
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00220513