Abstract
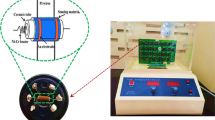
Fe2O3–In2O3 (9:1 mol.%) heterostructure nanoparticles were prepared by the thermal decomposition of stoichiometric amounts of Fe2C2O4 2H2O and In(OH)3 at 400°C for 20 h. The sample was characterized by x-ray diffraction (XRD), Fourier-transform infrared spectrometry, thermogravimetry–differential thermal analyzer, scanning electron microscopy/transmission electron microscopy, and a superconducting quantum interference device magnetometer. The XRD pattern could be indexed to both the rhombohedral α–Fe2O3 and cubic bixbyite In2O3 phases. This heterostructure system showed ferromagnetic properties (due to the presence of γ–Fe2O3 phase) from 5 to 300 K and a spin-glass-like behavior of magnetization versus temperature under zero-field-cooled and field-cooled conditions. H2 gas-sensing property was observed from 100 ppm to 2 ppm at 200 °C and 250 °C. Response and recovery times were about 275 s and 500 s, respectively, and the sensitivity varied from 2% to 21% as the H2 increased from 2 to 100 ppm. The Ra/Rg varied from 1 to 1.3 and the plot of Ra/Rg versus H2 could be fitted to the sigmoidal logistic function, y = A2 + (A1 − A2) / (1 + (x/x0)p.
Graphic Abstract









Similar content being viewed by others
References
I. Dincer, and C. Acar, Int. J. Hydrogen Energy 43, 8579 (2018). https://doi.org/10.1016/j.ijhydene.2018.03.120.
T. Kamal, J. Alloys Compd. 729, 1058 (2017). https://doi.org/10.1016/j.jallcom.2017.09.124.
J.H. Kim, and A. Mirzaei, H Woo Kim, P Wu, and SS KimSens. Actuators B Chem. 293, 210 (2019). https://doi.org/10.1016/j.snb.2019.04.113.
A. Mirzaei, J.H. Kim, H.W. Kim, and S.S. Kim, Appl. Sci. 9, 1775 (2019). https://doi.org/10.3390/app9091775.
A. Katoch, J.H. Kim, Y.J. Kwon, H.W. Kim, S.S. Kim, and A.C.S. Appl, Mater. Interfaces 7, 11351 (2015). https://doi.org/10.1021/acsami.5b01817.
N. Van Toan, N. Viet Chien, N. Van Duy, H. Si Hong, H. Nguyen, H. Duc Hoa, and N. Van Hieu, J. Hazard. Mater. 301, 433 (2016). https://doi.org/10.1016/j.jhazmat.2015.09.013.
T. Hübert, L. Boon-Brett, V. Palmisano, and M.A. Bader, Int. J. Hydrogen Energy 39, 20474 (2014). https://doi.org/10.1016/j.ijhydene.2014.05.042.
X. Li, J. Feng, W. Mao, F. Yin, and J. Jiang, J. Mater. Chem. C 8, 14386 (2020). https://doi.org/10.1039/d0tc03622f.
T. Hübert, L. Boon-Brett, G. Black, and U. Banach, Sensors Actuators B Chem. 157, 329 (2011). https://doi.org/10.1016/j.snb.2011.04.070.
E. Brauns, E. Morsbach, S. Kunz, M. Bäumer, and W. Lang, Sens. Actuators B Chem. 193, 895 (2014). https://doi.org/10.1016/j.snb.2013.11.048.
R. Tabassum, and B.D. Gupta, Appl. Opt. 54, 1032 (2015). https://doi.org/10.1364/AO.54.001032.
K. Wetchakun, T. Samerjai, N. Tamaekong, C. Liewhiran, C. Siriwong, V. Kruefu, A. Wisitsoraat, A. Tuantranont, and S. Phanichphant, Sens. Actuators B Chem. 160, 580 (2011). https://doi.org/10.1016/j.snb.2011.08.032.
A. Gurlo, Nanoscale 3, 154 (2011). https://doi.org/10.1039/c0nr00560f.
B. Cao, J. Chen, X. Tang, and W. Zhou, J. Mater. Chem. 19, 2323 (2009). https://doi.org/10.1039/b816646c.
H.J. Song, X.H. Jia, H. Qi, X.F. Yang, H. Tang, and C.Y. Min, J. Mater. Chem. 22, 3508 (2012). https://doi.org/10.1039/C2JM13574D.
