Abstract
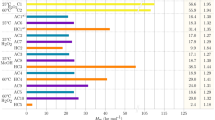
Embroidery industry wastewater contains high levels of polyvinyl alcohol (PVA), making it resistant to biodegradation and conventional treatment methods such as coagulation and adsorption. Chemical oxidation processes have been successfully used to treat PVA-containing effluents, but the high chemical demand raises significant environmental and sustainability concerns. Recent studies are focused on finding innovative and efficient chemical approaches to effectively treat PVA-containing effluents. This work used an electrooxidation process (EO) with boron-doped diamond electrodes to treat wastewater containing PVA for the first time in the literature. The effects of pH (2.0–8.0), temperature (25–45 °C), current (3.0–6.0 A), electrolyte concentration (NaCl of 3.0–8.0 g/L), and oxidation time (0–6000 min) on the removal of total organic Carbon (TOC) were investigated. The pollutant removal mechanism of the process was elucidated using high-performance size exclusion chromatography, Fourier-transform infrared, Ultraviolet–visible, and Fluorescence Spectroscopy methods. The results showed that TOC concentrations significantly dropped from 6584 to 159.10 mg/L after 10 h of treatment at 6.0 A, pH 5.01, and 25, with an estimated 97.6% removal efficiency. Characterization studies showed that much of the wastewater’s PVA is broken down, and the halogens are bound to the degraded part of the polymer. The raw wastewater used contained a wide range of pollutants (103–1010 Da). The results showed that the pollutants in the wastewater were successfully removed by EO irrespective of the molecular size. These results demonstrate the superiority of EO over treatment techniques that exhibit selectivity for pollutants with different molecular weights.
Graphical abstract









Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
Anglada A, Urtiaga A, Ortiz I (2009) Contributions of electrochemical oxidation to waste-water treatment: fundamentals and review of applications. J Chem Technol Biotechnol 84(12):1747–1755
Arslan H, Gun M, Akarsu C, Bilici Z, Dizge N (2023) Treatment of turnip juice wastewater by electrocoagulation/electroflotation and electrooxidation with aluminum, iron, boron-doped diamond, and graphite electrodes. Int J Environ Sci Technol 20(1):53–62. https://doi.org/10.1007/s13762-022-03994-3
Brycht M, Lochyński P, Barek J et al (2016) Electrochemical study of 4-chloro-3-methylphenol on anodically pretreated boron-doped diamond electrode in the absence and presence of a cationic surfactant. J Electroanal Chem 771:1–9. https://doi.org/10.1016/j.jelechem.2016.03.031
Can OT, Gengec E, Kobya M (2019) TOC and COD removal from instant coffee and coffee products production wastewater by chemical coagulation assisted electrooxidation. J Water Process Eng. https://doi.org/10.1016/j.jwpe.2019.01.002
Chaabene R, Khannous L, Samet Y (2022) Electrochemical degradation of aqueous metformin at boron-doped diamond electrode: kinetic study and phytotoxicity tests. Int J Environ Sci Technol. https://doi.org/10.1007/s13762-022-04325-2
Chen Y, Sun Z, Yang Y, Ke Q (2001) Heterogeneous photocatalytic oxidation of polyvinyl alcohol in water. J Photochem Photobiol A Chem 142:85–89. https://doi.org/10.1016/S1010-6030(01)00477-4
Chou WL, Wang CT, Huang KY (2010) Investigation of process parameters for the removal of polyvinyl alcohol from aqueous solution by iron electrocoagulation. Desalination 251:12–19. https://doi.org/10.1016/j.desal.2009.10.008
Dong Y, Bian L, Wang P (2019) Accelerated degradation of polyvinyl alcohol via a novel and cost effective heterogeneous system based on Na2S2O8 activated by Fe complex functionalized waste PAN fiber and visible LED irradiation. Chem Eng J 358:1489–1498
