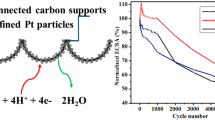
Abstract
This work presents simple post-treatment methods to selectively and partially remove the Pd core of Pd–Pt core–shell (Pt@Pd/C) catalysts. The proton exchange membrane fuel cell with the post-treated Pt@Pd/C cathode (Pt loading: 0.10 mg·cm−2) delivers an impressive peak power density of 1.2 W·cm−2. The partial removal of Pd core endows an ultrahigh oxygen reduction reaction (ORR) mass activity of 0.32 A·mgPGM−1 when normalized to the platinum group metal (PGM) mass, or equivalently 0.55 A·mgPt−1 at 0.9 V measured in a fuel cell. The post-treatment thickens the Pt shells and mitigates the Pd dissolution during potential cycling. As a result, the post-treated core–shell catalyst demonstrates superior durability in ORR mass activity and polarization power density retention than untreated core–shell catalyst and benchmark Pt/C. In-situ inductively coupled plasma-mass spectrometry (ICP-MS) results highlight that the amount of dissolved Pd in post-treated core–shell catalyst is 17-times lower than that of the untreated one. Our findings highlight the importance of structural tuning of catalysts in enhancing their mass activity and durability.

Similar content being viewed by others
References
Wagner, F. T.; Lakshmanan, B.; Mathias, M. F. Electrochemistry and the future of the automobile. J. Phys. Chem. Lett. 2010, 1, 2204–2219.
Debe, M. K. Electrocatalyst approaches and challenges for automotive fuel cells. Nature 2012, 486, 43–51.
Kongkanand, A.; Mathias, M. F. The priority and challenge of high-power performance of low-platinum proton-exchange membrane fuel cells. J. Phys. Chem. Lett. 2016, 7, 1127–1137.
Li, Y.; Chen, M. Y.; Lu, B. A.; Zhang, J. N. Recent advances in exploring highly active & durable PGM-free oxygen reduction catalysts. J. Electrochem. 2023, 29, 2215002.
Singh, K.; Tetteh, E. B.; Lee, H. Y.; Kang, T. H.; Yu, J. S. Tailor-made Pt catalysts with improved oxygen reduction reaction stability/durability. ACS Catal. 2019, 9, 8622–8645.
Papadias, D. D.; Ahluwalia, R. K.; Kariuki, N.; Myers, D.; More, K. L.; Cullen, D. A.; Sneed, B. T.; Neyerlin, K. C.; Mukundan, R.; Borup, R. L. Durability of Pt-Co alloy polymer electrolyte fuel cell cathode catalysts under accelerated stress tests. J. Electrochem. Soc. 2018, 165, F3166–F3177.
Lohse-Busch, H.; Stutenberg, K.; Duoba, M.; Iliev, S. Technology assessment of a fuel cell vehicle: 2017 Toyota Mirai. Argonne National Laboratory: Argonne, 2018.
Borup, R. L.; Kusoglu, A.; Neyerlin, K. C.; Mukundan, R.; Ahluwalia, R. K.; Cullen, D. A.; More, K. L.; Weber, A. Z.; Myers, D. J. Recent developments in catalyst-related PEM fuel cell durability. Curr. Opin. Electrochem. 2020, 21, 192–200.
Shin, J.; Choi, J. H.; Cha, P. R.; Kim, S. K.; Kim, I.; Lee, S. C.; Jeong, D. S. Catalytic activity for oxygen reduction reaction on platinum-based core–shell nanoparticles: All-electron density functional theory. Nanoscale 2015, 7, 15830–15839.
Mavrikakis, M.; Hammer, B.; Nørskov, J. K. Effect of strain on the reactivity of metal surfaces. Phys. Rev. Lett. 1998, 81, 2819–2822.
Zhang, J. L.; Vukmirovic, M. B.; Xu, Y.; Mavrikakis, M.; Adzic, R. R. Controlling the catalytic activity of platinum-monolayer electrocatalysts for oxygen reduction with different substrates. Angew. Chem. 2005, 117, 2170–2173.
Sasaki, K.; Naohara, H.; Cai, Y.; Choi, Y. M.; Liu, P.; Vukmirovic, M. B.; Wang, J. X.; Adzic, R. R. Core-protected platinum monolayer shell high-stability electrocatalysts for fuel-cell cathodes. Angew. Chem. 2010, 122, 8784–8789.