J. Li, H. Fan, and X. Jia, J. Phys. Chem. C 114, 14684 (2010). https://doi.org/10.1021/jp100792c.
T. Waitz, T. Wagner, T. Sauerwald, C.D. Kohl, and M. Tiemann, Adv. Funct. Mater. 19, 653 (2009). https://doi.org/10.1002/adfm.200801458.
S. Vallejos, T. Stoycheva, P. Umek, C. Navio, R. Snyders, C. Bittencourt, E. Llobet, C. Blackman, S. Moniz, and X. Correig, Chem. Commun. 47, 565 (2011). https://doi.org/10.1039/c0cc02398a.
Y. Wang, X. Jiang, and Y. Xia, J. Am. Chem. Soc. 125, 16176 (2003). https://doi.org/10.1021/ja037743f.
D. Chu, Y.-P. Zeng, D. Jiang, and Y. Masuda, Sensors Actuators B Chem. 137, 630 (2009). https://doi.org/10.1016/j.snb.2008.12.063.
P. Sun, Y. Cai, S. Du, X. Xu, L. You, J. Ma, F. Liu, X. Liang, Y. Sun, and G. Lu, Sens. Actuators B Chem. 182, 336 (2013). https://doi.org/10.1016/j.snb.2013.03.019.
H.-L. Yu, L. Li, X.M. Gao, Y. Zhang, F. Meng, T.-S. Wang, G. Xiao, Y.J. Chen, and C.L. Zhu, Sens. Actuators B Chem. 171–172, 679 (2012). https://doi.org/10.1016/j.snb.2012.05.054.
S. Aygün, and D. Cann, J. Phys. Chem. B 109, 7878 (2005). https://doi.org/10.1021/jp044481a.
M. Ivanovskaya, D. Kotsikau, G. Faglia, P. Nelli, and S. Irkaev, Sens. Actuators B Chem. 93, 422 (2003). https://doi.org/10.1016/S0925-4005(03)00175-8.
M. Ivanovskaya, D. Kotsikau, G. Faglia, and P. Nelli, Sens. Actuators B Chem. 96, 498 (2003). https://doi.org/10.1016/S0925-4005(03)00624-5.
T. Zhou, L. Wei, Y. Xie, Q. Li, G. Hu, Y. Chen, S. Yan, G. Liu, L. Mei, and J. Jiao, Nanoscale Res. Lett. 7, 661 (2012). https://doi.org/10.1186/1556-276X-7-661.
G. Han, Q. Lu, G. Liu, X. Ye, S. Lin, Y. Song, B. Liu, X. Yang, and G. Li, J. Mater. Sci. Mater. Electron. 23, 1616 (2012). https://doi.org/10.1007/s10854-012-0638-4.
P. Li, Y. Cai, and H. Fan, RSC Adv. 3, 22239 (2013). https://doi.org/10.1039/C3RA43616K.
R.K. Gupta, K. Ghosh, and P.K. Kahol, Mater. Lett. 64, 2022 (2010). https://doi.org/10.1016/j.matlet.2010.06.026.
D. Bérardan, and E. Guilmeau, J. Phys. Condens. Matter 19, 236224 (2007). https://doi.org/10.1088/0953-8984/19/23/236224.
A. Singhal, S.N. Achary, J. Manjanna, O.D. Jayakumar, R.M. Kadam, and A.K. Tyagi, J. Phys. Chem. C 113, 3600 (2009). https://doi.org/10.1021/jp8097846.
S. Kohiki, Y. Murakawa, K. Hori, H. Shimooka, T. Tajiri, H. Deguchi, M. Oku, M. Arai, M. Mitome, and Y. Bando, Jpn. J. Appl. Phys. 44, L979 (2005). https://doi.org/10.1143/JJAP.44.L979.
T.-R. Rashid, D.-T. Phan, and G.-S. Chung, Sens. Actuators B Chem. 185, 777 (2013). https://doi.org/10.1016/j.snb.2013.01.015.
C. Fahed, S.B. Qadri, H. Kim, A. Piqué, M. Miller, N.A. Mahadik, M.V. Rao, and M. Osofsky, Phys. Status Solidi 7, 2298 (2010). https://doi.org/10.1002/pssc.200983703.
