Abstract
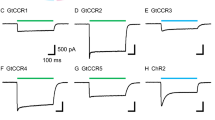
The study aimed to analyze the role and sources of calcium ions during the retinal light response in the freshwater pond snail Lymnaea stagnalis. We explored the effect of the following pharmacological agents modulating intracellular Ca2+ concentration on the electroretinogram (ERG) of an isolated eye: 2-aminoethyl diphenyl borate, a Ca2+ current modulator; EGTA, a Ca2+ chelator; (+)-cis-dilthiazem and Cd2+, Ca2+ channel blockers. All these agents suppressed the slow ERG wave and associated impulse activity with varying degrees of effectiveness and reversibility. It was concluded that light response of microvillar photoreceptors in L. stagnalis is accompanied by an elevation of the cytoplasmic Ca2+ concentration. Together with ultrastructural features of photoreceptors, our results suggest that during the light response calcium ions are not only released from the apical intracellular Ca2+ stores, but, at least in part, enter from the extracellular medium.







Similar content being viewed by others
REFERENCES
Yau KW, Hardie RC (2009) Phototransduction motifs and variations. Cell 139(2):246–264. https://doi.org/10.1016/j.cell.2009.09.029
Abbas F, Vinberg F (2009) Transduction and adaptation mechanisms in the cilium or microvilli of photoreceptors and olfactory receptors from insects to humans. Front Cell Neurosci 15:662453. https://doi.org/10.3389/fncel.2021.662453
Katz B, Minke B (2009) Drosophila photoreceptors and signaling mechanisms. Front Cell Neurosci 3:2. https://doi.org/10.3389/neuro.03.002.2009
Fu Y, Yau KW (2007) Phototransduction in mouse rods and cones. Pflugers Arch 454(5):805-819. https://doi.org/10.1007/s00424-006-0194-y
Nasi E, del Pilar Gomez M, Payne R (2000) Phototransduction mechanisms in microvillar and ciliary photoreceptors of invertebrates. In: Stavenga DG, DeGrip WJ, Pugh EN (eds). Handbook of Biological Physics. V.3. Elsevier Science BV, North-Holland. 389–448. https://doi.org/10.1016/S1383-8121(00)80011-1
Fain GL, Hardie R, Laughlin SB (2010) Phototransduction and the evolution of photoreceptors. Curr Biol 20(3): R114–R124. https://doi.org/10.1016/j.cub.2009.12.006
Arendt D, Tessmar-Raible K, Snyman H, Dorresteijn AW, Wittbrodt J (2004) Ciliary photoreceptors with a vertebrate-type opsin in an invertebrate brain. Science 306:869–871. https://doi.org/10.1126/science.1099955
Gotow T, Nishi T (2009) A new photosensory function for simple photoreceptors, the intrinsically photoresponsive neurons of the sea slug Onchidium. Front Cell Neurosci 3:18. https://doi.org/10.3389/neuro.03.018.2009
Kartelija G, Nedeljkovic M, Radenovic L (2003) Photosensitive neurons in mollusks. Comp Biochem Physiol A 134(3):483–495. https://doi.org/10.1016/s1095-6433(02)00351-3
Katagiri Y, Katagiri Y (2008) A multiple photoreceptive system in a marine gastropod, Onchidium: 1) Morphological characteristics and photoresponse of four kinds of photoreceptor cells. Hikaku seiri seikagaku (Comparative Physiology and Biochemistry) 25(1):4–10. https://doi.org/10.3330/hikakuseiriseika.25.4
Sudoplatov KA, Zhukov VV (1999) Electrical responses of peripheral nerves of the mollusc Lymnaea stagnalis to photostimulation of skin surface. J Evol Biochem Physiol 35(4):360–369. https://www.researchgate.net/publication/289523282
Sakakibara M, Aritaka T, Iizuka A, Suzuki H, Horikoshi T, Lukowiak K (2005) Electrophysiological responses to light of neurons in the eye and statocyst of Lymnaea stagnalis. J Neurophysiol. 93(1):493–507. https://doi.org/10.1152/jn.00692.2004
Zhukov VV, Fedorenko AD, Lavrova AI, Postnikov EB (2017) Electrical responses of Lymnaea stagnalis to light stimulation: Effect of divalent cations. J Evol Biochem Physiol 53(5):404–413. https://doi.org/10.1134/S0022093017050064
Takigami S, Sunada H, Horikoshi T, Sakakibara M (2014) Morphological and physiological characteristics of dermal photoreceptors in Lymnaea stagnalis. Biophysics (Nagoya-shi) 10:77–88. https://doi.org/10.2142/biophysics.10.77
Sunada H, Sakaguchi T, Horikoshi T, Lukowiak K, Sakakibara M (2010) The shadow-induced withdrawal response, dermal photoreceptors, and their input to the higher-order interneuron RPeD11 in the pond snail Lymnaea stagnalis. J Exp Biol 213(20):3409–3415. https://doi.org/10.1242/jeb.043521
Pankey S, Sunada H, Horikoshi T, Sakakibara M (2010) Cyclic nucleotide-gated channels are involved in phototransduction of dermal photoreceptors in Lymnaea stagnalis. J Comp Physiol B 180(8):1205–1211. https://doi.org/10.1007/s00360-010-0490-x
Stoll CJ (1973) Observations on the ultrastructure of the eye of the basommatophoran snail Lymnaea stagnalis (L.). Proc Kon Ned Akad Wet 76:414–424. https://eurekamag.com/research/023/225/023225797.php
Bobkova MV, Gál J, Zhukov VV, Shepeleva IP, Meyer-Rochow VB (2004) Variations in the Retinal Designs of Pulmonate Snails (Mollusca, Gastropoda): Squaring Phylogenetic Background and Ecophysiological Needs (I). Invertebrate Biology 123(2):101–115. https://doi.org/10.1111/j.1744-7410.2004.tb00146.x
Zhukov VV, Saphonov MV (2020) Activation of IP3 receptors is a component of phototransduction in gastropods retina. J Evol Biochem Physiol 56(7):811. https://doi.org/10.31857/S0044452920072978
Maruyama T, Kanaji T, Nakade S, Kanno T, Mikoshiba K (1997) 2APB, 2-aminoethoxydiphenyl borate, a membrane-penetrable modulator of Ins(1,4,5)P3-induced Ca2+ release. J Biochem 122(3):498–505. https://doi.org/10.1093/oxfordjournals.jbchem.a021780
Lansman JB, Hess P, Tsien RW (1986) Blockade of current through single calcium channels by Cd2+, Mg2+, and Ca2+. Voltage and concentration dependence of calcium entry into the pore. J Gen Physiol 88(3):321–347. https://doi.org/10.1085/jgp.88.3.321
Dobrev D, Milde AS, Andreas K, Ravens U (1999) The effects of verapamil and diltiazem on N-, P- and Q-type calcium channels mediating dopamine release in rat striatum. Br J Pharmacol 127(2):576-582. https://doi.org/10.1038/sj.bjp.0702574
Laryushkin DP, Maiorov SA, Zinchenko VP, Gaidin SG, Kosenkov AM (2021) Role of L-Type Voltage-Gated Calcium Channels in Epileptiform Activity of Neurons. Int J Mol Sci 22(19):10342. https://doi.org/10.3390/ijms221910342.