Sasaki, K.; Wang, J. X.; Naohara, H.; Marinkovic, N.; More, K.; Inada, H.; Adzic, R. R. Recent advances in platinum monolayer electrocatalysts for oxygen reduction reaction: Scale-up synthesis, structure, and activity of Pt shells on Pd cores. Electrochim. Acta 2010, 55, 2645–2652.
Zhang, J.; Mo, Y.; Vukmirovic, M. B.; Klie, R.; Sasaki, K.; Adzic, R. R. Platinum monolayer electrocatalysts for O2 reduction: Pt monolayer on Pd (111) and on carbon-supported Pd nanoparticles. J. Phys. Chem. B 2004, 108, 10955–10964.
Branković, S. R.; Wang, J. X.; Adžić, R. R. New methods of controlled monolayer-to-multilayer deposition of Pt for designing electrocatalysts at an atomic level. J. Serb. Chem. Soc. 2001, 66, 887–898.
Zhou, W. P.; Sasaki, K.; Su, D.; Zhu, Y. M.; Wang, J. X.; Adzic, R. R. Gram-scale- synthesized Pd2Co-supported Pt monolayer electrocatalysts for oxygen reduction reaction. J. Phys. Chem. C 2010, 114, 8950–8957.
Shao, M. H.; Protsailo, L. V. Platinum monolayer on hollow, porous nanoparticles with high surface areas and method of making. U.S. Patent 8,921.260 B2, December 30, 2014.
Shao, M. H.; Weidner, J. W.; Odell, J. H. Method and system for core–shell catalyst processing. U.S. Patent 10,541,425 B2, January 21, 2020.
Kongkanand, A.; Subramanian, N. P.; Yu, Y. C.; Liu, Z. Y.; Igarashi, H.; Muller, D. A. Achieving high-power PEM fuel cell performance with an ultralow-Pt-content core–shell catalyst. ACS Catal. 2016, 6, 1578–1583.
Zhang, J.; Lima, F. H. B.; Shao, M. H.; Sasaki, K.; Wang, J. X.; Hanson, J.; Adzic, R. R. Platinum monolayer on nonnoble metal-noble metal core–shell nanoparticle electrocatalysts for O2 reduction. J. Phys. Chem. B 2005, 709, 22701–22704.
Zhou, W. P.; Yang, X. F.; Vukmirovic, M. B.; Koel, B. E.; Jiao, J.; Peng, G. W.; Mavrikakis, M.; Adzic, R. R. Imoroning electrocatalysts for O2 reduction by fine-tuning the Pt-support interaction: Pt monolayer on the surfaces of a Pd3Fe (111) single-crystal alloy. J. Am. Chem. Soc. 2009, 131, 12755–12762.
Knupp, S. L.; Vukmirovic, M. B.; Haldar, P.; Herron, J. A.; Mavrikakis, M.; Adzic, R. R. Platinum monolayer electrocatalysts for O2 reduction: Pt monolayer on carbon-supported PdIr nanoparticles. Electrocatalysis 2010, 1, 213–223.
Kuttiyiel, K. A.; Sasaki, K.; Su, D.; Vukmirovic, M. B.; Marinkovic, N. S.; Adzic, R. R. Pt monolayer on Au-stabilized PdNi core–shell nanoparticles for oxygen reduction reaction. Electrochim. Acta 2013, 110, 267–272.
Kuttiyiel, K. A.; Choi, Y.; Hwang, S. M.; Park, G. G.; Yang, T. H.; Su, D.; Sasaki, K.; Liu, P.; Adzic, R. R. Enhancement of the oxygen reduction on nitride stabilized pt–M (M = Fe, Co, and Ni) core–shell nanoparticle electrocatalysts. Nano Energy 2015, 13, 442–449.
Hu, J.; Kuttiyiel, K. A.; Sasaki, K.; Su, D.; Yang, T. H.; Park, G. G.; Zhang, C. X.; Chen, G. Y.; Adzic, R. R. Pt monolayer shell on nitrided alloy core—A path to highly stable oxygen reduction catalyst. Catalysts 2015, 5, 1321–1332.
Zhang, Y.; Ma, C.; Zhu, Y. M.; Si, R.; Cai, Y.; Wang, J. X.; Adzic, R. R. Hollow core supported Pt monolayer catalysts for oxygen reduction. Catal. Today 2013, 202, 50–54.
