Abstract
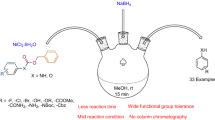
A green and eco-friendly route for the almost quantitative BOC protection of a large variety of aliphatic and aromatic amines, amino acids, and amino alcohols is reported in catalyst and solvent-free media under mild reaction conditions. The products were confirmed by 1H, 13C NMR, IR spectroscopy, and in some cases, elemental analysis. This protocol does not require any water quenches, solvent separations, and purification steps, such as recrystallization and column chromatography.



Similar content being viewed by others
References
P.T. Anastas, T.C. Williamson, Green Chemistry: Frontiers in Chemical Synthesis and Processes (Oxford University Press, Oxford, 1998)
M. Lancaster, Green Chemistry: An Introductory Text (Royal Society of Chemistry, Cambridge, 2002)
J. Clark, D. Macquarrie, Handbook of Green Chemistry and Technology (Blackwell, Abingdon, 2002)
R.A. Sheldon, Industrial Environmental Chemistry (Plenum, New York, 1992)
M. Shailaja, A. Manjula, B.V. Rao, Synth. Commun. 41, 2073 (2011)
Y. Basel, A. Hasners, J. Org. Chem. 65, 6368 (2000)
C. Lutz, V. Lutz, P. Knochel, Tetrahedron 54, 6385 (1998)
T.A. Kelly, D.W. McNeil, Tetrahedron Lett. 35, 9003 (1994)
D.L. Boger, J.A. McKie, J. Org. Chem. 60, 1271 (1995)
J.M. Muchowski, M.C. Venuti, J. Org. Chem. 45, 4798 (1980)
A. Heydari, S.E. Hosseini, Adv. Synth. Catal. 347, 1929 (2005)
R.K. Pandey, S.P. Dagade, R.K. Upadhaya, M.K. Dongare, P. Kumar, ARKIVOC vii, 28 (2002)
S. Sunitha, S. Kanjilal, S.P. Reddy, B.N. Prasad, Tetrahedron Lett. 49, 2527 (2008)
D.J. Upadhyaya, A. Barge, R. Stefania, G. Cravottoa, Tetrahedron Lett. 48, 8318 (2007)
B. Das, K. Venkateswarlu, M. Krishnaiah, H. Holla, Tetrahedron Lett. 47, 7551 (2006)
M.S. Reddy, M. Narender, Y.V.D. Nageswar, K.R. Rao, Synlett 7, 1110 (2006)
S. Khaksar, A. Heydari, M. Tajbakhsh, S.M. Vahdat, Tetrahedron Lett. 49, 3527 (2008)
N. Inahashi, A. Matsumiya, T. Sato, Synlett 2, 294 (2008)
A. Heydari, R.K. Shiroodi, H. Hamadi, M. Esfandyari, M. Pourayoubi, Tetrahedron Lett. 48, 5865 (2007)
S.V. Chankeshwara, A.S. Chakraborti, J. Mol. Catal. A Chem. 253, 198 (2006)
N.G. Khaligh, RSC Adv. 2, 12364 (2012)
S.V. Chankeshwara, A.K. Chakraborti, Org. Lett. 8, 3259 (2006)
M. Raju, S. Mäeorg, O. Tšubrik, U. Mäeorg, ARKIVOC vi, 291 (2009)
M.M. Mojtahedi, N. Niknejad, H. Veisi, Lett. Org. Chem. 10, 121 (2013)
X. Jia, Q. Huang, J. Li, S. Li, Q. Yang, Synlett 5, 0806 (2007)
M.A. Zolfigol, V. Khakyzadeh, A.R.M. Zare, G. Chehardoli, F.D. Panah, A. Zare, O. Khaledian, Sci. Iran. Tran. C Chem. Chem. Eng. 18, 1584 (2012)
Z. Cheraiet, S. Ouarna, S. Hessainia, M. Berredjem, N.E. Aouf, ISRN Org. Chem. 2012, 404235 (2012). doi:10.5402/2012/404235
R. Varala, S. Nuvula, S.R. Adapa, J. Org. Chem. 71, 8283 (2006)
G. Bartoli, M. Bosco, M. Locatelli, E. Marcantoni, M. Massaccesi, P. Melchiorre, L. Sambri, Synlett 10, 1794 (2004)
F. Shirini, M.A. Zolfigol, M. Abedini, J. Iran. Chem. Soc. 7, 603 (2010)
F. Shirini, N.G. Khaligh, O.G. Jolodar, J. Iran. Chem. Soc. 10, 181 (2013)
S. Karimian, H. Tajik, Chin. Chem. Lett. 25, 218 (2014)
G.P. Kumar, D. Rambabu, M.V. Basaveswara Rao, M. Pal, J. Chem. 2013, 916960 (2013). doi:10.1155/2013/916960
F. Shirini, N.G. Khaligh, J. Mol. Liq. 177, 386 (2013)
F. Shirini, O.G. Jolodar, M. Seddighi, H.T. Borujeni, RSC Adv. 5, 19790 (2015)
Acknowledgments
Dr. B. Viswanadham thanks the University of KwaZulu-Natal for the award of an AES Postdoctoral Fellow.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Viswanadham, B., Mahomed, A.S., Friedrich, H.B. et al. Efficient and expeditious chemoselective BOC protection of amines in catalyst and solvent-free media. Res Chem Intermed 43, 1355–1363 (2017). https://doi.org/10.1007/s11164-016-2702-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-016-2702-9