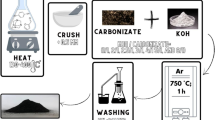
Abstract
Nowadays, the pollution relating to organic dyes imposes many endangering risk on the aqueous ecosystems and mankind. There is a raising interest in the purification of synthetic dyes-polluted waters by the advanced technologies. Herein, we reported the direct and facile synthesis of N-doped porous carbons by pyrolyzing ZIF-8 as a self-sacrificial precursor for removing a wide range of synthetic dyes involving crystal violet, Congo red, malachite green, methyl orange. N-doped porous carbons were featured by some advanced analytic techniques. Moreover, the influence of pyrolyzing temperature (600–800 °C), adsorption time (0–180 min), and dyes content (10–100 mg/L) on uptake capacity on N-doped porous carbons was optimized. Nonlinearized kinetic and isotherm fittings aided to more understand the nature of organic dyes uptake. The highlight outcome indicated the presence of vital surface functional groups such as phenolic, carboxyl, lactone, and basic groups on N-doped porous carbon. Adsorption kinetics indicated the adherence of both pseudo-first-order equation, and pseudo-second-order equations. Equilibrium uptake of crystal violet, Congo red, as well as methyl orange, obeyed Langmuir model, while that of malachite green followed Freundlich model. With five cycle recyclability and sufficient adsorption capacity, N-doped porous carbon may be a suitable adsorbent for removing organic dyes in water.







Similar content being viewed by others
References
Ahmad A, Khan N, Giri BS et al (2020) Removal of methylene blue dye using rice husk, cow dung and sludge biochar: Characterization, application, and kinetic studies. Bioresour Technol 306:123202. https://doi.org/10.1016/j.biortech.2020.123202
Ahmed I, Bhadra BN, Lee HJ, Jhung SH (2018) Metal-organic framework-derived carbons: preparation from ZIF-8 and application in the adsorptive removal of sulfamethoxazole from water. Catal Today 301:90–97. https://doi.org/10.1016/j.cattod.2017.02.011
Alizadeh N, Shariati S, Besharati N (2017) Adsorption of crystal violet and methylene blue on azolla and fig leaves modified with magnetite iron oxide nanoparticles. Int J Environ Res 11:197–206. https://doi.org/10.1007/s41742-017-0019-1
Altintig E, Onaran M, Sarı A et al (2018) Preparation, characterization and evaluation of bio-based magnetic activated carbon for effective adsorption of malachite green from aqueous solution. Mater Chem Phys 220:313–321. https://doi.org/10.1016/j.matchemphys.2018.05.077
Altıntıg E, Yenigun M, Sarı A et al (2021) Facile synthesis of zinc oxide nanoparticles loaded activated carbon as an eco-friendly adsorbent for ultra-removal of malachite green from water. Environ Technol Innov 21:101305. https://doi.org/10.1016/j.eti.2020.101305
An HJ, Park JM, Khan NA, Jhung SH (2020) Adsorptive removal of bulky dye molecules from water with mesoporous polyaniline-derived carbon. Beilstein J Nanotechnol 11:597–605. https://doi.org/10.3762/bjnano.11.47
Bhadra BN, Ahmed I, Kim S, Jhung SH (2017) Adsorptive removal of ibuprofen and diclofenac from water using metal-organic framework-derived porous carbon. Chem Eng J 314:50–58. https://doi.org/10.1016/j.cej.2016.12.127
Butova VV, Budnik AP, Bulanova EA, Soldatov AV (2016) New microwave-assisted synthesis of ZIF-8. Mendeleev Commun 26:43–44. https://doi.org/10.1016/j.mencom.2016.01.017
Caicedo DF, dos Reis GS, Lima EC et al (2020) Efficient adsorbent based on construction and demolition wastes functionalized with 3-aminopropyltriethoxysilane (APTES) for the removal ciprofloxacin from hospital synthetic effluents. J Environ Chem Eng 8:103875
