Abstract
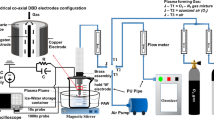
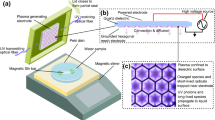
Cold atmospheric plasma (CAP) has gaining potential, and very effective to curb or deactivate the various microorganisms such as bacteria and virus. Lately, the major outbreak SARS-CoV-2 infection has affected humanity largely with added complexity of its ability to mutate to variants such as Omicron. We have earlier shown the effectiveness of CAP on SARS-CoV-2 spike protein and, in this study, we have evaluated the effectiveness of CAP to deactivate Omicron. We studied the ability of the binding of Angiotensin converting Enzyme Protein (ACE2) protein to the plasma treated spike S1-S2 protein and spike Receptor binding domain (RBD) using Cold atmospheric plasma direct treatment as well as Plasma activated water (PAW). Results have shown the binding efficiency of Omicron spike protein to ACE2 decrease with increase in treatment time with both direct treatment and PAW as evidenced using spectroscopic techniques. The reactive species (RONS) play a major role in the efficient deactivation of the binding of ACE2 to the Omicron spike protein. Correspondingly, the comparison between the efficiency between direct treatment and PAW has also been discussed.





Similar content being viewed by others
Data Availability
Not applicable.
References
Adedoyin OB, Soykan E (2020) Covid-19 pandemic and online learning: the challenges and opportunities. Interact Learn Environ 3:1–3
World Health Organization (2023) Geneva, Swizerland. https://www.who.int/health-topics/coronavirus#tab=tab_1. Accessed 21 June 2023
Burki T (2021) Understanding variants of SARS-CoV-2. The Lancet 397(10273):462
Li M, Lou F, Fan H (2021) SARS-CoV-2 variants of concern Delta: a great challenge to prevention and control of COVID-19. Signal Transduct Target Therapy 6:1–3
Li M, Lou F, Fan H (2022) SARS-CoV-2 variant Omicron: currently the most complete escapee from neutralization by antibodies and vaccines. Signal Transduct Target Therapy 7:1–3
World Health Organization (2020) Infection prevention and control during health care when novel coronavirus ( nCoV) infection is suspected: interim guidance. World Health Organization
Loizou C, Kniazeva V, Apostolou T, Kornev A, Kostevitch S, Roslyakov E, Constantinou C, Hadjihannas L (2022) Effect of cold atmospheric plasma on SARS-CoV-2 inactivation: a pilot study in the hospital environment. COVID 2:1396–1404
Chen Z, Garcia G Jr, Arumugaswami V, Wirz RE (2020) Cold atmospheric plasma for SARS-CoV-2 inactivation. Phys Fluids 32:111702
Ganesh Subramanian PS, Harsha R, Manju DK, Hemanth M, Lakshminarayana R, Anand MS, Dasappa S (2019) Characterization of plasma activated water for medical applications. Adv Mater Lett 10:919–923
Bisag A, Isabelli P, Laurita R, Bucci C, Capelli F, Dirani G, Gherardi M, Laghi G, Paglianti A, Sambri V, Colombo V (2020) Cold atmospheric plasma inactivation of aerosolized microdroplets containing bacteria and purified SARS-CoV‐2 RNA to contrast airborne indoor transmission. Plasma Processes Polym 17:2000154
Bisag A, Isabelli P, Laghi G, Laurita R, Dirani G, Taddei F, Bucci C, Capelli F, Gherardi M, Paglianti A, Sambri V (2022) Cold atmospheric plasma decontamination of SARS-CoV‐2 bioaerosols. Plasma Processes Polym 19:e2100133
Jin T, Xu Y, Dai C, Zhou X, Xu Q, Wu Z (2021) Cold atmospheric plasma: A non-negligible strategy for viral RNA inactivation to prevent SARS-CoV-2 environmental transmission AIP advances 11
Kaushik N, Mitra S, Baek EJ, Nguyen LN, Bhartiya P, Kim JH, Choi EH, Kaushik NK (2023) The inactivation and destruction of viruses by reactive oxygen species generated through physical and cold atmospheric plasma techniques: current status and perspectives. J Adv Res 43:59–71
Khanikar RR, Kalita M, Kalita P, Kashyap B, Das S, Khan MR, Bailung H, Sankaranarayanan K (2022) Cold atmospheric pressure plasma for attenuation of SARS-CoV-2 spike protein binding to ACE2 protein and the RNA deactivation. RSC Adv 12:9466–9472
Filipić A, Gutierrez-Aguirre I, Primc G, Mozetič M, Dobnik D (2020) Cold plasma, a new hope in the field of virus inactivation. Trends Biotechnol 38:1278–1291
Qin H, Qiu H, He ST, Hong B, Liu K, Lou F, Li M, Hu P, Kong X, Song Y, Liu Y (2022) Efficient disinfection of SARS-CoV-2-like coronavirus, pseudotyped SARS-CoV-2 and other coronaviruses using cold plasma induces spike protein damage. J Hazard Mater 430:128414
