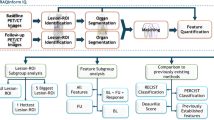
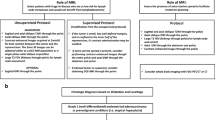
Abstract
Introduction
The advent of image-guided radiation therapy (IGRT) has recently changed the workflow of radiation treatments by ensuring highly collimated treatments. Artificial intelligence (AI) and radiomics are tools that have shown promising results for diagnosis, treatment optimization and outcome prediction. This review aims to assess the impact of AI and radiomics on modern IGRT modalities in RT.
Methods
A PubMed/MEDLINE and Embase systematic review was conducted to investigate the impact of radiomics and AI to modern IGRT modalities. The search strategy was “Radiomics” AND “Cone Beam Computed Tomography”; “Radiomics” AND “Magnetic Resonance guided Radiotherapy”; “Radiomics” AND “on board Magnetic Resonance Radiotherapy”; “Artificial Intelligence” AND “Cone Beam Computed Tomography”; “Artificial Intelligence” AND “Magnetic Resonance guided Radiotherapy”; “Artificial Intelligence” AND “on board Magnetic Resonance Radiotherapy” and only original articles up to 01.11.2022 were considered.
Results
A total of 402 studies were obtained using the previously mentioned search strategy on PubMed and Embase. The analysis was performed on a total of 84 papers obtained following the complete selection process. Radiomics application to IGRT was analyzed in 23 papers, while a total 61 papers were focused on the impact of AI on IGRT techniques.
Discussion
AI and radiomics seem to significantly impact IGRT in all the phases of RT workflow, even if the evidence in the literature is based on retrospective data. Further studies are needed to confirm these tools' potential and provide a stronger correlation with clinical outcomes and gold-standard treatment strategies.

Similar content being viewed by others
References
Ramesh AN, Kambhampati C, Monson JRT, Drew PJ (2004) Artificial intelligence in medicine. Ann R Coll Surg Engl 86:334–338. https://doi.org/10.1308/147870804290
Vandewinckele L, Claessens M, Dinkla A et al (2020) Overview of artificial intelligence-based applications in radiotherapy: recommendations for implementation and quality assurance. Radiother Oncol 153:55–66. https://doi.org/10.1016/j.radonc.2020.09.008
Huynh E, Hosny A, Guthier C et al (2020) Artificial intelligence in radiation oncology. Nat Rev Clin Oncol 17:771–781. https://doi.org/10.1038/s41571-020-0417-8
Cesario A, D’Oria M, Calvani R et al (2021) The role of artificial intelligence in managing multimorbidity and cancer. J Pers Med 11:314. https://doi.org/10.3390/jpm11040314
Cusumano D, Lenkowicz J, Votta C et al (2020) A deep learning approach to generate synthetic CT in low field MR-guided adaptive radiotherapy for abdominal and pelvic cases. Radiother Oncol 153:205–212. https://doi.org/10.1016/j.radonc.2020.10.018
Gambacorta MA, Valentini C, Dinapoli N et al (2013) Clinical validation of atlas-based auto-segmentation of pelvic volumes and normal tissue in rectal tumors using auto-segmentation computed system. Acta Oncol 52:1676–1681. https://doi.org/10.3109/0284186X.2012.754989
Fionda B, Boldrini L, D’Aviero A et al (2020) Artificial intelligence (AI) and interventional radiotherapy (brachytherapy): state of art and future perspectives. J Contemp Brachyther 12:497–500. https://doi.org/10.5114/jcb.2020.100384
Nardone V, Reginelli A, Grassi R et al (2021) Delta radiomics: a systematic review. Radiol Med 126:1571–1583. https://doi.org/10.1007/s11547-021-01436-7
Cusumano D, Boldrini L, Yadav P et al (2021) Delta radiomics analysis for local control prediction in pancreatic cancer patients treated using magnetic resonance guided radiotherapy. Diagnostics 11:72. https://doi.org/10.3390/diagnostics11010072