Chahinez H-O, Abdelkader O, Leila Y, Tran HN (2020) One-stage preparation of palm petiole-derived biochar: Characterization and application for adsorption of crystal violet dye in water. Environ Technol Innov 19:100872. https://doi.org/10.1016/j.eti.2020.100872
Chen Y-Z, Cai G, Wang Y et al (2016) Palladium nanoparticles stabilized with N-doped porous carbons derived from metal–organic frameworks for selective catalysis in biofuel upgrade: the role of catalyst wettability. Green Chem 18:1212–1217. https://doi.org/10.1039/C5GC02530C
Chen H, Liu T, Meng Y et al (2020) Novel graphene oxide/aminated lignin aerogels for enhanced adsorption of malachite green in wastewater. Coll Surf A Physicochem Eng Asp 603:125281. https://doi.org/10.1016/j.colsurfa.2020.125281
Choudhary M, Kumar R, Neogi S (2020) Activated biochar derived from Opuntia ficus-indica for the efficient adsorption of malachite green dye, Cu+2 and Ni+2 from water. J Hazard Mater 392:122441. https://doi.org/10.1016/j.jhazmat.2020.122441
Dang HH, Nguyen DTC, Nguyen TT et al (2021) Zeolitic-imidazolate framework-derived N-self-doped porous carbons with ultrahigh theoretical adsorption capacities for tetracycline and ciprofloxacin. J Environ Chem Eng 9:104938. https://doi.org/10.1016/j.jece.2020.104938
Darwish AAA, Rashad M, AL-Aoh HA (2019) Methyl orange adsorption comparison on nanoparticles: Isotherm, kinetics, and thermodynamic studies. Dye Pigment 160:563–571. https://doi.org/10.1016/j.dyepig.2018.08.045
Deb A, Kanmani M, Debnath A et al (2019) Ultrasonic assisted enhanced adsorption of methyl orange dye onto polyaniline impregnated zinc oxide nanoparticles: kinetic, isotherm and optimization of process parameters. Ultrason Sonochem 54:290–301. https://doi.org/10.1016/j.ultsonch.2019.01.028
Donkadokula NY, Kola AK, Naz I, Saroj D (2020) A review on advanced physico-chemical and biological textile dye wastewater treatment techniques. Rev Environ Sci Bio/technology 19:543–560. https://doi.org/10.1007/s11157-020-09543-z
Du X-D, Wang C-C, Liu J-G et al (2017) Extensive and selective adsorption of ZIF-67 towards organic dyes: Performance and mechanism. J Colloid Interface Sci 506:437–441
Ebrahimi AK, Sheikhshoaie I, Mehran M (2017) Facile synthesis of a new metal-organic framework of copper (II) by interface reaction method, characterization, and its application for removal of Malachite Green. J Mol Liq 240:803–809. https://doi.org/10.1016/j.molliq.2017.06.097
Eltaweil AS, Ali Mohamed H, Abd El-Monaem EM, El-Subruiti GM (2020) Mesoporous magnetic biochar composite for enhanced adsorption of malachite green dye: characterization, adsorption kinetics, thermodynamics and isotherms. Adv Powder Technol 31:1253–1263. https://doi.org/10.1016/j.apt.2020.01.005
Freundlich HMF (1906) Over the adsorption in solution. J Phys Chem 57:1100–1107
Gai F, Zhu D, Wu Y et al (2021) Tailored N-doped porous carbons via a MOF assembly process for high-performance CO2 uptake. Mater Adv 2:692–699. https://doi.org/10.1039/D0MA00789G
Gao M, Wang Z, Yang C et al (2019) Novel magnetic graphene oxide decorated with persimmon tannins for efficient adsorption of malachite green from aqueous solutions. Coll Surf A Physicochem Eng Asp 566:48–57. https://doi.org/10.1016/j.colsurfa.2019.01.016
Gopinath KP, Vo D-VN, Gnana Prakash D et al (2021) Environmental applications of carbon-based materials: a review. Environ Chem Lett 19:557–582. https://doi.org/10.1007/s10311-020-01084-9
Han L-J, Ge F-Y, Sun G-H et al (2019) Effective adsorption of Congo red by a MOF-based magnetic material. Dalt Trans 48:4650–4656. https://doi.org/10.1039/C9DT00813F