El-Bashir SM, Yahia IS, Binhussain MA, AlSalhi MS (2017) Designing of PVA/Rose Bengal long-pass optical window applications. Res Phys 7:1238–1244. https://doi.org/10.1016/j.rinp.2017.03.033
Ergan BT, Gengec E (2020) Dye degradation and kinetics of online electro-fenton system with thermally activated carbon fiber cathodes. J Environ Chem Eng 8:104217. https://doi.org/10.1016/j.jece.2020.104217
Gates FL (1934) The ultraviolet absorption spectrum of pepsin. J Gen Physiol 17:797–801. https://doi.org/10.1085/jgp.17.6.797
Gengec E (2015) Color removal from anaerobic/aerobic treatment effluent of bakery yeast wastewater by polyaniline/beidellite composite materials. J Environ Chem Eng. https://doi.org/10.1016/j.jece.2015.09.009
Gengec E (2017) Treatment of highly toxic cardboard plant wastewater by a combination of electrocoagulation and electrooxidation processes. Ecotoxicol Environ Saf. https://doi.org/10.1016/j.ecoenv.2017.07.032
Gengec E, Kobya M (2013) Treatment of Baker’s yeast wastewater by electrocoagulation and evaluation of molecular weight distribution with HPSEC. Sep Sci Technol 48:2880–2889. https://doi.org/10.1080/01496395.2013.804087
Gengec E, Ozdemir U, Ozbay B et al (2013) Optimizing dye adsorption onto a waste-derived (modified charcoal ash) adsorbent using box-behnken and central composite design procedures. Water Air Soil Pollut. https://doi.org/10.1007/s11270-013-1751-6
Gengec NA, Isgoren M, Kobya M et al (2018) Optimization of beidellite/polyaniline production conditions by central composite design for removal of acid yellow 194. J Polym Environ 26:2619–2631. https://doi.org/10.1007/s10924-017-1157-4
Guo Y, Lai B, Zhou YX (2016) Pretreatment of polyvinyl alcohol-containing desizing wastewater by the fenton process: oxidation and coagulation. Environ Eng Sci 33:160–166. https://doi.org/10.1089/EES.2015.0327
Hamad D, Mehrvar M, Dhib R (2014) Experimental study of polyvinyl alcohol degradation in aqueous solution by UV/H2O2 process. Polym Degrad Stab 103:75–82
Hamad D, Dhib R, Mehrvar M (2021) Identification and model predictive control (MPC) of aqueous polyvinyl alcohol degradation in UV/H2O2 photochemical reactors. J Polym Environ 29:2572–2584. https://doi.org/10.1007/s10924-020-02031-z
He Y, Wang X, Huang W et al (2016) Application of porous boron-doped diamond electrode towards electrochemical mineralization of triphenylmethane dye. J Electroanal Chem 775:292–298. https://doi.org/10.1016/j.jelechem.2016.06.023
Hurwitz G, Hoek EMV, Liu K et al (2014) Photo-assisted electrochemical treatment of municipal wastewater reverse osmosis concentrate. Chem Eng J 249:180–188. https://doi.org/10.1016/j.cej.2014.03.084
İskurt Ç, Aliyev E, Gengec E et al (2022) Electrochemical oxidation of pretreated landfill leachate nanofiltration concentrate in terms of pollutants removal and formation of by-products. Chemosphere 307:135954. https://doi.org/10.1016/J.CHEMOSPHERE.2022.135954
Jipa IM, Stoica A, Stroescu M et al (2012) Potassium sorbate release from poly(vinyl alcohol)-bacterial cellulose films. Chem Pap 66:138–143. https://doi.org/10.2478/s11696-011-0068-4
Kac FU, Kobya M, Gengec E (2017) Removal of humic acid by fixed-bed electrocoagulation reactor: studies on modelling, adsorption kinetics and HPSEC analyses. J Electroanal Chem 804:199–211. https://doi.org/10.1016/j.jelechem.2017.10.009
Karatas O, Gengec NA, Gengec E et al (2022) High-performance carbon black electrode for oxygen reduction reaction and oxidation of atrazine by electro-fenton process. Chemosphere 287:132370. https://doi.org/10.1016/j.chemosphere.2021.132370
Kuchtová G, Chýlková J, Váňa J et al (2020) Electro-oxidative decolorization and treatment of model wastewater containing acid Blue 80 on boron doped diamond and platinum anodes. J Electroanal Chem. https://doi.org/10.1016/j.jelechem.2020.114036