M. Ristić, S. Popović, M. Tonković, and S. Musić, J. Mater. Sci. 26, 4225 (1991). https://doi.org/10.1007/BF02402973.
M. Sorescu, T. Xu, L. Diamandescu, and D. Hileman, Hyperfine Interact. 199, 365 (2011). https://doi.org/10.1007/s10751-011-0267-y.
M.I. Ivanovskaya, D.A. Kotsikau, A. Taurino, and P. Siciliano, Sens. Actuators B Chem. 124, 133 (2007).
Z. Jing, Mater. Sci. Eng. A 441, 176 (2006). https://doi.org/10.1016/j.msea.2006.08.013.
J.-M. Tulliani, and P. Bonville, Ceram. Int. 31, 507 (2005). https://doi.org/10.1016/j.ceramint.2004.06.015.
G. Neri, A. Bonavita, N. Galvagno, N. Donato, and A. Caddemi, Sensors Actuators B Chem. 111–112, 71 (2005). https://doi.org/10.1016/j.snb.2005.06.061.
I. Nodari, A. Alebouyeh, J.F. Brice, R. Gérardin, and O. Evrard, Mater. Res. Bull. 23, 1039 (1988). https://doi.org/10.1016/0025-5408(88)90060-8.
F. Brown, M.J. Flores, N. Kimizuka, Y. Michiue, M. Onoda, T. Mohri, M. Nakamura, and N. Ishizawa, J. Solid State Chem. 144, 91 (1999). https://doi.org/10.1006/jssc.1998.8123.
M. Pernet, J.C. Joubert, and C. Berthet-Colominas, Solid State Commun. 17, 1505 (1975). https://doi.org/10.1016/0038-1098(75)90983-7.
A. Gurlo, M. Ivanovskaya, N. Barsan, and U. Weimar, Inorg. Chem. Commun. 6, 569 (2003). https://doi.org/10.1016/S1387-7003(03)00047-9.
J. Feng, X. Li, G. Zhu, Q.J. Wang A.C.S. Appl. Mater. Interfaces 12, 43098 (2020). https://doi.org/10.1021/acsami.0c12907.
E.E. Gutman, Sens. Actuators B: Chem. 23, 209 (1995). https://doi.org/10.1016/0925-4005(94)01276-N.
U. Schwertmann, and R. M. Cornell, Iron Oxides in the Laboratory ‐ Preparation and Characterization, ISBN 978-3-527-26991-6, 2nd Ed., Wiley-VCH, 2008.
J.L. Jambor, and J.E. Dutrizac, Chem. Rev. 98, 2549 (1998). https://doi.org/10.1021/cr970105t.
L. E. Smart, E. A. Moore, Solid State Chemistry: An Introduction, ISBN 0-203-49635-3, 3rd Ed., CRC, 2005.
R.K. Chava, S.Y. Oh, and Y.T. Yu, CrystEngComm 18, 3655 (2016). https://doi.org/10.1039/C6CE00352D.
J. Hu, Y. Sun, Y. Xue, M. Zhang, P. Li, K. Lian, S. Zhuiykov, W. Zhang, and Y. Chen, Sens. Actuators B Chem. 257, 124 (2018). https://doi.org/10.1016/j.snb.2017.10.139.
X. Li, X. Li, N. Chen, X. Li, J. Zhang, J. Yu, J. Wang, and Z. Tang, J. Nanomater. 2014, 973156 (2014). https://doi.org/10.1155/2014/973156.
Z.Q. Zheng, L.F. Zhu, and B. Wang, Nanoscale Res. Lett. 10, 293 (2015). https://doi.org/10.1186/s11671-015-1002-4.
K. Hara, and N. Nishida, Sens. Actuators B Chem. 20, 181 (1994). https://doi.org/10.1016/0925-4005(94)01181-8.
J.-H. Lee, J.-H. Kim, J.-Y. Kim, A. Mirzaei, H.W. Kim, and S.S. Kim, Sensors 19, 4276 (2019). https://doi.org/10.3390/s19194276.
Z. Li, S. Yan, Z. Wu, H. Li, J. Wang, W. Shen, Z. Wang, and Y. Fu, Int. J. Hydrogen Energy 43, 22746 (2018). https://doi.org/10.1016/j.ijhydene.2018.10.101.
C. Wang, X. Cheng, X. Zhou, P. Sun, X. Hu, K. Shimanoe, G. Lu, N. Yamazoe, A.C.S. Appl, Mater. Interfaces 6, 12031 (2014). https://doi.org/10.1021/am501063z.