Casà C, Piras A, D’Aviero A et al (2022) The impact of radiomics in diagnosis and staging of pancreatic cancer. Ther Adv Gastrointest Endosc 15:26317745221081596. https://doi.org/10.1177/26317745221081596
Cusumano D, Boldrini L, Yadav P et al (2021) Delta radiomics for rectal cancer response prediction using low field magnetic resonance guided radiotherapy: an external validation. Phys Med 84:186–191. https://doi.org/10.1016/j.ejmp.2021.03.038
Alongi P, Stefano A, Comelli A et al (2021) Radiomics analysis of 18F-Choline PET/CT in the prediction of disease outcome in high-risk prostate cancer: an explorative study on machine learning feature classification in 94 patients. Eur Radiol 31:4595–4605. https://doi.org/10.1007/s00330-020-07617-8
Chiloiro G, Rodriguez-Carnero P, Lenkowicz J et al (2020) Delta radiomics can predict distant metastasis in locally advanced rectal cancer: the challenge to personalize the cure. Front Oncol 10:595012. https://doi.org/10.3389/fonc.2020.595012
Boldrini L, Cusumano D, Chiloiro G et al (2019) Delta radiomics for rectal cancer response prediction with hybrid 0.35 T magnetic resonance-guided radiotherapy (MRgRT): a hypothesis-generating study for an innovative personalized medicine approach. Radiol Med 124:145–153. https://doi.org/10.1007/s11547-018-0951-y
Lambin P, Rios-Velazquez E, Leijenaar R et al (2012) Radiomics: extracting more information from medical images using advanced feature analysis. Eur J Cancer 48:441–446. https://doi.org/10.1016/j.ejca.2011.11.036
Gardin I, Grégoire V, Gibon D et al (2019) Radiomics: principles and radiotherapy applications. Crit Rev Oncol Hematol 138:44–50. https://doi.org/10.1016/j.critrevonc.2019.03.015
Luh JY, Albuquerque KV, Cheng C et al (2020) ACR-ASTRO practice parameter for image-guided radiation therapy (IGRT). Am J Clin Oncol 43:459–468. https://doi.org/10.1097/COC.0000000000000697
Gillan C, Giuliani M, Harnett N et al (2016) Image guided radiation therapy: unlocking the future through knowledge translation. Int J Radiat Oncol Biol Phys 96:248–250. https://doi.org/10.1016/j.ijrobp.2016.05.028
Sharma M, Nano TF, Akkati M et al (2022) A systematic review and meta-analysis of liver tumor position variability during SBRT using various motion management and IGRT strategies. Radiother Oncol 166:195–202. https://doi.org/10.1016/j.radonc.2021.11.022
Lee J, Liu S-H, Lin J-B et al (2018) Image-guided study of inter-fraction and intra-fraction set-up variability and margins in reverse semi-decubitus breast radiotherapy. Radiat Oncol 13:254. https://doi.org/10.1186/s13014-018-1200-1
Corradini S, Alongi F, Andratschke N et al (2019) MR-guidance in clinical reality: current treatment challenges and future perspectives. Radiat Oncol 14:92. https://doi.org/10.1186/s13014-019-1308-y
Boldrini L, Cusumano D, Cellini F et al (2019) Online adaptive magnetic resonance guided radiotherapy for pancreatic cancer: state of the art, pearls and pitfalls. Radiat Oncol 14:71. https://doi.org/10.1186/s13014-019-1275-3
Zhang Y, Liang Y, Ding J et al (2022) A prior knowledge-guided, deep learning-based semiautomatic segmentation for complex anatomy on magnetic resonance imaging. Int J Radiat Oncol Biol Phys 114:349–359. https://doi.org/10.1016/j.ijrobp.2022.05.039
D’Aviero A, Re A, Catucci F et al (2022) Clinical validation of a deep-learning segmentation software in head and neck: an early analysis in a Developing Radiation Oncology Center. Int J Environ Res Public Health 19:9057. https://doi.org/10.3390/ijerph19159057
