Abstract
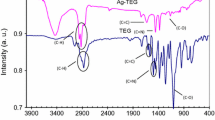
A convenient and uncomplicated scheme has been projected for the quantitative determination of essential diamines putrescine (PUT) and cadaverine (CAD) via sodium dodecyl sulfate protected silver nanoparticles (SDS-AgNPs). This scheme is based on the chemical interaction of a SDS-AgNPs probe with PUT and CAD, leading to a color change from yellow to red or reddish brown. The interaction was investigated through different techniques such as using a UV-visible spectrophotometer, Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), energy dispersive X-ray spectroscopy (EDX), dynamic light scattering spectroscopy (DLS) and the zeta potential. Both amines possess a close resemblance in structure (except for the addition of one more methylene group in CAD), and no any distinguishable color change was noted. However, the maximum absorption band at 580 and 600 nm was demonstrated for PUT and CAD correspondingly. The methodical response was observed at absorption ratios of 580/410 and 600/410 nm, with the linear regression within 4–12 and 6–14 μg/mL for PUT and CAD. The detection limits calculated for both the diamines PUT and CAD were 0.333 and 1.638 μg/mL. The scheme was successfully applied for determinations in biological samples, including spiked blood plasma and urine. Putrescine exhibited % recovery within 95.717–105.200%, while cadaverine was within 95.940–105.109%, respectively. The scheme was reproducible and precise with inter-day RSD (n = 5) within 1.126, 0.018% and the intraday RSD (n= 5) was within 0.005, 0.002% for PUT and CAD, respectively.
Similar content being viewed by others
References
B. D. RioLargar, B. Redruello, D. M. Linares, V. Ladero, P. Ruas-Madiedo, M. Fernandez, and M. A. Alvarez, Sci. Rep., 2019, 9, 1.
A. Önal, Food Chem., 2007, 103, 1475.
A. Halàsz, A. Barath, L. Simon-Sarkadi, and W. Holzapfel, Trends Food Sci. Technol., 1994, 5, 42.
V. Ladero, M. Coton, M. Fernández, N. Buron, M. C. Martin, H. Guichard, E. Coton, and M. A. Alvarez, Food Microb., 2011, 28, 554.
M. Marino, M. Maifreni, S. Moret, and G. Rondinini, Lett. Appl. Microb., 2000, 31, 169.
L. Prester, Food Addit. Contam., 2011, 28, 1547.
G. Suzzi and F. Gardini, Int. J. Food Microb., 2003, 88, 41.
B. Ten Brink, C. Damink, H. M. L. J. Joosten, and J. H. J. Huis In’t Veld, Int. J. Food Microb., 1990, 11, 73.
N. Jornet-Martinez, M. González-Béjar, Y. Moliner-Martínez, P. Campins-Falco, and J. Pérez-Prieto, Anal. Chem., 2014, 86, 1347.
Y. Chen, J. Zhang, Y. Gao, J. Lee, H. Chen, and Y. Yin, Biosens. Bioelectron., 2015, 72, 306.
M. Y. Khuhawar and G. A. Qureshi, J. Chromatogr. B, 2001, 764, 385.
L. F. Bjeldanes, D. E. Schutz, and M. M. Morris, Food Cosmet. Toxicol., 1978, 16, 157.
J. Donthuan, S. Yunchalard, and S. Srijaranai, J. Sep. Sci., 2014, 37, 3164.
A. Calvo-Pérez, O. Domínguez-Renedo, M. A. Alonso-Lomillo, and M. J. Arcos-Martínez, Microchim. Acta, 2013, 180, 253.
M. Saaid, B. Saad, N. H. Hashim, A. S. M. Ali, and M. I. Saleh, Food Chem., 2009, 113, 1356.
M. Mohammadi, M. Kamankesh, Z. Hadian, A. M. Mortazavian, and A. Mohammadi, Chromatographia, 2017, 80, 119.
M. Y. Khuhawar, A. A. Memon, P. D. Jaipal, and M. I. Bhanger, J. Chromatogr. B, 1999, 723, 17.
J. Plotka-Wasylka, V. Simeonov, and J. Namiesnik, J. Chromatogr. A, 2016, 1453, 10.
J. Lapa-Guimarães and J. Pickova, J. Chromatogr. A, 2004, 1045, 223.
M. S. Steiner, R. J. Meier, C. Spangler, A. Duerkop, and O. S. Wolfbeis, Microchim. Acta, 2009, 67, 259.
V. Valderrey, A. Bonasera, S. Fredrich, and S. Hecht, Angew. Chem. Int. Ed., 2017, 56, 1914.
J. Tu, S. Sun, and Y. Xu, Chem. Commun., 2016, 52, 1040.
K. M. A. El-Nour, E. T. A. Salam, H. M. Soliman, and A. S. Orabi, Nanoscale Res. Lett., 2017, 12, 231.
Y. He, X. E. Zhao, R. Wang, N. Wei, J. Sun, J. Dang, G. Chen, Z. Liu, S. Zhu, and J. You, J. Agric. Food Chem., 2016, 64, 8225.