Bliznakov, S.; Vukmirovic, M.; Yang, L. J.; Sutter, E.; Adzic, R. R. Pt monolayer on electrodeposited Pd nanostructures-advanced cathode catalysts for PEM fuel cells. ECS Trans. 2016, 41, 1055–1066.
Park, J.; Zhang, L.; Choi, S. I.; Roling, L. T.; Lu, N.; Herron, J. A.; Xie, S. F.; Wang, J. G.; Kim, M. J.; Mavrikakis, M. et al. Atomic layer-by-layer deposition of platinum on palladium octahedra for enhanced catalysts toward the oxygen reduction reaction. ACS Nano 2015, 9, 2635–2647.
Park, J.; Vara, M.; Xia, Y. N. A systematic study of the catalytic durability of Pd@Pt2-3L nano-sized octahedra toward oxygen reduction. Catal. Today 2017, 280, 266–273.
Zhang, J. L.; Vukmirovic, M. B.; Sasaki, K.; Nilekar, A. U.; Mavrikakis, M.; Adzic, R. R. Mixed-metal Pt monolayer electrocatalysts for enhanced oxygen reduction kinetics. J. Am. Chem. Soc. 2005, 127, 12480–12481.
Choi, S. I.; Shao, M. H.; Lu, N.; Ruditskiy, A.; Peng, H. C.; Park, J.; Guerrero, S.; Wang, J. G.; Kim, M. J.; Xia, Y. N. Synthesis and characterization of Pd@Pt-Ni core–shell octahedra with high activity toward oxygen reduction. ACS Nano 2014, 8, 10363–10371.
Zhao, X.; Chen, S.; Fang, Z. C.; Ding, J.; Sang, W.; Wang, Y. C.; Zhao, J.; Peng, Z. M.; Zeng, J. Octahedral Pd@Pt18Ni core–shell nanocrystals with ultrathin PtNi alloy shells as active catalysts for oxygen reduction reaction. J. Am. Chem. Soc. 2015, 137, 2804–2807.
Xie, S. F.; Choi, S. I.; Lu, N.; Roling, L. T.; Herron, J. A.; Zhang, L.; Park, J.; Wang, J. G.; Kim, M. J.; Xie, Z. X. et al. Atomic layer-by-layer deposition of Pt on Pd nanocubes for catalysts with enhanced activity and durability toward oxygen reduction. Nano Lett. 2014, 14, 3570–3576.
Wang, J. X.; Inada, H.; Wu, L. J.; Zhu, Y. M.; Choi, Y.; Liu, P.; Zhou, W. P.; Adzic, R. R. Oxygen reduction on well-defined core–shell nanocatalysts: Particle size, facet, and Pt shell thickness effects. J. Am. Chem. Soc. 2009, 131, 17298–17302.
Wang, X.; Choi, S. I.; Roling, L. T.; Luo, M.; Ma, C.; Zhang, L.; Chi, M. F.; Liu, J. Y.; Xie, Z. X.; Herron, J. A. et al. Palladium-platinum core–shell icosahedra with substantially enhanced activity and durability towards oxygen reduction. Nat. Commun. 2015, 6, 7594.
Zhang, L. L.; Zhu, S. Q.; Chang, Q. W.; Su, D.; Yue, J.; Du, Z.; Shao, M. H. Palladium–platinum core–shell electrocatalysts for oxygen reduction reaction prepared with the assistance of citric acid. ACS Catal. 2016, 6, 3428–3432.
Zhu, S. Q.; Yue, J.; Qin, X. P.; Shao, M. H. Synthesis and evaluation of core–shell electrocatalysts for oxygen reduction reaction. ECS Trans. 2016, 75, 731–740.
Khateeb, S.; Guerreo, S.; Su, D.; Darling, R. M.; Protsailo, L. V.; Shao, M. H. Fuel cell performance of palladium-platinum core–shell electrocatalysts synthesized in gram-scale batches. J. Electrochem. Soc. 2016, 163, F708–F713.
Cochell, T.; Manthiram, A. Pt@PdxCuy/C core–shell electrocatalysts for oxygen reduction reaction in fuel cells. Langmuir 2012, 28, 1579–1587.