Brunt AM, Haviland JS, Wheatley DA et al (2020) Hypofractionated breast radiotherapy for 1 week versus 3 weeks (FAST-Forward): 5-year efficacy and late normal tissue effects results from a multicentre, non-inferiority, randomised, phase 3 trial. The Lancet 395:1613–1626. https://doi.org/10.1016/S0140-6736(20)30932-6
Piras A, Menna S, D’Aviero A et al (2021) New fractionations in breast cancer: a dosimetric study of 3D-CRT versus VMAT. J Med Radiat Sci. https://doi.org/10.1002/jmrs.530
Ling DC, Vargo JA, Beriwal S (2020) Breast, prostate, and rectal cancer: Should 5–5-5 be a new standard of care? Int J Radiat Oncol Biol Phys 108:390–393. https://doi.org/10.1016/j.ijrobp.2020.06.049
Piras A, Boldrini L, Menna S et al (2021) Hypofractionated radiotherapy in head and neck cancer elderly patients: a feasibility and safety systematic review for the clinician. Front Oncol 11:761393. https://doi.org/10.3389/fonc.2021.761393
Massaccesi M, Boldrini L, Piras A et al (2020) Spatially fractionated radiotherapy (SFRT) targeting the hypoxic tumor segment for the intentional induction of non-targeted effects: an in silico study to exploit a new treatment paradigm. Tech Innov Patient Support Radiat Oncol 14:11–14. https://doi.org/10.1016/j.tipsro.2020.02.003
Piras A, Venuti V, D’Aviero A et al (2022) Covid-19 and radiotherapy: a systematic review after 2 years of pandemic. Clin Transl Imaging 66:1–20. https://doi.org/10.1007/s40336-022-00513-9
Page MJ, McKenzie JE, Bossuyt PM et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71. https://doi.org/10.1136/bmj.n71
Tomaszewski MR, Latifi K, Boyer E et al (2021) Delta radiomics analysis of magnetic resonance guided radiotherapy imaging data can enable treatment response prediction in pancreatic cancer. Radiat Oncol 16:237. https://doi.org/10.1186/s13014-021-01957-5
Chiloiro G, Boldrini L, Preziosi F et al (2022) A predictive model of 2yDFS during MR-guided RT neoadjuvant chemoradiotherapy in locally advanced rectal cancer patients. Front Oncol 12:831712. https://doi.org/10.3389/fonc.2022.831712
Simpson G, Jin W, Spieler B et al (2022) Predictive value of delta-radiomics texture features in 0.35 Tesla magnetic resonance setup images acquired during stereotactic ablative radiotherapy of pancreatic cancer. Front Oncol 12:807725. https://doi.org/10.3389/fonc.2022.807725
Xue C, Yuan J, Poon DM et al (2021) Reliability of MRI radiomics features in MR-guided radiotherapy for prostate cancer: repeatability, reproducibility, and within-subject agreement. Med Phys 48:6976–6986. https://doi.org/10.1002/mp.15232
van Timmeren JE, van Elmpt W, Leijenaar RTH et al (2019) Longitudinal radiomics of cone-beam CT images from non-small cell lung cancer patients: evaluation of the added prognostic value for overall survival and locoregional recurrence. Radiother Oncol 136:78–85. https://doi.org/10.1016/j.radonc.2019.03.032
Qin Q, Shi A, Zhang R et al (2020) Cone-beam CT radiomics features might improve the prediction of lung toxicity after SBRT in stage I NSCLC patients. Thorac Cancer 11:964–972. https://doi.org/10.1111/1759-7714.13349
Bosetti DG, Ruinelli L, Piliero MA et al (2020) Cone-beam computed tomography-based radiomics in prostate cancer: a mono-institutional study. Strahlenther Onkol 196:943–951. https://doi.org/10.1007/s00066-020-01677-x
Shi L, Rong Y, Daly M et al (2020) Cone-beam computed tomography-based delta-radiomics for early response assessment in radiotherapy for locally advanced lung cancer. Phys Med Biol 65:015009. https://doi.org/10.1088/1361-6560/ab3247
Morgan HE, Wang K, Dohopolski M et al (2021) Exploratory ensemble interpretable model for predicting local failure in head and neck cancer: the additive benefit of CT and intra-treatment cone-beam computed tomography features. Quant Imaging Med Surg 11:4781–4796. https://doi.org/10.21037/qims-21-274