Hasanzadeh M, Simchi A, Far HS (2019) Kinetics and adsorptive study of organic dye removal using water-stable nanoscale metal organic frameworks. Mater Chem Phys. https://doi.org/10.1016/j.matchemphys.2019.05.050
Ho Y-S, McKay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34:451–465. https://doi.org/10.1016/s0032-9592(98)00112-5
Huang D, Xin Q, Ni Y et al (2018) Synergistic effects of zeolite imidazole framework@graphene oxide composites in humidified mixed matrix membranes on CO2 separation. RSC Adv 8:6099–6109. https://doi.org/10.1039/C7RA09794H
Ibrahim RK, El-Shafie A, Hin LS et al (2019) A clean approach for functionalized carbon nanotubes by deep eutectic solvents and their performance in the adsorption of methyl orange from aqueous solution. J Environ Manage 235:521–534. https://doi.org/10.1016/j.jenvman.2019.01.070
Jiang M, Cao X, Zhu D et al (2016) Hierarchically porous N-doped carbon derived from ZIF-8 nanocomposites for electrochemical applications. Electrochim Acta 196:699–707. https://doi.org/10.1016/j.electacta.2016.02.094
Karami A, Sabouni R, Al-Sayah MH, Aidan A (2021) Adsorption potentials of iron-based metal–organic framework for methyl orange removal: batch and fixed-bed column studies. Int J Environ Sci Technol 18:3597–3612. https://doi.org/10.1007/s13762-020-03103-2
Kasera N, Hall S, Kolar P (2021) Effect of surface modification by nitrogen-containing chemicals on morphology and surface characteristics of N-doped pine bark biochars. J Environ Chem Eng 9:105161. https://doi.org/10.1016/j.jece.2021.105161
Katheresan V, Kansedo J, Lau SY (2018) Efficiency of various recent wastewater dye removal methods: a review. J Environ Chem Eng 6:4676–4697. https://doi.org/10.1016/j.jece.2018.06.060
Kausar A, Iqbal M, Javed A et al (2018) Dyes adsorption using clay and modified clay: a review. J Mol Liq 256:395–407. https://doi.org/10.1016/j.molliq.2018.02.034
Khorasani AC, Shojaosadati SA (2019) Magnetic pectin-Chlorella vulgaris biosorbent for the adsorption of dyes. J Environ Chem Eng 7:103062. https://doi.org/10.1016/j.jece.2019.103062
Klinthongchai Y, Prichanont S, Praserthdam P, Jongsomjit B (2021) Effect of TMB/P123 ratios on physicochemical properties of mesocellular foam carbon (MCF-C) as catalyst for ethanol dehydrogenation. Mater Today Chem 20:100466. https://doi.org/10.1016/j.mtchem.2021.100466
Konicki W, Aleksandrzak M, Mijowska E (2017) Equilibrium, kinetic and thermodynamic studies on adsorption of cationic dyes from aqueous solutions using graphene oxide. Chem Eng Res Des 123:35–49. https://doi.org/10.1016/j.cherd.2017.03.036
Kundu S, Chowdhury IH, Naskar MK (2017) Synthesis of hexagonal shaped nanoporous carbon for efficient adsorption of methyl orange dye. J Mol Liq 234:417–423. https://doi.org/10.1016/j.molliq.2017.03.090
Lafi R, Montasser I, Hafiane A (2018) Adsorption of congo red dye from aqueous solutions by prepared activated carbon with oxygen-containing functional groups and its regeneration. Adsorpt Sci Technol 37:160–181. https://doi.org/10.1177/0263617418819227
Lagergren SK (1898) About the theory of so-called adsorption of soluble substances. Sven Vetenskapsakad Handingarl 24:1–39
Langmuir I (1916) The constitution and fundamental properties of solids and liquids. Part I Solids. J Am Chem Soc 38:2221–2295
Li J, Zhu Q-L, Xu Q (2015) Pd nanoparticles supported on hierarchically porous carbons derived from assembled nanoparticles of a zeolitic imidazolate framework (ZIF-8) for methanol electrooxidation. Chem Commun 51:10827–10830. https://doi.org/10.1039/C5CC03008K