H.D. Chen, K.L. Jin, P.F. Wang, J.C. Xu, Y.B. Han, H.X. Jin, D.F. Jin, X.L. Peng, B. Hong, J. Li, Y.T. Yang, J. Gong, H.L. Ge, and X.Q. Wang, J. Phys. Chem. Solids 120, 271 (2018). https://doi.org/10.1016/j.jpcs.2018.05.004.
M.S. Kiruba, A.S. Jose, K. Prajwal, P. Chowdhury, and H.C. Barshilia, Sensors Actuators B Chem. 310, 127830 (2020). https://doi.org/10.1016/j.snb.2020.127830.
F. Zhang, X. Li, Q. Zhao, Q. Zhang, M. Tadé, and S. Liu, J. Colloid Interface Sci. 457, 18 (2015). https://doi.org/10.1016/j.jcis.2015.06.008.
J. Cai, S. Li, H. Pan, Y. Liu, and G. Qin, J. Mater. Sci. 51, 8148 (2016). https://doi.org/10.1007/s10853-016-0085-3.
D. Kotsikau, and M. Ivanovskaya, Mater. Chem. Phys. 160, 337 (2015). https://doi.org/10.1016/j.matchemphys.2015.04.047.
M. Sorescu, L. Diamandescu, and D. Tarabasanu-Mihaila, J. Phys. Chem. Solids 65, 1719 (2004). https://doi.org/10.1016/j.jpcs.2004.05.002.
M. Hermanek, R. Zboril, M. Mashlan, L. Machala, and O. Schneeweiss, J. Mater. Chem. 16, 1273 (2006). https://doi.org/10.1039/b514565a.
K.S. Rane, A.K. Nikumbh, and A.J. Mukhedkar, J. Mater. Sci. 16, 2387 (1981). https://doi.org/10.1007/BF01113573.
M. Aliahmad, and N. Nasiri Moghaddam, MaterSci. 31, 264 (2013). https://doi.org/10.2478/s13536-012-0100-6.
Z. Li, H.-F. Fei, Y. Tan, X. Zhang, Z. Xie, and Z. Zhang, RSC Adv. 5, 38093 (2015). https://doi.org/10.1039/C5RA05968B.
J. Manjanna, and G. Venkateswaran, Ind. Eng. Chem. Res. 41, 3053 (2002). https://doi.org/10.1021/ie010344d.
G. Hosamani, B.N. Jagadale, J. Manjanna, S.M. Shivaprasad, D.K. Shukla, and J.S. Bhat, J. Electron. Mater. 50, 52 (2021). https://doi.org/10.1007/s11664-020-08553-5.
D. Cao, H. Li, L. Pan, J. Li, X. Wang, P. Jing, X. Cheng, W. Wang, J. Wang, and Q. Liu, Sci. Rep. 6, 32360 (2016). https://doi.org/10.1038/srep32360.
A. Mirzaei, B. Hashemi, and K. Janghorban, J. Mater. Sci. Mater. Electron. 27, 3109 (2016). https://doi.org/10.1007/s10854-015-4200-z.
D.M. Chethana, T.C. Thanuja, H.M. Mahesh, M.S. Kiruba, A.S. Jose, H.C. Barshilia, and J. Manjanna, Ceram. Int. 47, 10381 (2020). https://doi.org/10.1016/j.ceramint.2020.06.129.
W. Wang, Y. Zhang, J. Zhang, G. Li, D. Leng, Y. Gao, J. Gao, H. Lu, and X. Li, Sens. Actuators B Chem. 328, 129045 (2021). https://doi.org/10.1016/j.snb.2020.129045.
Acknowledgments
Authors thank Prof. Satoru Kobayashi, Faculty of Engineering, Iwate University, Japan, and Mr. Dinesh S. Patil, Dept. of Chemistry, RCU, India, for their help during this work. Prof. J Manjanna gratefully acknowledge the financial support from DST-FIST, Govt. of India [SR/FST/CSI-273/2016].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chethana, D.M., Thanuja, T.C., Mahesh, H.M. et al. Heterostructure Fe2O3–In2O3 Nanoparticles as Hydrogen Gas Sensor. Journal of Elec Materi 50, 4313–4323 (2021). https://doi.org/10.1007/s11664-021-08951-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-021-08951-3