Du F, Tang N, Cui Y et al (2020) A novel nomogram model based on cone-beam CT radiomics analysis technology for predicting radiation pneumonitis in esophageal cancer patients undergoing radiotherapy. Front Oncol 10:596013. https://doi.org/10.3389/fonc.2020.596013
Sellami S, Bourbonne V, Hatt M et al (2022) Predicting response to radiotherapy of head and neck squamous cell carcinoma using radiomics from cone-beam CT images. Acta Oncol 61:73–80. https://doi.org/10.1080/0284186X.2021.1983207
Iliadou V, Kakkos I, Karaiskos P et al (2022) Early prediction of planning adaptation requirement indication due to volumetric alterations in head and neck cancer radiotherapy: a machine learning approach. Cancers 14:3573. https://doi.org/10.3390/cancers14153573
Zhang R, Cai Z, Luo Y et al (2022) Preliminary exploration of response the course of radiotherapy for stage III non-small cell lung cancer based on longitudinal CT radiomics features. Eur J Radiol Open 9:100391. https://doi.org/10.1016/j.ejro.2021.100391
van Timmeren JE, Leijenaar RTH, van Elmpt W et al (2017) Feature selection methodology for longitudinal cone-beam CT radiomics. Acta Oncol 56:1537–1543. https://doi.org/10.1080/0284186X.2017.1350285
van Timmeren JE, Leijenaar RTH, van Elmpt W et al (2017) Survival prediction of non-small cell lung cancer patients using radiomics analyses of cone-beam CT images. Radiother Oncol 123:363–369. https://doi.org/10.1016/j.radonc.2017.04.016
Delgadillo R, Spieler BO, Ford JC et al (2021) Repeatability of CBCT radiomic features and their correlation with CT radiomic features for prostate cancer. Med Phys 48:2386–2399. https://doi.org/10.1002/mp.14787
Bagher-Ebadian H, Siddiqui F, Liu C et al (2017) On the impact of smoothing and noise on robustness of CT and CBCT radiomics features for patients with head and neck cancers. Med Phys 44:1755–1770. https://doi.org/10.1002/mp.12188
Fave X, Mackin D, Yang J et al (2015) Can radiomics features be reproducibly measured from CBCT images for patients with non-small cell lung cancer? Med Phys 42:6784–6797. https://doi.org/10.1118/1.4934826
Wang H, Zhou Y, Wang X et al (2021) Reproducibility and repeatability of CBCT-derived radiomics features. Front Oncol 11:773512. https://doi.org/10.3389/fonc.2021.773512
Gu J, Zhu J, Qiu Q et al (2018) The feasibility study of Megavoltage Computed Tomographic (MVCT) image for texture feature analysis. Front Oncol 8:586. https://doi.org/10.3389/fonc.2018.00586
Scholey JE, Rajagopal A, Vasquez EG et al (2022) Generation of synthetic megavoltage CT for MRI-only radiotherapy treatment planning using a 3D deep convolutional neural network. Med Phys 49:6622–6634. https://doi.org/10.1002/mp.15876
Liang X, Chen L, Nguyen D et al (2019) Generating synthesized computed tomography (CT) from cone-beam computed tomography (CBCT) using CycleGAN for adaptive radiation therapy. Phys Med Biol 64:125002. https://doi.org/10.1088/1361-6560/ab22f9
Yuan N, Dyer B, Rao S et al (2020) Convolutional neural network enhancement of fast-scan low-dose cone-beam CT images for head and neck radiotherapy. Phys Med Biol 65:035003. https://doi.org/10.1088/1361-6560/ab6240
Irmak S, Zimmermann L, Georg D et al (2021) Cone beam CT based validation of neural network generated synthetic CTs for radiotherapy in the head region. Med Phys 48:4560–4571. https://doi.org/10.1002/mp.14987
Chen L, Liang X, Shen C et al (2021) Synthetic CT generation from CBCT images via unsupervised deep learning. Phys Med Biol. https://doi.org/10.1088/1361-6560/ac01b6
Gao L, Xie K, Wu X et al (2021) Generating synthetic CT from low-dose cone-beam CT by using generative adversarial networks for adaptive radiotherapy. Radiat Oncol 16:202. https://doi.org/10.1186/s13014-021-01928-w