Li S, Zhang X, Huang Y (2017) Zeolitic imidazolate framework-8 derived nanoporous carbon as an effective and recyclable adsorbent for removal of ciprofloxacin antibiotics from water. J Hazard Mater 321:711–719. https://doi.org/10.1016/j.jhazmat.2016.09.065
Li Y, Kim J, Wang J et al (2018) High performance capacitive deionization using modified ZIF-8-derived, N-doped porous carbon with improved conductivity. Nanoscale 10:14852–14859. https://doi.org/10.1039/C8NR02288G
Li N, Zhou L, Jin X et al (2019) Simultaneous removal of tetracycline and oxytetracycline antibiotics from wastewater using a ZIF-8 metal organic-framework. J Hazard Mater 366:563–572. https://doi.org/10.1016/j.jhazmat.2018.12.047
Liang C, Tang Y, Zhang X et al (2020) ZIF-mediated N-doped hollow porous carbon as a high performance adsorbent for tetracycline removal from water with wide pH range. Environ Res 182:109059. https://doi.org/10.1016/j.envres.2019.109059
Liu J, Wang N, Zhang H, Baeyens J (2019) Adsorption of Congo red dye on FexCo3-xO4 nanoparticles. J Environ Manage 238:473–483. https://doi.org/10.1016/j.jenvman.2019.03.009
Liu W, Cui G, Wang H et al (2020) Efficient and selective adsorption of dye in aqueous environment employing a functional Zn(II)-based metal organic framework. J Solid State Chem 292:121740. https://doi.org/10.1016/j.jssc.2020.121740
Liu X, Wu Y, Ye H et al (2021) Modification of sugar-based carbon using lanthanum and cobalt bimetal species for effective adsorption of methyl orange. Environ Technol Innov 23:101769. https://doi.org/10.1016/j.eti.2021.101769
Lu P, Hu X, Li Y et al (2018) One-step preparation of a novel SrCO3/g-C3N4 nano-composite and its application in selective adsorption of crystal violet. RSC Adv 8:6315–6325. https://doi.org/10.1039/C7RA11565B
Lum PT, Foo KY, Zakaria NA, Palaniandy P (2020) Ash based nanocomposites for photocatalytic degradation of textile dye pollutants: a review. Mater Chem Phys 241:122405. https://doi.org/10.1016/j.matchemphys.2019.122405
Lv S-W, Liu J-M, Ma H et al (2019) Simultaneous adsorption of methyl orange and methylene blue from aqueous solution using amino functionalized Zr-based MOFs. Micropor Mesoporous Mater 282:179–187. https://doi.org/10.1016/j.micromeso.2019.03.017
Mashkoor F, Nasar A (2019) Preparation, characterization and adsorption studies of the chemically modified Luffa aegyptica peel as a potential adsorbent for the removal of malachite green from aqueous solution. J Mol Liq 274:315–327. https://doi.org/10.1016/j.molliq.2018.10.119
Mullick A, Neogi S (2016) Synthesis of potential biosorbent from used stevia leaves and its application for malachite green removal from aqueous solution: kinetics, isotherm and regeneration studies. RSC Adv 6:65960–65975. https://doi.org/10.1039/C6RA15225B
Munagapati VS, Yarramuthi V, Kim Y et al (2018) Removal of anionic dyes (Reactive Black 5 and Congo Red) from aqueous solutions using Banana Peel Powder as an adsorbent. Ecotoxicol Environ Saf 148:601–607. https://doi.org/10.1016/j.ecoenv.2017.10.075
Nasar A, Mashkoor F (2019) Application of polyaniline-based adsorbents for dye removal from water and wastewater—a review. Environ Sci Pollut Res 26:5333–5356. https://doi.org/10.1007/s11356-018-3990-y
Nguyen DTC, Dang HH, Vo D-VN et al (2021) Biogenic synthesis of MgO nanoparticles from different extracts (flower, bark, leaf) of Tecoma stans (L) and their utilization in selected organic dyes treatment. J Hazard Mater 404:124146. https://doi.org/10.1016/j.jhazmat.2020.124146
Nguyen DTC, Le HTN, Nguyen TT et al (2021) Multifunctional ZnO nanoparticles bio-fabricated from Canna indica L flowers for seed germination, adsorption, and photocatalytic degradation of organic dyes. J Hazard Mater 420:126586. https://doi.org/10.1016/j.jhazmat.2021.126586