Sasaki, K.; Naohara, H.; Choi, Y.; Cai, Y.; Chen, W. F.; Liu, P.; Adzic, R. R. Highly stable Pt monolayer on PdAu nanoparticle electrocatalysts for the oxygen reduction reaction. Nat. Commun. 2012, 3, 1115.
Shao, M. H.; Smith, B. H.; Guerrero, S.; Protsailo, L.; Su, D.; Kaneko, K.; Odell, J. H.; Humbert, M. P.; Sasaki, K.; Marzullo, J. et al. Core-shell catalysts consisting of nanoporous cores for oxygen reduction reaction. Phys. Chem. Chem. Phys. 2013, 15, 15078–15090.
Adzic, R. R. Platinum monolayer electrocatalysts: Tunable activity, stability, and self-healing properties. Electrocatalysis 2012, 3, 163–169.
Pan, L. J.; Ott, S.; Dionigi, F.; Strasser, P. Current challenges related to the deployment of shape-controlled Pt alloy oxygen reduction reaction nanocatalysts into low Pt-loaded cathode layers of proton exchange membrane fuel cells. Curr. Opin. Electrochem. 2019, 18, 61–71.
Shao, M. H.; Chang, Q. W.; Dodelet, J. P.; Chenitz, R. Recent advances in electrocatalysts for oxygen reduction reaction. Chem. Rev. 2016, 116, 3594–3657.
Ball, S. Core-shell catalysts in PEMFC cathode environments. In Electrocatalysis in Fuel Cells: A Non- and Low-Platinum Approach. Shao, M. H., Ed.; Springer: London, 2013; pp 561–587.
Shao, M. H. Method to prepare full monolayer of platinum on palladium based core nanoparticles. U.S. Patent 9.246,176 B2, January 26, 2016.
Kaneko, K.; Kimura, H.; Adachi, M. Method for producing core–shell catalyst particles. U.S. Patent 10,263,259 B2, April 16, 2019.
Thambidurai, C.; Gebregziabiher, D. K.; Liang, X. H.; Zhang, Q.; H. Ivanova, V.; Haumesser, P. H.; Stickney, J. L. E-ALD of Cu nanofilms on Ru/Ta wafers using surface limited redox replacement. J. Electrochem. Soc. 2010, 157, D466–D471.
Zhu, S. Q.; Yue, J.; Qin, X. P.; Wei, Z. D.; Liang, Z. X.; Adzic, R. R.; Brankovic, S. R.; Du, Z.; Shao, M. H. The role of citric acid in perfecting platinum monolayer on palladium nanoparticles during the surface limited redox replacement reaction. J. Electrochem. Soc. 2016, 163, D3040–D3046.
Shao, M. H.; Odell, J. H.; Choi, S. I.; Xia, Y. N. Electrochemical surface area measurements of platinum- and palladium-based nanoparticles. Electrochem. Commun. 2013, 31, 46–48.
Sassin, M. B.; Garsany, Y.; Gould, B. D.; Swider-Lyons, K. E. Fabrication method for laboratory-scale high-performance membrane electrode assemblies for fuel cells. Anal. Chem. 2017, 89, 511–518.
Kabir, S.; Myers, D. J.; Kariuki, N.; Park, J.; Wang, G. X.; Baker, A.; Macauley, N.; Mukundan, R.; More, K. L.; Neyerlin, K. C. Elucidating the dynamic nature of fuel cell electrodes as a function of conditioning: An ex situ material characterization and in situ electrochemical diagnostic study. ACS Appl. Mater. Interfaces 2019, 11, 45016–45030.
Neyerlin, K. C.; Gu, W. B.; Jorne, J.; Gasteiger, H. A. Determination of catalyst unique parameters for the oxygen reduction reaction in a PEMFC. J. Electrochem. Soc. 2006, 153, A1955–A1963.
Ott, S.; Orfanidi, A.; Schmies, H.; Anke, B.; Nong, H. N.; Hübner, J.; Gernert, U.; Gliech, M.; Lerch, M.; Strasser, P. Ionomer distribution control in porous carbon-supported catalyst layers for high-power and low Pt-loaded proton exchange membrane fuel cells. Nat. Mater. 2020, 19, 77–85.
Xiao, F.; Wang, Y. A.; Xu, G. L.; Yang, F.; Zhu, S. Q.; Sun, C. J.; Cui, Y. D.; Xu, Z. W.; Zhao, Q. L.; Jang, J. et al. Fe-N-C boosts the stability of supported platinum nanoparticles for fuel cells. J. Am. Chem. Soc. 2022, 144, 20372–20384.