Qiu RLJ, Lei Y, Shelton J et al (2021) Deep learning-based thoracic CBCT correction with histogram matching. Biomed Phys Eng Express. https://doi.org/10.1088/2057-1976/ac3055
Xue X, Ding Y, Shi J et al (2021) Cone Beam CT (CBCT) based synthetic CT generation using deep learning methods for dose calculation of nasopharyngeal carcinoma radiotherapy. Technol Cancer Res Treat 20:15330338211062416. https://doi.org/10.1177/15330338211062415
Liu J, Yan H, Cheng H et al (2021) CBCT-based synthetic CT generation using generative adversarial networks with disentangled representation. Quant Imaging Med Surg 11:4820–4834. https://doi.org/10.21037/qims-20-1056
Zhang Y, Yue N, Su M-Y et al (2021) Improving CBCT quality to CT level using deep learning with generative adversarial network. Med Phys 48:2816–2826. https://doi.org/10.1002/mp.14624
Chen L, Liang X, Shen C et al (2020) Synthetic CT generation from CBCT images via deep learning. Med Phys 47:1115–1125. https://doi.org/10.1002/mp.13978
Uh J, Wang C, Acharya S et al (2021) Training a deep neural network coping with diversities in abdominal and pelvic images of children and young adults for CBCT-based adaptive proton therapy. Radiother Oncol 160:250–258. https://doi.org/10.1016/j.radonc.2021.05.006
Lemus OMD, Wang Y-F, Li F et al (2022) Dosimetric assessment of patient dose calculation on a deep learning-based synthesized computed tomography image for adaptive radiotherapy. J Appl Clin Med Phys 23:e13595. https://doi.org/10.1002/acm2.13595
Wu W, Qu J, Cai J, Yang R (2022) Multiresolution residual deep neural network for improving pelvic CBCT image quality. Med Phys 49:1522–1534. https://doi.org/10.1002/mp.15460
Kurosawa T, Nishio T, Moriya S et al (2020) Feasibility of image quality improvement for high-speed CBCT imaging using deep convolutional neural network for image-guided radiotherapy in prostate cancer. Phys Med 80:84–91. https://doi.org/10.1016/j.ejmp.2020.10.012
Thummerer A, Seller Oria C, Zaffino P et al (2021) Clinical suitability of deep learning based synthetic CTs for adaptive proton therapy of lung cancer. Med Phys 48:7673–7684. https://doi.org/10.1002/mp.15333
Li Y, Zhu J, Liu Z et al (2019) A preliminary study of using a deep convolution neural network to generate synthesized CT images based on CBCT for adaptive radiotherapy of nasopharyngeal carcinoma. Phys Med Biol 64:145010. https://doi.org/10.1088/1361-6560/ab2770
Thummerer A, de Jong BA, Zaffino P et al (2020) Comparison of the suitability of CBCT- and MR-based synthetic CTs for daily adaptive proton therapy in head and neck patients. Phys Med Biol 65:235036. https://doi.org/10.1088/1361-6560/abb1d6
Maspero M, Houweling AC, Savenije MHF et al (2020) A single neural network for cone-beam computed tomography-based radiotherapy of head-and-neck, lung and breast cancer. Phys Imaging Radiat Oncol 14:24–31. https://doi.org/10.1016/j.phro.2020.04.002
Sibolt P, Andersson LM, Calmels L et al (2021) Clinical implementation of artificial intelligence-driven cone-beam computed tomography-guided online adaptive radiotherapy in the pelvic region. Phys Imaging Radiat Oncol 17:1–7. https://doi.org/10.1016/j.phro.2020.12.004
Li R, Roy A, Bice N et al (2021) Managing tumor changes during radiotherapy using a deep learning model. Med Phys 48:5152–5164. https://doi.org/10.1002/mp.14925
Han X, Hong J, Reyngold M et al (2021) Deep-learning-based image registration and automatic segmentation of organs-at-risk in cone-beam CT scans from high-dose radiation treatment of pancreatic cancer. Med Phys 48:3084–3095. https://doi.org/10.1002/mp.14906
