Abstract
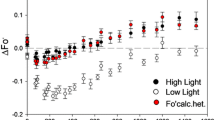
The effective absorption cross section of inactive photosystem II (PS II) centers, which is the product of the effective antenna size and the quantum yield for photochemistry, was investigated by comparing the light saturation curves of inactive PS II and active reaction centers in intact chloroplasts and thylakoid membranes of spinach (Spinacia oleracea). Inactive PS II centers are defined as the impaired PS II reaction centers that require greater than 50 ms for the reoxidation of QA − subsequent to a single turnover flash. Active reaction centers are defined as the rapidly turning over PS II centers (recovery time less than 50 ms) and all of the PS I centers. The electrochromic shift, measured by the flash-induced absorbance increase at 518 nm, was used to probe the activity of the reaction centers. Light saturation curves were generated for inactive PS II centers and active reaction centers by measuring the extent of the absorbance increase at 518 nm induced by red actinic flashes of variable energy. The light saturation curves show that inactive PS II centers required over twice as many photons as active reaction centers to achieve the same yield. The ratio of the flash energy required for 50% saturation for active reaction centers (PS II active + PS I) compared to inactive PS II centers was 0.45±0.04 in intact chloroplasts, and 0.54±0.11 in thylakoid membranes. Analysis of the light saturation curves using a Poisson statistical model in which the ratio of the antenna size of active PS II centers to that of PS I is considered to range from 1 to 1.5, indicates that the effective absorption cross section of inactive PS II centers was 0.54–0.37 times that of active PS II centers. If the quantum yield for photochemistry is assumed to be one, we estimate that the antenna system serving the inactive PS II centers contains approx. 110 chlorophyll molecules.
Similar content being viewed by others
Abbreviations
- BSA:
-
bovine serum albumin
- DCMU:
-
3[3,4-dichlorophenyl]-1,1dimethylurea
- Hepes:
-
4-(2-hydroxyethyl)-1-piperazineethanesufonic acid
- P680:
-
primary donor in photosystem II
- QA :
-
primary quinone acceptor in photosystem II
- QB :
-
secondary quinone acceptor in photosystem II
References
Anderson J and Melis A (1983) Localization of different photosystems in separate regions of chloroplast membranes. Proc Natl Acad Sci 80: 745–749
Andreasson E, Svwensson P, Claes W and Albertsson P (1988) Separation and characterization of stroma and grana membranes evidence for heterogeneity in antenna size of both Photosystem I and Photosystem II. Biochim Biophys Acta 936: 339–350
Armond PA and Arntzen CJ (1977) Localization and characterization of photosystem II in grana and stroma lamellae. Plant Physiol 59: 398–404
Björkman O and Demmig B (1987) Photon yield of O2 evolution and chlorophyll fluorescence at 77 K among vascular plants of diverse origins. Planta 170: 489–504
Black MT, Brearly TH and Horton P (1986) Heterogeneity in chloroplast photosystem II. Photosynth Res 8: 193–207
Cho HM, Mancino LJ and Blankenship RE (1984) Light saturation curves and quantum yields in reaction centers from photosynthetic bacteria. Biophys J 45: 455–461
Chylla RA and Whitmarsh J (1989) Inactive Photosystem II Complexes in Leaves: Turnover Rate and Quantitation. Plant Phys 90: 765–772
Chylla RA and Whitmarsh J (1990) Measurement of the complete oxidation kinetics of QA − in spinach leaves using flash fluorescence. Proc. VIIIth International Congress on Photosynthesis (ed) M. Baltscheffsky. In press
Chylla RA, Garab G and Whitmarsh J (1987) Evidence for slow turnover in a fraction of photosystem II complexes in thylakoid membranes. Biochim Biophys Acta 894: 562–571
Diner BA and Petrouleas V (1987) Q400, the non-heme iron of the photosystem II iron-quinone complex. A spectroscopic probe of the quinone and inhibitor binding to the reaction center. Biochim Biophys Acta 895: 107–125
Emerson R and Arnold W (1932) The photochemical reaction in photosynthesis. J Gen Physiol 16: 191–205
Ghirardi ML and Melis A (1988) Chlorophyll b deficiency in soybean mutants. I. Effects on photosystem stoichiometry and chlorophyll antenna size. Biochim Biophys Acta 932: 130–137
Graan T (1986) The interaction of silicomolybdate with the photosystem II herbicide-binding site. FEBS Lett 206: 9–14
Graan T and Ort DR (1986) Detection of oxygen-evolving photosystem II centers inactive in plastoquinone reduction. Biochim Biophys Acta 852: 320–330
Guenther JE, Nemson JA and Melis A (1988) Photosystem stoichiometry and chlorophyll antenna size in Dunaliella salina (green algae). Biochim Biophys Acta 934: 108–117