Liu Y, Geever LM, Kennedy JE et al (2010) Thermal behavior and mechanical properties of physically crosslinked PVA/Gelatin hydrogels. J Mech Behav Biomed Mater 3:203–209. https://doi.org/10.1016/j.jmbbm.2009.07.001
Mieloszyk J, Drabent R, Siódmiak J (1987) Phosphorescence and fluorescence of poly(vinyl alcohol) films. J Appl Polym Sci 34:1577–1580. https://doi.org/10.1002/app.1987.070340418
Neto SA, De AAR (2009) Electrooxidation of glyphosate herbicide at different DSA® compositions : pH, concentration and supporting electrolyte effect. Electrochim Acta 54:2039–2045. https://doi.org/10.1016/j.electacta.2008.07.019
Oh SY, Kim HW, Park JM et al (2009) Oxidation of polyvinyl alcohol by persulfate activated with heat, Fe2+, and zero-valent iron. J Hazard Mater 168:346–351. https://doi.org/10.1016/j.jhazmat.2009.02.065
Özcan A, Şahin Y, Koparal AS, Oturan MA (2008) Propham mineralization in aqueous medium by anodic oxidation using boron-doped diamond anode: influence of experimental parameters on degradation kinetics and mineralization efficiency. Water Res 42:2889–2898. https://doi.org/10.1016/j.watres.2008.02.027
Palahouane B, Drouiche N, Aoudj S, Bensadok K (2015) Cost-effective electrocoagulation process for the remediation of fluoride from pretreated photovoltaic wastewater. J Ind Eng Chem 22:127–131. https://doi.org/10.1016/j.jiec.2014.06.033
Pan Y, Liu Y, Wu D et al (2020) Application of Fenton pre-oxidation, Ca-induced coagulation, and sludge reclamation for enhanced treatment of ultra-high concentration poly(vinyl alcohol) wastewater. J Hazard Mater 389:121866. https://doi.org/10.1016/j.jhazmat.2019.121866
Panizza M, Cerisola G (2001) Removal of organic pollutants from industrial wastewater by electrogenerated Fenton’s reagent. Water Res 35:3987–3992. https://doi.org/10.1016/S0043-1354(01)00135-X
Prabhakaran D, Kannadasan T, Ahmed Basha C (2009) Treatability of resin effluents by electrochemical oxidation using batch recirculation reactor. Int J Environ Sci Technol 6:491–498. https://doi.org/10.1007/BF03326088
Raut AR, Murhekar GH (2014) International journal of chemical studies optical and morphological study of modified polyvinyl alcohol 2:19–24
Rolsky C, Kelkar V (2021) Degradation of polyvinyl alcohol in US wastewater treatment plants and subsequent nationwide emission estimate. Int J Environ Res Public Health 18(11):6027
Särkkä H, Bhatnagar A, Sillanpää M (2015) Recent developments of electro-oxidation in water treatment: a review. J Electroanal Chem 754:46–56. https://doi.org/10.1016/j.jelechem.2015.06.016
Scialdone O (2009) Electrochemical oxidation of organic pollutants in water at metal oxide electrodes: a simple theoretical model including direct and indirect oxidation processes at the anodic surface. Electrochim Acta 54:6140–6147. https://doi.org/10.1016/j.electacta.2009.05.066
Sires I, Brillas E, Oturan MA et al (2014) Electrochemical advanced oxidation processes: today and tomorrow. A review. Environ Sci Pollut Res 21:8336–8367. https://doi.org/10.1007/s11356-014-2783-1
Solaro R, Corti A, Chiellini E (2000) Biodegradation of poly(vinyl alcohol) with different molecular weights and degree of hydrolysis. Polym Adv Technol 11:873–878. https://doi.org/10.1002/1099-1581(200008/12)11:8/12%3c873::AID-PAT35%3e3.0.CO;2-V
Song Q, Chen X, Tang L, Zhou W (2021) Treatment of polyvinyl alcohol containing wastewater in two stage spiral symmetrical stream anaerobic bioreactors coupled a sequencing batch reactor. Bioresour Technol 340:125702
Soni BD, Patel UD, Agrawal A, Ruparelia JP (2020) Electrochemical destruction of RB5 on Ti/PtOx–RuO2–SnO2–Sb2O5 electrodes: a comparison of two methods for electrode preparation. Int J Environ Sci Technol 17:903–916. https://doi.org/10.1007/s13762-019-02393-5
Sun W, Tian J, Chen L et al (2012) Improvement of biodegradability of PVA-containing wastewater by ionizing radiation pretreatment. Environ Sci Pollut Res 19:3178–3184. https://doi.org/10.1007/s11356-012-0821-4