Topalov, A. A.; Cherevko, S.; Zeradjanin, A. R.; Meier, J. C.; Katsounaros, I.; Mayrhofer, K. J. J. Towards a comprehensive understanding of platinum dissolution in acidic media. Chem. Sci. 2014, 5, 631–638.
Meier, J. C.; Galeano, C.; Katsounaros, I.; Topalov, A. A.; Kostka, A.; Schüth, F.; Mayrhofer, K. J. J. Degradation mechanisms of Pt/C fuel cell catalysts under simulated start-stop conditions. ACS Catal. 2012, 2, 832–843.
Lopes, P. P.; Strmcnik, D.; Tripkovic, D.; Connell, J. G.; Stamenkovic, V.; Markovic, N. M. Relationships between atomic level surface structure and stability/activity of platinum surface atoms in aqueous environments. ACS Catal. 2016, 6, 2536–2544.
Bard, A. J.; Faulkner, L. R. Electrochemical Methods: Fundamentals and Applications; 2nd ed. John Wiley: New York, 2001; pp 580–632.
Xie, S. F.; Lu, N.; Xie, Z. X.; Wang, J. G.; Kim, M. J.; Xia, Y. N. Synthesis of Pd–Rh core-frame concave nanocubes and their conversion to Rh cubic nanoframes by selective etching of the Pd cores. Angew. Chem. 2012, 124, 10412–10416.
Xia, X. H.; Wang, Y.; Ruditskiy, A.; Xia, Y. N. 25th anniversary article: Galvanic replacement: A simple and versatile route to hollow nanostructures with tunable and well-controlled properties. Adv. Mater. 2013, 25, 6313–6333
Zheng, Y. Q.; Zeng, J.; Ruditskiy, A.; Liu, M. C.; Xia, Y. N. Oxidative etching and its role in manipulating the nucleation and growth of noble-metal nanocrystals. Chem. Mater. 2014, 26, 22–33.
Dursun, A.; Pugh, D. V.; Corcoran, S. G. Dealloying of Ag-Au alloys in halide-containing electrolytes: Affect on critical potential and pore size. J. Electrochem. Soc. 2003, 150, B355–B360.
Xia, Y. N.; Kim, E.; Whitesides, G. M. Microcontact printing of alkanethiols on silver and its application in microfabrication. J. Electrochem. Soc. 1996, 143, 1070–1079.
Xiong, Y. J.; Chen, J. Y.; Wiley, B.; Xia, Y. N.; Aloni, S.; Yin, Y. D. Understanding the role of oxidative etching in the polyol synthesis of Pd nanoparticles with uniform shape and size. J. Am. Chem. Soc. 2005, 127, 7332–7333.
Xiong, Y. J.; Chen, J. Y.; Wiley, B.; Xia, Y. N.; Yin, Y. D.; Li, Z. Y. Size-dependence of surface plasmon resonance and oxidation for Pd nanocubes synthesized via a seed etching process. Nano Lett. 2005, 5, 1237–1242.
Cheng, H.; Gui, R. J.; Yu, H.; Wang, C.; Liu, S.; Liu, H. F.; Zhou, T. P.; Zhang, N.; Zheng, X. S.; Chu, W. S. et al. Subsize Pt-based intermetallic compound enables long-term cyclic mass activity for fuel-cell oxygen reduction. Proc. Natl. Acad. Sci. USA 2021, 118, e2104026118.
Guan, J. Y.; Yang, S. X.; Liu, T. T.; Yu, Y. H.; Niu, J.; Zhang, Z. P.; Wang, F. Intermetallic FePt@PtBi core–shell nanoparticles for oxygen reduction electrocatalysis. Angew. Chem. 2021, 133, 22070–22075.
Liang, J. S.; Zhao, Z. L.; Li, N.; Wang, X. M.; Li, S. Z.; Liu, X.; Wang, T. Y.; Lu, G.; Wang, D. L.; Hwang, B. J. et al. Biaxial strains mediated oxygen reduction electrocatalysis on fenton reaction resistant L10-PtZn fuel cell cathode. Adv. Energy Mater. 2020, 10, 2000179.
