Abstract
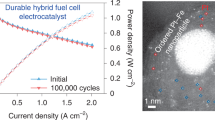
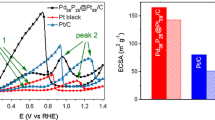
To settle the drawbacks of non-cost-effective and enhance the electrocatalytic performance of Pt/C, Fe@Pt core-shell nanoparticles supported by Vulcan XC-72 have been designed and synthetized successfully. The synthesized materials were characterized by X-ray diffraction, scanning electron microscopy, transmission electron microscopy, and X-ray photoelectron spectroscopy, whereas the electrochemical analyses were obtained by electrochemical impedance spectroscopy, cyclic voltammetry, linear sweep voltammetry, and chronoamperometry. Results suggest that the onset potential of oxygen reduction reaction on the Fe@Pt/C is about 0.6 V (vs. Hg/Hg2Cl2) in O2-saturated 0.5 M H2SO4, which shifts positively more than 30 mV compared with that of Pt/C. The corresponding electron transfer number is 4, meaning the reduction reaction mainly occurred through a 4-electron pathway. More importantly, the Fe@Pt/C has an excellent stability and better catalytic performance towards oxygen reduction reaction activity compared with Pt/C. Thus, Fe@Pt/C could be utilized as promising cathode catalysts in proton exchange membrane fuel cells.




Similar content being viewed by others
References
Santos LGRA, Oliveira CHF, Moraes IR, Ticianelli EA (2006) Oxygen reduction reaction in acid medium on Pt-Ni/C prepared by a microemulsion method. J Electroanal Chem 596:141–148
Yin SB, Cai M, Wang CQ, Shen PK (2011) Tungsten carbide promoted Pd-Fe as alcohol-tolerant electrocatalysts for oxygen reduction reactions. Energy Environ Sci 4:558–563
Zhao X, Yin M, Ma L, Ling L, Liu CP, Liao J-H, Lu TH, Xing W (2011) Recent advances in catalysts for direct methanol fuel cells. Energy Environ Sci 4:2736–2753
Wang Y, Song Q, Maragou V, Shen PK, Tsiakaras P (2009) High surface area tungsten carbide microspheres as effective Pt catalyst support for oxygen reduction reaction. Appl Catal B Environ 89:223–228
Mehta V, Cooper JS (2003) Review and analysis of PEM fuel cell design and manufacturing. J Power Sources 114:32–53
Negro E, Noto VD (2008) Polymer electrolyte fuel cells based on bimetallic carbon nitride electrocatalysts. J Power Sources 178:634–641
Costamagna P, Srinivasan S (2001) Quantum jumps in the PEMFC science and technology from the 1960s to the year 2000: Part II. Engineering, technology development and application aspects. J Power Sources 102:253–269
Zhang H, Jin MS, Xia YN (2012) Enhancing the catalytic and electrocatalytic properties of Pt-based catalysts by forming bimetallic nanocrystals with Pd. Chem Soc Rev 41:8035–8049
Srivastava R, Mani P, Hahn N, Strasser P (2007) Efficient oxygen reduction fuel cell electrocatalysis on voltammetrically dealloyed Pt-Cu-Co nanoparticles. Angew Chem Int Ed 46:8988–8991
Li XY, Liu XH, Wang WW, Li L, Lu XQ (2014) High loading Pt nanoparticles on functionalization of carbon nanotubes for fabricating nonenzyme hydrogen peroxide sensor. Biosens Bioelectron 59:221–226
Gasteiger HA, Kocha SS, Sompalli B, Wagner FT (2005) Activity benchmarks and requirements for Pt, Pt-alloy, and non-Pt oxygen reduction catalysts for PEMFCs. Appl Catal B Environ 56:9–35
Gasteiger HA, Markovic NM (2009) Just a dream—or future reality? Science 324:48–49
Stamenkovic VR, Mun BS, Arenz M, Mayerhofer KJJ, Lucas CA, Wang GF, Ross PN, Markovic NM (2007) Trends in electrocatalysis on extended and nanoscale Pt-bimetallic alloy surfaces. Nat Mater 6:241–247
Stamenkovic VR, Fowler B, Mun BS, Wang GF, Ross PN, Lucas CA, Markovic NM (2007) Improved oxygen reduction activity on Pt3Ni(111) via increased surface site availability. Science 315:493–497
Debe MK (2012) Electrocatalyst approaches and challenges for automotive fuel cells. Nature 486:43–51
Jung N, Chung DY, Ryu J, Yoo SJ, Sung YE (2014) Pt-based nanoarchitecture and catalyst design for fuel cell applications. Nano Today 9:433–456
Suo Y, Zhuang L, Lu JT (2007) First-principles considerations in the design of Pd-alloy catalysts for oxygen reduction. Angew Chem Int Ed 46:2862–2864
Liu YQ, Xu C-X (2013) Nanoporous PdTi alloys as non-platinum oxygen reduction reaction electrocatalysts with enhanced activity and durability. Chem Sus Chem 6:78–84
Han B, Xu C (2014) Nanoporous PdFe alloy as highly active and durable electrocatalyst for oxygen reduction reaction. Int J Hydrog Energy 39:18247–18255
He ZB, Maurice JL, Gohier A, Lee CS, Pribat D, Cojocaru CS (2011) Iron catalysts for the growth of carbon nanofibers: Fe, Fe3C or both. Chem Mater 23:5379–5387