Sun W, Chen L, Zhang Y, Wang J (2015) Synergistic effect of ozonation and ionizing radiation for PVA decomposition. J Environ Sci (China) 34:63–67. https://doi.org/10.1016/j.jes.2015.01.020
Sun W, Chen L, Wang J (2017) Degradation of PVA(polyvinyl alcohol) in wastewater by advanced oxidation processes. J Adv Oxid Technol. https://doi.org/10.1515/jaots-2017-0018
Tan G, Sağlam S, Emül E et al (2016) Synthesis and characterization of silver nanoparticles integrated in polyvinyl alcohol nanofibers for bionanotechnological applications. Turk J Biol 40:643–651. https://doi.org/10.3906/biy-1505-71
Wu W, Huang Z-H, Lim T-T (2014) Recent development of mixed metal oxide anodes for electrochemical oxidation of organic pollutants in water. Appl Catal A Gen 480:58–78. https://doi.org/10.1016/j.apcata.2014.04.035
Yan Z, Zhu J, Hua X, Liang D, Dong D, Guo Z, Zhang L (2020) Catalytic ozonation for the degradation of polyvinyl alcohol in aqueous solution using catalyst based on copper and manganese. J Clean Prod 272:122856
Yao Y, Chen Q, Zhou J (2022) Influence of typical electrolytes on electrooxidation of bio-refractory reactive dye. Int J Environ Sci Technol 19:1799–1810. https://doi.org/10.1007/s13762-021-03184-7
Ye B, Li Y, Chen Z et al (2017) Degradation of polyvinyl alcohol (PVA) by UV/chlorine oxidation: Radical roles, influencing factors, and degradation pathway. Water Res 124:381–387. https://doi.org/10.1016/j.watres.2017.05.059
Yu H, Gu G, Song L (1996) Degradation of polyvinyl alcohol in sequencing batch reactors. Environ Technol (UK) 17:1261–1267. https://doi.org/10.1080/09593331708616496
Zhai S, Zheng Q, Ge M (2021) Highly efficient activation of peroxymonosulfate by illite-based ultrafine cobalt oxide for the degradation of polyvinyl alcohol. Sep Purif Technol 269:118759. https://doi.org/10.1016/j.seppur.2021.118759
Zhang SJ, Yu HQ (2004) Radiation-induced degradation of polyvinyl alcohol in aqueous solutions. Water Res 38:309–316. https://doi.org/10.1016/j.watres.2003.09.020
Zhang Y, Rong W, Fu Y, Ma X (2011) Photocatalytic degradation of poly(vinyl alcohol) on Pt/TiO2 with fenton reagent. J Polym Environ 19:966–970. https://doi.org/10.1007/s10924-011-0350-0
Zhang Y, Zhu G, Ge M (2021) Degradation of PVA wastewater by CuxMnxFe3−2xO4/MWCNT catalyst. J Mater Sci Mater Electron 32:18616–18627. https://doi.org/10.1007/s10854-021-06321-7
Zhu G, Ge M (2021) Study on efficient degradation of polyvinyl alcohol in aqueous solution. Environ Chall 4:100176. https://doi.org/10.1016/j.envc.2021.100176
Zimmermann, Wolfgang Schindler H (1979) Process for the separation of polyvinyl alcohol from aqueous solutions
Zou J, Peng X, Li M et al (2017) Electrochemical oxidation of COD from real textile wastewaters: kinetic study and energy consumption. Chemosphere 171:332–338. https://doi.org/10.1016/j.chemosphere.2016.12.065
Acknowledgements
We thank the Scientific and Technical Research Council of Turkey (TUBITAK, Grant Number: 115Y305) for funding the research project.
Author contributions
EG: Conceptualization, methodology, data curation, writing—review and editing.: SKB: Conceptualization, investigation, supervision, writing—review and editing.: HH: Conceptualization, investigation, data curation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no confict of interest.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Editorial responsibility: Maryam Shabani.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gengec, E., Karayunlu Bozbas, S. & Ayhan, H.H. Treatment of embroidery wastewater containing poly(vinyl alcohol) by electrooxidation process. Int. J. Environ. Sci. Technol. 21, 1223–1234 (2024). https://doi.org/10.1007/s13762-023-05210-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-023-05210-2