Jiang J, Riyahi Alam S, Chen I et al (2021) Deep cross-modality (MR-CT) educed distillation learning for cone beam CT lung tumor segmentation. Med Phys 48:3702–3713. https://doi.org/10.1002/mp.14902
Alam SR, Li T, Zhang P et al (2021) Generalizable cone beam CT esophagus segmentation using physics-based data augmentation. Phys Med Biol 66:065008. https://doi.org/10.1088/1361-6560/abe2eb
Liang X, Bibault J-E, Leroy T et al (2021) Automated contour propagation of the prostate from pCT to CBCT images via deep unsupervised learning. Med Phys 48:1764–1770. https://doi.org/10.1002/mp.14755
Schreier J, Genghi A, Laaksonen H et al (2020) Clinical evaluation of a full-image deep segmentation algorithm for the male pelvis on cone-beam CT and CT. Radiother Oncol 145:1–6. https://doi.org/10.1016/j.radonc.2019.11.021
Åström LM, Behrens CP, Calmels L et al (2022) Online adaptive radiotherapy of urinary bladder cancer with full re-optimization to the anatomy of the day: Initial experience and dosimetric benefits. Radiother Oncol 171:37–42. https://doi.org/10.1016/j.radonc.2022.03.014
Wang C, Alam RS, Zhang S et al (2020) Predicting spatial esophageal changes in a multimodal longitudinal imaging study via a convolutional recurrent neural network. Phys Med Biol 65:235027. https://doi.org/10.1088/1361-6560/abb1d9
Lalonde A, Winey B, Verburg J et al (2020) Evaluation of CBCT scatter correction using deep convolutional neural networks for head and neck adaptive proton therapy. Phys Med Biol. https://doi.org/10.1088/1361-6560/ab9fcb
Harms J, Lei Y, Wang T et al (2020) Cone-beam CT-derived relative stopping power map generation via deep learning for proton radiotherapy. Med Phys 47:4416–4427. https://doi.org/10.1002/mp.14347
Luximon DC, Ritter T, Fields E et al (2022) Development and interinstitutional validation of an automatic vertebral-body misalignment error detector for cone-beam CT-guided radiotherapy. Med Phys 49:6410–6423. https://doi.org/10.1002/mp.15927
Liang X, Zhao W, Hristov DH et al (2020) A deep learning framework for prostate localization in cone beam CT-guided radiotherapy. Med Phys 47:4233–4240. https://doi.org/10.1002/mp.14355
Fu Y, Wang T, Lei Y et al (2021) Deformable MR-CBCT prostate registration using biomechanically constrained deep learning networks. Med Phys 48:253–263. https://doi.org/10.1002/mp.14584
Zhang S, Lv B, Zheng X et al (2022) Dosimetric study of deep learning-guided ITV prediction in cone-beam CT for lung stereotactic body radiotherapy. Front Public Health 10:860135. https://doi.org/10.3389/fpubh.2022.860135
Kai Y, Arimura H, Ninomiya K et al (2020) Semi-automated prediction approach of target shifts using machine learning with anatomical features between planning and pretreatment CT images in prostate radiotherapy. J Radiat Res 61:285–297. https://doi.org/10.1093/jrr/rrz105
Dohopolski M, Wang K, Morgan H et al (2022) Use of deep learning to predict the need for aggressive nutritional supplementation during head and neck radiotherapy. Radiother Oncol 171:129–138. https://doi.org/10.1016/j.radonc.2022.04.016
Cusumano D, Placidi L, Teodoli S et al (2020) On the accuracy of bulk synthetic CT for MR-guided online adaptive radiotherapy. Radiol Med 125:157–164. https://doi.org/10.1007/s11547-019-01090-0
Lenkowicz J, Votta C, Nardini M et al (2022) A deep learning approach to generate synthetic CT in low field MR-guided radiotherapy for lung cases. Radiother Oncol 176:31–38. https://doi.org/10.1016/j.radonc.2022.08.028
Terpstra ML, Maspero M, d’Agata F et al (2020) Deep learning-based image reconstruction and motion estimation from undersampled radial k-space for real-time MRI-guided radiotherapy. Phys Med Biol 65:155015. https://doi.org/10.1088/1361-6560/ab9358