Haehnel W, Pröpper A and Krause H (1980a) Evidence for complexed plastocyanin as the immediate electron donor of P700. Biochim Biophys Acta 593: 384–399
Haehnel W, Hesse V and Pröpper A (1980b) Electron transfer from plastocyanin to P700. FEBS Lett 111: 79–82
Jensen RG and Bassham JA (1966) Photosynthesis by isolated chloroplasts. Proc Natl Acad Sci 56: 1095–1101
Junge W (1964) Electrogenic reactions and proton pumping in green plant photosynthesis. Curr Topics in Membranes Transport 16: 431–465
Lavergne J (1982a) Two types of primary acceptor in chloroplasts photosystem II: I. Different recombination properties. Photobiochem Photobiophys 3: 257–271
Lavergne J (1982b) Two types of primary acceptor in chloroplasts photosystem II: II. Reduction in two successive photoacts. Photobiochem Photobiophys 3: 273–285
Lee WJ and Whitmarsh J (1989) The photosynethetic apparatus of pea thylakoid membranes: Response to growth light intensity. Plant Physiol 89: 932–940
Ley A and Mauzerall D (1982) Absolute absorption cross-sections for photosystem II and the minimum quantum requirement for photosynthesis in Chlorella vulgaris. Biochim Biophys Acta 680: 95–106
Mauzerall D (1982) Statistical theory of the effect of multiple excitation in photosynthetic systems. In: Biological Events Probed by Ultrafast Laser Spectroscopy. Alfano R (ed) pp 215–235. Academic Press, New York
Mauzerall D and Greenbaum NL (1989) The absolute size of a photosynthetic unit. Biochim Biophys Acta 974: 119–140
Mäempää P, Andersson B and Sundby C (1987) Difference in sensitivity to photoinhibition between photosystem II in appressed and non-appressed thylakoid regions. FEBS Lett 215: 31–36
Melis A (1985) Functional properties of Photosystem IIβ in spinach chloroplasts. Biochim Biophys Acta 808: 334–342
Melis A and Anderson JM (1983) Structural and functional organization of the photosystems in spinach chloroplasts: antenna size, relative-electron transport capacity, and chlorophyll composition. Biochim Biophys Acta 724: 473–484
Melis A and Duysens LNM (1979) Biphasic energy conversion kinetics and absorbance difference spectra of photosystem II of chloroplasts. Evidence for two different photosystem II reaction centers. Photochem Photobiol 29: 373–382
Melis A and Homann PH (1975) Kinetic analysis of the fluorescence induction in 3-(3,4-dichlorophenyl)-1,1-dimethylurea poisoned chloroplasts. Photochem Photobiol 21: 431–437
Ort DR and Whitmarsh J (1989) Inactive photosystem II centers: A resolution of discrepencies in photosystem II quantitation? Photosynth Res 23: 101–104
Percival MP, Webber AN and Baker NR (1984) Evidence for role of the light-harvesting chlorophyll a/b protein complex in photosystem II heterogeneity. Biochim Biophys Acta 767: 582–589
Robinson HH and Crofts AR (1983) Kinetics of the oxidation-reduction reactions of the photosystem II quinone acceptor complex, and the pathway for deactivation. FEBS Lett 153: 221–226
Spencer D and Wildman SG (1962) Observations on the structure of grana-containing chloroplasts and a proposed model of chloroplast structure. Aust J Biol Sci 15: 599–610
Thielen APGM and van Gorkom HJ (1981a) Quantum efficiency and antenna size of photosystem IIα, IIβ and I in tobacco chloroplasts. Biochim Biophys Acta 635: 111–120
Thielen APGM and van Gorkom HJ (1981b) Energy transfer and quantum yield in photosystem II. Biochim Biophys Acta 637: 439–446
Thielen APGM and van Gorkom HJ (1981c) Redox potentials of electron acceptors in photosystem IIα and photosystem IIβ. FEBS Lett 129: 205–209
Weaver EC and Weaver HE (1969) Paramagnetic unit in spinach subchloroplast particles: Estimation of size. Science 165: 906–907
Whitmarsh J and Ort DR (1984) Stoichiometry of electron transfer complexes in spinach chloroplasts. Arch Biochem Biophys 231: 378–389
Wong D, Govindjee and Jursinic P (1974) Analysis of microsecond fluorescence yield and delayed light emission changes after a single flash in pea chloroplasts: Effect of mono- and divalent cations. Photochem Photobiol 28: 963–974
Ziegler R and Egle K (1965) Zur quantitativen analyse der Chloroplastenpigmente I. Kritische Uberprufung der Spektralphotometrischen Chlorophyll-Bestimmung. Beitr Biol Pflanz 4: 11–37
Author information
Authors and Affiliations
Additional information
Photosynthesis Research Unit, USDA, Agricultural Research Service, 289 Morrill Hall, 505 S. Goodwin Ave., Urbana, Illinois 61801, USA
Rights and permissions
About this article
Cite this article
Chylla, R.A., Whitmarsh, J. Light saturation response of inactive photosystem II reaction centers in spinach. Photosynth Res 25, 39–48 (1990). https://doi.org/10.1007/BF00051734
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00051734