Chen Z, Garcia G, Arumugaswami V, Wirz RE (2020) Cold atmospheric plasma for SARS-CoV-2 inactivation. Phys Fluids 1:32
Cortázar OD, Megía-Macías A, Moreno S, Brun A, Gómez-Casado E (2022) Vulnerability of SARS-CoV-2 and PR8 H1N1 virus to cold atmospheric plasma activated media. Sci Rep 12:263
Han I, Mumtaz S, Choi EH (2022) Nonthermal biocompatible plasma inactivation of coronavirus SARS-CoV-2: prospects for future antiviral applications. Viruses 14:2685
Adamovich I, Agarwal S, Ahedo E, Alves LL, Baalrud S, Babaeva N, Bogaerts A, Bourdon A, Bruggeman PJ, Canal C, Choi EH (2022) The 2022 plasma roadmap: low temperature plasma science and technology. J Phys D 55:373001
Tang X, Staack D (2019) Bioinspired mechanical device generates plasma in water via cavitation. Sci Adv 5:eaau7765
Guo L, Yao Z, Yang L, Zhang H, Qi Y, Gou L, Xi W, Liu D, Zhang L, Cheng Y, Wang X (2021) Plasma-activated water: an alternative disinfectant for S protein inactivation to prevent SARS-CoV-2 infection. Chem Eng J 421:127742
Fridman G, Friedman G, Gutsol A, Shekhter AB, Vasilets VN, Fridman A (2008) Applied plasma medicine. Plasma Processes Polym 5:503–533
Von Woedtke T, Schmidt A, Bekeschus S, Wende K, Weltmann KD (2019) Plasma medicine: a field of applied redox biology. In Vivo 33:1011–1026
Nehra V, Kumar A, Dwivedi HK (2008) Atmospheric non-thermal plasma sources. Int J Eng 2:53–68
Feizollahi E, Misra NN, Roopesh MS (2021) Factors influencing the antimicrobial efficacy of dielectric barrier discharge (DBD) atmospheric cold plasma (ACP) in food processing applications. Crit Rev Food Sci Nutr 61:666–689
Khanikar RR, Boruah PJ, Bailung H (2020) Development and optical characterization of an atmospheric pressure non-thermal plasma jet for superhydrophobic surface fabrication. Plasma Res Express 2:045002
Bharati AJ, Khanikar RR, Bailung H, Sankaranarayanan K (2023) Investigating the effect of atmospheric plasma on protein fibrinogen: spectroscopic and biophysical analysis. Plasma Processes Polym e2300127
Lan J, Ge J, Yu J, Shan S, Zhou H, Fan S, Zhang Q, Shi X, Wang Q, Zhang L, Wang X (2020) Structure of the SARS-CoV-2 spike receptor-binding domain bound to the ACE2 receptor. Nature 581:215–220
Journal of Experimental Botany (2006) 57(8):1711–1718. https://doi.org/10.1093/jxb/erj180
Hydrogen peroxide induce modifications of human extracellular superoxide dismutase that results in enzyme inhibition (2013) Redox Biology 1(1):24–31
Imaging and Profiling of proteins under oxidative conditions in cells and tissues by hydrogen-peroxide-responsive labeling (2020) J Am Chem Soc 142(37):15711–15721
Bruggeman P, Leys C (2009) Non-thermal plasmas in and in contact with liquids. J Phys D 42:053001
Yang X, Zhang C, Li Q, Cheng JH (2023) Physicochemical properties of plasma-activated water and its control effects on the quality of strawberries. Molecules 28:2677
Hoeben WF, Van Ooij PP, Schram DC, Huiskamp T, Pemen AJ, Lukeš PJ (2019) On the possibilities of straightforward characterization of plasma activated water. Plasma Chem Plasma Process 39:597–626
Li Z, Zhang X, Qi M, Zhao X, Qu Z, Wang X, Li W, Xu D (2023) Assessment of microbial species inactivation and purification of sewage by a gas–liquid diaphragm discharge plasma. J Appl Phys 134(9)
Acknowledgements
Author KS thanks DST-IASST, Guwahati for the In-house project grant, SERB, Govt. of India for the SRG grant (SRG/2020/001894 dt. 11.11.2020) and ICMR Project grant (File No. 17 × (3)/Ad-hoc/19/2022-ITR dt. 23.12.2022). Author Reema thanks UGC, Govt. of India and DB thanks IASST DST, Govt. of India for the support for the Junior Research Fellowship. Reema and DB also thanks AcSIR for Ph.D registration.
Funding
In-house project grant, SERB, Govt. of India for the SRG grant (SRG/2020/001894 dt. 11.11.2020) and ICMR Project grant (File No. 17 × (3)/Ad-hoc/19/2022-ITR dt. 23.12.2022).
Author information
Authors and Affiliations
Contributions
Reema and DB performed the experiments and HB, KS designed the experiments and all the authors corrected the manuscript.
Corresponding author
Ethics declarations
Ethical Approval
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Reema, Basumatary, D., Bailung, H. et al. Comparative Analysis of Direct Cold Atmospheric Plasma Treatment vs. Plasma Activated Water for the Deactivation of Omicron Variant of SARS-CoV-2. Plasma Chem Plasma Process 44, 1019–1030 (2024). https://doi.org/10.1007/s11090-024-10449-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-024-10449-9