Chun J, Zhang H, Gach HM et al (2019) MRI super-resolution reconstruction for MRI-guided adaptive radiotherapy using cascaded deep learning: In the presence of limited training data and unknown translation model. Med Phys 46:4148–4164. https://doi.org/10.1002/mp.13717
Olberg S, Chun J, Su Choi B et al (2021) Abdominal synthetic CT reconstruction with intensity projection prior for MRI-only adaptive radiotherapy. Phys Med Biol. https://doi.org/10.1088/1361-6560/ac279e
Olberg S, Zhang H, Kennedy WR et al (2019) Synthetic CT reconstruction using a deep spatial pyramid convolutional framework for MR-only breast radiotherapy. Med Phys 46:4135–4147. https://doi.org/10.1002/mp.13716
Thomas MA, Fu Y, Yang D (2020) Development and evaluation of machine learning models for voxel dose predictions in online adaptive magnetic resonance guided radiation therapy. J Appl Clin Med Phys 21:60–69. https://doi.org/10.1002/acm2.12884
Chen X, Ma X, Yan X et al (2022) Personalized auto-segmentation for magnetic resonance imaging-guided adaptive radiotherapy of prostate cancer. Med Phys 49:4971–4979. https://doi.org/10.1002/mp.15793
Tong N, Gou S, Yang S et al (2019) Shape constrained fully convolutional densenet with adversarial training for multi-organ segmentation on head and neck CT and low field MR images. Med Phys 46:2669–2682. https://doi.org/10.1002/mp.13553
Friedrich F, Hörner-Rieber J, Renkamp CK et al (2021) Stability of conventional and machine learning-based tumor auto-segmentation techniques using undersampled dynamic radial bSSFP acquisitions on a 0.35 T hybrid MR-linac system. Med Phys 48:587–596. https://doi.org/10.1002/mp.14659
Kawula M, Hadi I, Nierer L et al (2022) Patient-specific transfer learning for auto-segmentation in adaptive 0.35 T MRgRT of prostate cancer: a bi-centric evaluation. Med Phys. https://doi.org/10.1002/mp.16056
Chun J, Park JC, Olberg S et al (2022) Intentional deep overfit learning (IDOL): a novel deep learning strategy for adaptive radiation therapy. Med Phys 49:488–496. https://doi.org/10.1002/mp.15352
Liang F, Qian P, Su K-H et al (2018) Abdominal, multi-organ, auto-contouring method for online adaptive magnetic resonance guided radiotherapy: An intelligent, multi-level fusion approach. Artif Intell Med 90:34–41. https://doi.org/10.1016/j.artmed.2018.07.001
Eppenhof K, a. J, Maspero M, Savenije MHF, et al (2020) Fast contour propagation for MR-guided prostate radiotherapy using convolutional neural networks. Med Phys 47:1238–1248. https://doi.org/10.1002/mp.13994
Künzel LA, Nachbar M, Hagmüller M et al (2021) First experience of autonomous, un-supervised treatment planning integrated in adaptive MR-guided radiotherapy and delivered to a patient with prostate cancer. Radiother Oncol 159:197–201. https://doi.org/10.1016/j.radonc.2021.03.032
Hague C, McPartlin A, Lee LW et al (2021) An evaluation of MR based deep learning auto-contouring for planning head and neck radiotherapy. Radiother Oncol 158:112–117. https://doi.org/10.1016/j.radonc.2021.02.018
Chen Y, Ruan D, Xiao J et al (2020) Fully automated multiorgan segmentation in abdominal magnetic resonance imaging with deep neural networks. Med Phys 47:4971–4982. https://doi.org/10.1002/mp.14429
Huang L, Li M, Gou S et al (2021) Automated segmentation method for low field 3D stomach MRI using transferred learning image enhancement network. Biomed Res Int 2021:6679603. https://doi.org/10.1155/2021/6679603
Luximon DC, Abdulkadir Y, Chow PE et al (2022) Machine-assisted interpolation algorithm for semi-automated segmentation of highly deformable organs. Med Phys 49:41–51. https://doi.org/10.1002/mp.15351