Leybaert L (2016) IP3, still on the move but now in the slow lane. Sci Signal 9(453):fs17. https://www.science.org/doi/10.1126/scisignal.aal1929
Wang Y, Deshpande M, Payne R (2002) 2-Aminoethoxydiphenyl borate inhibits phototransduction and blocks voltage-gated potassium channels in Limulus ventral photoreceptors. Cell Calcium 32(4):209–216. https://doi.org/10.1016/S0143416002001562
Montell C (2012) Drosophila visual transduction. Trends Neurosci 35(6):356–363. https://doi.org/10.1016/j.tins.2012.03.004
Hardie RC (2012) Phototransduction mechanisms in Drosophila microvillar photoreceptors. WIREs Membr Transp Signal 1:162–187. https://doi.org/10.1002/wmts.20
del Pilar Gomez M, Nasi E (2009) Prolonged calcium influx after termination of light-induced calcium release in invertebrate photoreceptors. J Gen Physiol 134(3):177–189. https://doi.org/10.1085/jgp.200910214
Van Kerkhove E, Pennemans V, Swennen Q (2010) Cadmium and transport of ions and substances across cell membranes and epithelia. Biometals 23(5):823-855. https://doi.org/10.1007/s10534-010-9357-6
Bootman MD, Collins TJ, Mackenzie L, Roderick HL, Berridge MJ, Peppiatt CM (2002) 2-aminoethoxydiphenyl borate (2-APB) is a reliable blocker of store-operated Ca2+ entry but an inconsistent inhibitor of InsP3-induced Ca2+ release. FASEB J 16(10):1145–1150. https://doi.org/10.1096/fj.02-0037rev
Prakriya M, Lewis RS (2001) Potentiation and inhibition of Ca(2+) release-activated Ca(2+) channels by 2-aminoethyldiphenyl borate (2-APB) occurs independently of IP(3) receptors. J Physiol 536 (Pt 1):3–19. https://doi.org/10.1111/j.1469-7793.2001.t01-1-00003.x
Dickinson GD, Ellefsen KL, Dawson SP, Pearson JE, Parker I (2016) Hindered cytoplasmic diffusion of inositol trisphosphate restricts its cellular range of action. Sci Signal 9(453):ra108. https://doi.org/10.1126/scisignal.aag1625
Zhukov VV (2007) On the problem of retinal transmitters of the freshwater mollusc Lymnaea stagnalis. J Evol Biochem Phys 43(5):524–532. https://doi.org/10.1134/S0022093007050118
Zylstra U (1972) Distribution and ultrastructure of epidermal sensory cells in the freshwater snail Lymnaea stagnalis and Biomphalaria pheifferi. Neth J Zool 22:283–298. https://doi.org/10.1163/002829672X00103
ACKNOWLEDGMENT
The authors are grateful Prof. D. von Keyserling and Mrs. A. Agbedor (Institute of Anatomy, Medical Faculty, RWTH, Aachen) for the opportunity and assistance in carrying out electron microscopy.
Funding
M.V.S. was supported by the Baltic Federal University grant for graduate students (No. 879, 12-10-2021) and a starting grant for young scientists (program 5-100).
Author information
Authors and Affiliations
Contributions
Conceptualization and experimental design (V.V.Zh.), data collection (V.V.Zh. and M.V.S.), data processing (V.V.Zh. and M.V.S.), writing and editing a manuscript (V.V.Zh. and M.V.S.).
Corresponding author
Ethics declarations
CONFLICT OF INTEREST
The authors declare that they have no conflict of interest that might be associated with the publication of this article.
Additional information
Translated by A. Polyanovsky
Russian Text © The Author(s), 2022, published in Zhurnal Evolyutsionnoi Biokhimii i Fiziologii, 2022, Vol. 58, No. 3, pp. 196–208https://doi.org/10.31857/S004445292203007X.
Rights and permissions
About this article
Cite this article
Zhukov, V.V., Saphonov, M.V. Calcium Component of the Retinal Light Response in the Snail Lymnaea stagnalis: a Pharmacological and Ultrastructural Study. J Evol Biochem Phys 58, 652–665 (2022). https://doi.org/10.1134/S0022093022030036
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022093022030036