Nodehi R, Shayesteh H, Kelishami AR (2020) Enhanced adsorption of congo red using cationic surfactant functionalized zeolite particles. Microchem J 153:104281. https://doi.org/10.1016/j.microc.2019.104281
Parvin S, Biswas BK, Rahman MA et al (2019) Study on adsorption of Congo red onto chemically modified egg shell membrane. Chemosphere 236:124326. https://doi.org/10.1016/j.chemosphere.2019.07.057
Patra BR, Nanda S, Dalai AK, Meda V (2021) Taguchi-based process optimization for activation of agro-food waste biochar and performance test for dye adsorption. Chemosphere 285:131531. https://doi.org/10.1016/j.chemosphere.2021.131531
Paul A, Vyas G, Paul P, Srivastava DN (2018) Gold-nanoparticle-encapsulated ZIF-8 for a mediator-free enzymatic glucose sensor by amperometry. ACS Appl Nano Mater 1:3600–3607. https://doi.org/10.1021/acsanm.8b00748
Preetha BK, Vishalakshi B (2020) Microwave assisted synthesis of karaya gum based montmorillonite nanocomposite: Characterisation, swelling and dye adsorption studies. Int J Biol Macromol 154:739–750. https://doi.org/10.1016/j.ijbiomac.2020.03.107
Putri KNA, Keereerak A, Chinpa W (2020) Novel cellulose-based biosorbent from lemongrass leaf combined with cellulose acetate for adsorption of crystal violet. Int J Biol Macromol 156:762–772. https://doi.org/10.1016/j.ijbiomac.2020.04.100
Sarker M, Song JY, Jeong AR et al (2018) Adsorptive removal of indole and quinoline from model fuel using adenine-grafted metal-organic frameworks. J Hazard Mater 344:593–601
Schuepfer DB, Badaczewski F, Guerra-Castro JM et al (2020) Assessing the structural properties of graphitic and non-graphitic carbons by raman spectroscopy. Carbon N Y 161:359–372. https://doi.org/10.1016/j.carbon.2019.12.094
Sharifpour E, Khafri HZ, Ghaedi M et al (2018) Isotherms and kinetic study of ultrasound-assisted adsorption of malachite green and Pb2+ ions from aqueous samples by copper sulfide nanorods loaded on activated carbon: experimental design optimization. Ultrason Sonochem 40:373–382. https://doi.org/10.1016/j.ultsonch.2017.07.030
Sharma G, Kumar A, Naushad M et al (2018) Fabrication and characterization of Gum arabic-cl-poly(acrylamide) nanohydrogel for effective adsorption of crystal violet dye. Carbohydr Polym 202:444–453. https://doi.org/10.1016/j.carbpol.2018.09.004
Shi Z, Li L, Xiao Y et al (2017) Synthesis of mixed-ligand Cu–MOFs and their adsorption of malachite green. RSC Adv 7:30904–30910. https://doi.org/10.1039/C7RA04820C
Shi Z, Xu C, Guan H et al (2018) Magnetic metal organic frameworks (MOFs) composite for removal of lead and malachite green in wastewater. Coll Surf A Physicochem Eng Asp 539:382–390. https://doi.org/10.1016/j.colsurfa.2017.12.043
Soltani R, Marjani A, Hosseini M, Shirazian S (2020) Mesostructured hollow siliceous spheres for adsorption of dyes. Chem Eng Technol 43:392–402. https://doi.org/10.1002/ceat.201900470
Tang Y, Chen Q, Li W et al (2020) Engineering magnetic N-doped porous carbon with super-high ciprofloxacin adsorption capacity and wide pH adaptability. J Hazard Mater 388:122059
Tella AC, Olawale MD, Neuburger M, Obaleye JA (2017) Synthesis and crystal structure of Cd-based metal-organic framework for removal of methyl-orange from aqueous solution. J Solid State Chem 255:157–166. https://doi.org/10.1016/j.jssc.2017.07.019
Tran TV, Nguyen DTC, Le HTN et al (2019a) Response surface methodology-optimized removal of chloramphenicol pharmaceutical from wastewater using Cu3(BTC)2-derived porous carbon as an efficient adsorbent. Comptes Rendus Chim 22:794–803. https://doi.org/10.1016/j.crci.2019.09.004