Yarlagadda, V.; Carpenter, M. K.; Moylan, T. E.; Kukreja, R. S.; Koestner, R.; Gu, W. B.; Thompson, L.; Kongkanand, A. Boosting fuel cell performance with accessible carbon mesopores. ACS Energy Lett. 2018, 3, 618–621.
Padgett, E.; Yarlagadda, V.; Holtz, M. E.; Ko, M.; Levin, B. D. A.; Kukreja, R. S.; Ziegelbauer, J. M.; Andrews, R. N.; Ilavsky, J.; Kongkanand, A. et al. Mitigation of PEM fuel cell catalyst degradation with porous carbon supports. J. Electrochem. Soc. 2019, 166, F198–F207.
Yang, C. L.; Wang, L. N.; Yin, P.; Liu, J. Y.; Chen, M. X.; Yan, Q. Q.; Wang, Z. S.; Xu, S. L.; Chu, S. Q.; Cui, C. H. et al. Sulfur-anchoring synthesis of platinum intermetallic nanoparticle catalysts for fuel cells. Science 2021, 374, 459–464.
Han, B. H.; Carlton, C. E.; Kongkanand, A.; Kukreja, R. S.; Theobald, B. R.; Gan, L.; O’Malley, R.; Strasser, P.; Wagner, F. T.; Shao-Horn, Y. Record activity and stability of dealloyed bimetallic catalysts for proton exchange membrane fuel cells. Energy Environ. Sci. 2015, 8, 258–266.
Wang, R. Q.; Sui, S. Structure analysis of PEMFC cathode catalyst layer. J. Electrochem. 2021, 27, 595–604.
U.S. Department of Energy The fuel cell technologies office multi-year research, development, and demonstration plan. U.S. Department of Energy, 2016.
Liang, J. S.; Li, N.; Zhao, Z. L.; Ma, L.; Wang, X. M.; Li, S. Z.; Liu, X.; Wang, T. Y.; Du, Y. P.; Lu, G. et al. Tungsten-doped L10-PtCo ultrasmall nanoparticles as a high-performance fuel cell cathode. Angew. Chem., Int. Ed. 2019, 58, 15471–15477.
Zhao, X. R.; Xi, C.; Zhang, R.; Song, L.; Wang, C. Y.; Spendelow, J. S.; Frenkel, A. I.; Yang, J.; Xin, H. L.; Sasaki, K. High-performance nitrogen-doped intermetallic PtNi catalyst for the oxygen reduction reaction. ACS Catal. 2020, 10, 10637–10645.
Cai, X.; Lin, R.; Liu, X.; Zhao, Y. C. Effect of heat treatment on the surface structure of Pd@Pt-Ni core–shell catalysts for the oxygen reduction reaction. J. Alloys Compd. 2021, 884, 161059.
Kang, Y. Q.; Wang, J. Q.; Wei, Y. P.; Wu, Y. L.; Xia, D. S.; Gan, L. Engineering nanoporous and solid core–shell architectures of low-platinum alloy catalysts for high power density PEM fuel cells. Nano Res. 2022, 15, 6148–6155.
Li, J. R.; Xi, Z.; Pan, Y. T.; Spendelow, J. S.; Duchesne, P. N.; Su, D.; Li, Q.; Yu, C.; Yin, Z. Y.; Shen, B. et al. Fe stabilization by intermetallic L10-FePt and Pt catalysis enhancement in L10-FePt/Pt nanoparticles for efficient oxygen reduction reaction in fuel cells. J. Am. Chem. Soc. 2018, 140, 2926–2932.
Chong, L. N.; Wen, J. G.; Kubal, J.; Sen, F. G.; Zou, J. X.; Greeley, J.; Chan, M.; Barkholtz, H.; Ding, W. J.; Liu, D. J. Ultralow-loading platinum-cobalt fuel cell catalysts derived from imidazolate frameworks. Science 2018, 362, 1276–1281.
Dionigi, F.; Weber, C. C.; Primbs, M.; Gocyla, M.; Bonastre, A. M.; Spöri, C.; Schmies, H.; Hornberger, E.; Kühl, S.; Drnec, J. et al. Controlling near-surface Ni composition in octahedral PtNi(Mo) nanoparticles by Mo doping for a highly active oxygen reduction reaction catalyst. Nano Lett. 2019, 19, 6876–6885.