Lu AH, Nitz JJ, Comotti M, Weidenthaler C, Schlichte K, Lehmann CW, Terasaki O, Schüth F (2010) Spatially and size selective synthesis of Fe-based nanoparticles on ordered mesoporous supports as highly active and stable catalysts for ammonia decomposition. J Am Chem Soc 132:14152–14162
Mei H, Wu WQ, Yu BB, Wu HM, Wang SF, Xia QH (2016) Nonenzymatic electrochemical sensor based on Fe@Pt core-shell nanoparticles for hydrogen peroxide, glucose and formaldehyde. Sensors Actuators B Chem 223:68–75
Nadagouda MN, Varma RS, Greener A (2007) Synthesis of core (Fe, Cu)-shell (Au, Pt, Pd, and Ag) nanocrystals using aqueous vitamin C. Cryst Growth Des 7:2582–2587
Kristian N, Yu YL, Lee JM, Liu XW, Wang X (2010) Synthesis and characterization of Cocore-Ptshell electrocatalyst prepared by spontaneous replacement reaction for oxygen reduction reaction. Electrochim Acta 56:1000–1007
Zhu H, Li XW, Wang FH (2011) Synthesis and characterization of Cu@Pt/C core-shell structured catalysts for proton exchange membrane fuel cell. Int J Hydrog Energy 36:9151–9154
Sánchez-Padilla NM, Montemayor SM, Torres LA, Rodríguez Varela FJ (2013) Fast synthesis and electrocatalytic activity of M@Pt (M=Ru, Fe3O4, Pd) core-shell nanostructures for the oxidation of ethanol and methanol. Int J Hydrog Energy 38:12681–12688
Teng XW, Black D, Watkins NJ, Gao YL, Yang H (2003) Platinum-maghemite Core-shell nanoparticles using a sequential synthesis. Nano Lett 3:261–264
Sarkar A, Manthiram A (2010) Synthesis of Pt@Cu core-shell nanoparticles by galvanic displacement of Cu by Pt4+ ions and their application as electrocatalysts for oxygen reduction reaction in fuel cells. J Phys Chem C 114:4725–4732
Mei H, Wu HM, Wu WQ, Wang SF, Xia QH (2015) Ultrasensitive electrochemical assay of hydrogen peroxide and glucose based on PtNi alloy decorated MWCNTs. RSC Adv 5:102877–102884
Mei H, Wu WQ, Yu BB, Wu HM, Wang SF, Zhang XH, Xia QH (2016) Electrochemical sensor for detection of glucose based on Ni@Pt core-shell nanoparticles supported on carbon. Electroanalysis 28:671–678
Wu BH, Hu D, Kuang YJ, Liu B, Zhang XH, Chen JH (2009) Functionalization of carbon nanotubes by an ionic-liquid polymer: dispersion of Pt and PtRu nanoparticles on carbon nanotubes and their electrocatalytic oxidation of methanol. Angew Chem Int Ed 48:4751–4754
Lu J, Bo X, Wang H, Guo L (2013) Nitrogen-doped ordered mesoporous carbons synthesized from honey as metal-free catalyst for oxygen reduction reaction. Electrochim Acta 108:10–16
Mayrhofer KJJ, Strmcnik D, Blizanac BB, Stamenkovic V, Arenz M, Markovic NM (2008) Measurement of oxygen reduction activities via the rotating disc electrode method: from Pt model surfaces to carbon-supported high surface area catalysts. Electrochim Acta 53:3181–3188
Dai L, Xue Y, Qu L, Choi HJ, Baek JB (2015) Metal-free catalysts for oxygen reduction reaction. Chem Rev 115:4823–4892
Zheng Y, Jiao Y, Jaroniec M, Jin Y, Qiao SZ (2012) Nanostructured metal-free electrochemical catalysts for highly efficient oxygen reduction. Small 8:3550–3566
Shao M, Chang Q, Dodelet JP, Chenitz R (2016) Recent advances in electrocatalysts for oxygen reduction reaction. Chem Rev 116:3594–3657
Higgins D, Zamani P, Yu A, Chen Z (2016) The application of graphene and its composites in oxygen reduction electrocatalysis: a perspective and review of recent progress. Energy Environ Sci 9:357–390
Favaro M, Ferrighi L, Fazio G, Colazzo L, Valentin CD, Durante C, Sedona F, Gennaro A, Agnoli S, Granozzi G (2015) Single and multiple doping in graphene quantum dots: unraveling the origin of selectivity in the oxygen reduction reaction. ACS Catal 5:129–144
Acknowledgements
The authors acknowledge financial support from the National Natural Science Foundation of China through a project entitled “The synthesis of Pt-M/C nanoparticles and construction of non-enzymatic electrochemical biosensor” (Grant No. 21205030) and by the National Nature Science Foundation of China (51402096), and by the State Key Laboratory of Advanced Technology for Materials Synthesis and Processing (Wuhan University of Technology, 2015-KF-13), and from the Natural Science Fund for Creative Research Groups of Hubei Province of China (2014CFA015).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Huang, Y., Tan, Z., Wu, H. et al. Fe@Pt core-shell nanoparticles as electrocatalyst for oxygen reduction reaction in acidic media. Ionics 24, 229–236 (2018). https://doi.org/10.1007/s11581-017-2186-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-017-2186-7