Tran TV, Nguyen DTC, Nguyen H-TT et al (2019b) Application of Fe-based metal-organic framework and its pyrolysis products for sulfonamide treatment. Environ Sci Pollut Res 26:28106–28126
Tran TV, Nguyen DTC, Nguyen TT et al (2020a) Metal-organic framework HKUST-1-based Cu/Cu2O/CuO@C porous composite: Rapid synthesis and uptake application in antibiotics remediation. J Water Process Eng 36:101319. https://doi.org/10.1016/j.jwpe.2020.101319
Tran TV, Nguyen H, Le PHA et al (2020b) Microwave-assisted solvothermal fabrication of hybrid zeolitic–imidazolate framework (ZIF-8) for optimizing dyes adsorption efficiency using response surface methodology. J Environ Chem Eng 8:104189. https://doi.org/10.1016/j.jece.2020.104189
Tran TV, Nguyen VH, Nong LX et al (2020c) Hexagonal Fe-based MIL-88B nanocrystals with NH2 functional groups accelerating oxytetracycline capture via hydrogen bonding. Surf Interf 20:100605. https://doi.org/10.1016/j.surfin.2020.100605
Tran TV, Phan T-QT, Nguyen DTC et al (2020d) Recyclable Fe3O4@ C nanocomposite as potential adsorbent for a wide range of organic dyes and simulated hospital effluents. Environ Technol Innov 20:101122. https://doi.org/10.1016/j.eti.2020.101122
Tran TV, Nong LX, Nguyen H-TT et al (2021) Response surface methodology modeling for methylene blue removal by chemically modified porous carbon: adsorption mechanism and role of surface functional groups. Sep Sci Technol 56:2232–2242. https://doi.org/10.1080/01496395.2020.1820523
Vikrant K, Giri BS, Raza N et al (2018) Recent advancements in bioremediation of dye: current status and challenges. Bioresour Technol 253:355–367. https://doi.org/10.1016/j.biortech.2018.01.029
Wang D, Chen Y, Wang H et al (2018) N-doped porous carbon anchoring on carbon nanotubes derived from ZIF-8/polypyrrole nanotubes for superior supercapacitor electrodes. Appl Surf Sci 457:1018–1024. https://doi.org/10.1016/j.apsusc.2018.07.047
Wang J, Wang Y, Liang Y et al (2021) Nitrogen-doped carbons from in-situ glucose-coated ZIF-8 as efficient adsorbents for Rhodamine B removal from wastewater. Micropor Mesopo Mater 310:110662. https://doi.org/10.1016/j.micromeso.2020.110662
Wei J, Liu Y, Li J et al (2019) Adsorption and co-adsorption of tetracycline and doxycycline by one-step synthesized iron loaded sludge biochar. Chemosphere 236:124254
Wen G, Guo Z (2018) Facile modification of NH2-MIL-125(Ti) to enhance water stability for efficient adsorptive removal of crystal violet from aqueous solution. Coll Surf A Physicochem Eng Asp 541:58–67. https://doi.org/10.1016/j.colsurfa.2018.01.011
Wu K, Yu J, Jiang X (2017) Multi-walled carbon nanotubes modified by polyaniline for the removal of alizarin yellow R from aqueous solutions. Adsorpt Sci Technol 36:198–214. https://doi.org/10.1177/0263617416687564
Wu Y, Su M, Chen J et al (2019) Superior adsorption of methyl orange by h-MoS2 microspheres: isotherm, kinetics, and thermodynamic studies. Dye Pigment 170:107591. https://doi.org/10.1016/j.dyepig.2019.107591
Wu J, Yang J, Feng P et al (2020) High-efficiency removal of dyes from wastewater by fully recycling litchi peel biochar. Chemosphere 246:125734. https://doi.org/10.1016/j.chemosphere.2019.125734
Wu X, Xiong J, Liu S et al (2021) Investigation of hierarchically porous zeolitic imidazolate frameworks for highly efficient dye removal. J Hazard Mater 417:126011. https://doi.org/10.1016/j.jhazmat.2021.126011
Xie J, Lin R, Liang Z et al (2021) Effect of cations on the enhanced adsorption of cationic dye in Fe3O4-loaded biochar and mechanism. J Environ Chem Eng 9:105744. https://doi.org/10.1016/j.jece.2021.105744