Li, J. R.; Sharma, S.; Liu, X. M.; Pan, Y. T.; Spendelow, J. S.; Chi, M. F.; Jia, Y. K.; Zhang, P.; Cullen, D. A.; Xi, Z. et al. Hard-magnet L10-CoPt nanoparticles advance fuel cell catalysis. Joule 2019, 3, 124–135.
Gasteiger, H. A.; Panels, J. E.; Yan, S. G. Dependence of PEM fuel cell performance on catalyst loading. J. Power Sources 2004, 127, 162–171.
Kaiser, J.; Simonov, P. A.; Zaikovskii, V. I.; Hartnig, C.; Jörissen, L.; Savinova, E. R. Influence of carbon support on the performance of platinum based oxygen reduction catalysts in a polymer electrolyte fuel cell. J. Appl. Electrochem. 2007, 37, 1429–1437.
Wells, P. P.; Crabb, E. M.; King, C. R.; Wiltshire, R.; Billsborrow, B.; Thompsett, D.; Russell, A. E. Preparation, structure, and stability of Pt and Pd monolayer modified Pd and Pt electrocatalysts. Phys. Chem. Chem. Phys. 2009, 11, 5773–5781.
Shao, M. H. Palladium-based electrocatalysts for oxygen reduction reaction. In Electrocatalysis in Fuel Cells: A Non- and Low-Platinum Approach. Shao, M. H., Ed.; Springer: London, 2013; pp 513–531.
Zhang, A. J.; Birss, V. I.; Vanýsek, P. Impedance characterization of thin electrochemically formed palladium oxide films. J. Electroanal. Chem. 1994, 378, 63–75.
Butler, J. A. V.; Drever, G. The mechanism of electrolytic processes. Part I. The anodic oxidation of some metals of the platinum group. Trans. Faraday Soc. 1936, 32, 427–435.
Shao, M. H.; Yu, T.; Odell, J. H.; Jin, M. S.; Xia, Y. N. Structural dependence of oxygen reduction reaction on palladium nanocrystals. Chem. Commun. 2011, 47, 6566–6568.
Chausse, V.; Regull, P.; Victori, L. Formation of a higher palladium oxide in the oxygen evolution potential range. J. Electroanal. Chem. Interfacial Electrochem. 1987, 238, 115–128.
Zhang, A. J.; Gaur, M.; Birss, V. I. Growth of thin, hydrous oxide films at Pd electrodes. J. Electroanal. Chem. 1995, 389, 149–159.
Dall’Antonia, L. H.; Tremiliosi-Filho, G.; Jerkiewicz, G. Influence of temperature on the growth of surface oxides on palladium electrodes. J. Electroanal. Chem. 2001, 502, 72–81.
Grdeń, M.; Łukaszewski, M.; Jerkiewicz, G.; Czerwiński, A. Electrochemical behaviour of palladium electrode: Oxidation, electrodissolution, and ionic adsorption. Electrochim. Acta 2008, 53, 7583–7598.
Birss, V. I.; Chan, M.; Phan, T.; Vanýsek, P.; Zhang, A. An electrochemical study of the composition of thin, compact Pd oxide films. J. Chem. Soc. Faraday Trans. 1996, 92, 4041–4047.
Grdeń, M.; Piaścik, A.; Koczorowski, Z.; Czerwiński, A. Hydrogen electrosorption in Pd-Pt alloys. J. Electroanal. Chem. 2002, 532, 35–42.
Johnson, D. C.; Napp, D. T.; Bruckenstein, S. A ring-disk electrode study of the current/potential behaviour of platinum in 1.0 M sulphuric and 0.1 M perchloric acids. Electrochim. Acta 1970, 15, 1493–1509.
Acknowledgements
This work was supported by the National Key R&D Program of China (No. 2020YFB1505800), the Guangzhou Key Research and Development Program (No. 202103040002), and the Green Tech Fund (No. GTF202020092). M. G. would like to acknowledge the support from the National Key R&D Program of China (No. 2022YFA1503900) and the Guangdong Scientific Program (No. 2019QN01L057).
Author information
Authors and Affiliations
Corresponding authors
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Wu, H., Xiao, F., Wang, J. et al. Highly active and durable core–shell electrocatalysts for proton exchange membrane fuel cells. Nano Res. (2023). https://doi.org/10.1007/s12274-023-6297-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12274-023-6297-3