R. Romero-Gonzalez, M. I. Alarcon-Flores, J. L. M. Vidal, and A. G. Frenich, J. Agric. Food Chem., 2012, 60, 5324.
R. L. Self and W. H. Wu, J. Food Compost. Anal., 2012, 27, 169.
A. Önal, S. E. K. Tekkeli, and C. Önal, Food Chem., 2013, 138, 509.
R. Zhang, N. Li, J. Sun, and F. Gao, J. Agric. Food Chem., 2015, 63, 8947.
W. Zhang, Y. Tang, J. Liu, L. Jiang, W. Huang, F. W. Huo, and D. Tian, J. Agric. Food Chem., 2015, 63, 39.
H. Haick, Y. Y. Broza, P. Mochalski, V. Ruzsanyi, and A. Amann, Chem. Soc. Rev., 2014, 43, 1423.
Y. Y. Broza and H. Haick, Nanomedicine—UK, 2013, 8, 785.
A. P. Turner and N. Magan, Nat. Rev. Microbiol., 2004, 2, 161.
S. A. Ghoto, M. Y. Khuhawar, and T. M. Jahangir, Anal. Sci., 2019, 35, 631.
N. Kumar and R. N. Goyal, Sens. Actuators, B, 2018, 268, 383.
A. C. Burdusel, O. Gherasim, A. M. Grumezescu, L. Mogoanta, A. Ficai, and E. Andronescu, Nanomaterials, 2018, 8, 681.
F. Porcaro, L. Carlini, A. Ugolini, D. Visaggio, P. Visca, I. Fratoddi, I. Venditti, C. Menegini, L. Simon Elliott, C. Mari I, W. Olszewski, N. Ramanan, I. Luisetto, and C. Battocchio, Materials, 2016, 9, 1028.
M. Bordoloi, R. K. Sahoo, J. Tamuli, S. Saikia, and P. P. Dutta, Nano, 2020, 15, Article number 2030001.
L. Carlini, C. Fasolato, P. Postorino, I. Fratoddi, I. Venditte, G. Testa, and C. Battocchio, Colloids Surf., A, 2017, 532, 183.
P. Prosposito, L. Burratti, A. Bellingeri, G. Protano, C. Valerie, I. Corsi, C. Battocchio, G. Iucci, L. Tortola, V. Secchi, S. Franchi, and I. Venditti, Nanomaterials, 2019, 9, 1353.
A. Centeno, S. Rahmah Aid, and F. Xie, Chemosensors, 2018, 6, 4.
Q. Y. Chen, S. L. Xiao, S. Q. Shi, and L. P. Cai, Polymers, 2020, 12, 440.
P. Prosposito, L. Burratti, and I. Venditti, Chemsensors, 2020, 8, 26.
M. L. Firdaus, A. Parian, N. Meileza, M. Hitsmi, and R. Elvia, Chemosensors, 2019, 7, 25.
K. C. Song, S. M. Lee, T. S. Park, and B. S. Lee, Korean J. Chem. Eng., 2009, 26, 153.
L. Qi, Z. Xu, X. Jiang, C. Hu, and X. Zou, Carbohydr. Res., 2004, 339, 2693.
S. A. Ghoto, M. Y. Khuhawar, and T. M. Jahangir, Journal of Nanostructure in Chemistry, 2019, 9, 77.
S. Ribeiro, N. Hussain, and A. T. Florence, Int. J. Pharm., 2005, 298, 354.
N. Seiler, “Selected Topics from Neurochemistry”, 1985, Pergamon, 147.
C. Yuan, R. Liu, S. Wang, G. Han, M. Y. Han, C. Jiang, and Z. Zhang, J. Mater. Chem., 2011, 21, 16264.
K. A. Rawat, J. R. Bhamore, R. K. Singhal, and S. K. Kailasa, Biosens. Bioelectron., 2017, 88, 71.
J. Wang, Z. L. Wu, H. Z. Zhang, Y. F. Li, and C. Z. Huang, Talanta, 2017, 167, 193.
S. S. Gowda, S. Rajasowmiya, V. Vadivel, S. B. Devi, A. C. Jerald, S. Marimuthu, and N. Devipriya, Toxicol. in Vitro, 2018, 52, 170.
A. Contino, G. Maccarrone, M. Zimbone, R. Reitano, P. Musumeci, L. Calcagno, and I. P. Oliveri, J. Colloid Interface Sci., 2016, 462, 216.
X. Jiang and A. Yu, J. Nanosci. Nanotechnol., 2010, 10, 7643.
H. Li and Y. Bian, Nanotechnology, 2009, 20, 145502.
C. Yuan, R. Liu, S. Wang, G. Han, M. Y. Han, C. Jiang, and Z. Zhang, J. Mater. Chem., 2011, 21, 16264.
D. S. Singare, S. Marella, K. Gowthamrajan, G. T. Kulkarni, R. Vooturi, and P. S. Rao, Int. J. Pharm., 2010, 402, 213.
R. H. Muller, C. Jacobs, and O. Kayser, Adv. Drug Delivery Rev., 2001, 47, 3.
R. A. E. F. Hamouda, M. A. El-Mongy, and K. F. Eid, Int. J. Pharmacol., 2018, 14, 359.
D. E. Koppel, J. Chem. Phys., 1972, 57, 4814.
B. J. Berne and R. Pecora, “Dynamic Light Scattering with Applications to Chemistry, Biology and Physics”, 2000, Dover Publications Inc, Minneola.
Acknowledgments
We are highly thankful to the Institute of Advanced Research Studies in Chemical Sciences, University of Sindh Jamshoro, for supporting this Research project.
Author information
Authors and Affiliations
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Ghoto, S.A., Khuhawar, M.Y. Silver Nanoparticles for a Colorimetric Determination of Putrescine and Cadaverine in Biological Samples. ANAL. SCI. 37, 267–274 (2021). https://doi.org/10.2116/analsci.20P153
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2116/analsci.20P153