Kajikawa T, Kadoya N, Tanaka S et al (2020) Dose distribution correction for the influence of magnetic field using a deep convolutional neural network for online MR-guided adaptive radiotherapy. Phys Med 80:186–192. https://doi.org/10.1016/j.ejmp.2020.11.002
Li M, Shan S, Chandra SS et al (2020) Fast geometric distortion correction using a deep neural network: Implementation for the 1 Tesla MRI-Linac system. Med Phys 47:4303–4315. https://doi.org/10.1002/mp.14382
Cerviño LI, Du J, Jiang SB (2011) MRI-guided tumor tracking in lung cancer radiotherapy. Phys Med Biol 56:3773. https://doi.org/10.1088/0031-9155/56/13/003
Liu L, Shen L, Johansson A et al (2022) Real time volumetric MRI for 3D motion tracking via geometry-informed deep learning. Med Phys 49:6110–6119. https://doi.org/10.1002/mp.15822
Gao Y, Ghodrati V, Kalbasi A et al (2021) Prediction of soft tissue sarcoma response to radiotherapy using longitudinal diffusion MRI and a deep neural network with generative adversarial network-based data augmentation. Med Phys 48:3262–3372. https://doi.org/10.1002/mp.14897
Fiorino C, Gumina C, Passoni P et al (2018) A TCP-based early regression index predicts the pathological response in neo-adjuvant radio-chemotherapy of rectal cancer. Radiother Oncol 128:564–568. https://doi.org/10.1016/j.radonc.2018.06.019
Salvestrini V, Greco C, Guerini AE et al (2022) The role of feature-based radiomics for predicting response and radiation injury after stereotactic radiation therapy for brain metastases: a critical review by the Young Group of the Italian Association of Radiotherapy and Clinical Oncology (yAIRO). Transl Oncol 15:101–275. https://doi.org/10.1016/j.tranon.2021.101275
Chiloiro G, Cusumano D, Boldrini L et al (2022) THUNDER 2: THeragnostic Utilities for Neoplastic DisEases of the Rectum by MRI guided radiotherapy. BMC Cancer 22:67. https://doi.org/10.1186/s12885-021-09158-9
Tang B, Liu M, Wang B et al (2022) Improving the clinical workflow of a MR-Linac by dosimetric evaluation of synthetic CT. Front Oncol 12:66
Votta C, Cusumano D, Boldrini L et al (2021) Delivery of online adaptive magnetic resonance guided radiotherapy based on isodose boundaries. Phys Imaging Radiat Oncol 18:78–81. https://doi.org/10.1016/j.phro.2021.05.005
Placidi L, Cusumano D, Boldrini L et al (2020) Quantitative analysis of MRI-guided radiotherapy treatment process time for tumor real-time gating efficiency. J Appl Clin Med Phys 21:70–79. https://doi.org/10.1002/acm2.13030
Acknowledgements
The Authors thank the Scientific Committee and Board of the AIRO for the critical revision and final approval of the manuscript (Nr. 7/2023).
Funding
All authors received no specific funding for this work.
Author information
Authors and Affiliations
Contributions
Conception and design: L.B., V.S., A.D’. Data Collection, analysis, and interpretation of data: A.D’., A.P. Analysis Validation: L.B., F.DeF., I.D., R.G., C.G., G.C.I., V.N., V.S. Manuscript Writing: A.D’., A.P., L.B. The Final Manuscript Approval: all authors.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflicts of interests.
Ethical approval and consent to participate
No Ethical approval was needed due to the design of the study.
Consent for publication
No consent to publication was needed due to the design of the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Boldrini, L., D’Aviero, A., De Felice, F. et al. Artificial intelligence applied to image-guided radiation therapy (IGRT): a systematic review by the Young Group of the Italian Association of Radiotherapy and Clinical Oncology (yAIRO). Radiol med 129, 133–151 (2024). https://doi.org/10.1007/s11547-023-01708-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-023-01708-4