Xu S, Lv Y, Zeng X, Cao D (2017) ZIF-derived nitrogen-doped porous carbons as highly efficient adsorbents for removal of organic compounds from wastewater. Chem Eng J 323:502–511. https://doi.org/10.1016/j.cej.2017.04.093
Yang J-M (2017) A facile approach to fabricate an immobilized-phosphate zirconium-based metal-organic framework composite (UiO-66-P) and its activity in the adsorption and separation of organic dyes. J Coll Interf Sci 505:178–185. https://doi.org/10.1016/j.jcis.2017.05.040
Yaseen DA, Scholz M (2019) Textile dye wastewater characteristics and constituents of synthetic effluents: a critical review. Int J Environ Sci Technol 16:1193–1226. https://doi.org/10.1007/s13762-018-2130-z
Yu J, Zhang X, Wang D, Li P (2018) Adsorption of methyl orange dye onto biochar adsorbent prepared from chicken manure. Water Sci Technol 77:1303–1312. https://doi.org/10.2166/wst.2018.003
Yuan X, Zhou F, Man R, Huang J (2019) Dendritic post-cross-linked resin for the adsorption of crystal violet from aqueous solution. J Chem Thermodyn 130:235–242. https://doi.org/10.1016/j.jct.2018.09.030
Zaidi NAHM, Lim LBL, Usman A (2019) Enhancing adsorption of malachite green dye using base-modified Artocarpus odoratissimus leaves as adsorbents. Environ Technol Innov 13:211–223. https://doi.org/10.1016/j.eti.2018.12.002
Zhai L, Bai Z, Zhu Y et al (2018) Fabrication of chitosan microspheres for efficient adsorption of methyl orange. Chinese J Chem Eng 26:657–666. https://doi.org/10.1016/j.cjche.2017.08.015
Zhang L, Su Z, Jiang F et al (2014) Highly graphitized nitrogen-doped porous carbon nanopolyhedra derived from ZIF-8 nanocrystals as efficient electrocatalysts for oxygen reduction reactions. Nanoscale 6:6590–6602. https://doi.org/10.1039/C4NR00348A
Zhang X, Qian L, Yang S et al (2020) Comparative studies of methyl orange adsorption in various metal-organic frameworks by nitrogen adsorption and positron annihilation lifetime spectroscopy. Micropor Mesopor Mater 296:109993. https://doi.org/10.1016/j.micromeso.2019.109993
Zheng Y, Zhu Y, Wang A (2014) Highly efficient and selective adsorption of malachite green onto granular composite hydrogel. Chem Eng J 257:66–73. https://doi.org/10.1016/j.cej.2014.07.032
Zhong S, Zhan C, Cao D (2015) Zeolitic imidazolate framework-derived nitrogen-doped porous carbons as high performance supercapacitor electrode materials. Carbon N Y 85:51–59. https://doi.org/10.1016/j.carbon.2014.12.064
Zhong S, Wang Q, Cao D (2016) ZIF-derived nitrogen-doped porous carbons for Xe adsorption and separation. Sci Rep 6:21295. https://doi.org/10.1038/srep21295
Zolgharnein J, Dermanaki Farahani S, Bagtash M, Amani S (2020) Application of a new metal-organic framework of [Ni2F2(4,4′-bipy)2(H2O)2](VO3)2.8H2O as an efficient adsorbent for removal of Congo red dye using experimental design optimization. Environ Res 182:109054. https://doi.org/10.1016/j.envres.2019.109054
Acknowledgements
This research is funded by Foundation for Science and Technology Development Nguyen Tat Thanh University under Grant No. 2021.01.165/HĐ-KHCN.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Editorial responsibility: Maryam Shabani.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nguyen, D.T.C., Vo, D.V.N., Nguyen, T.T. et al. Kinetic, equilibrium, adsorption mechanisms of cationic and anionic dyes on N-doped porous carbons produced from zeolitic-imidazolate framework. Int. J. Environ. Sci. Technol. 19, 10723–10736 (2022). https://doi.org/10.1007/s13762-021-03888-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-021-03888-w