Abstract
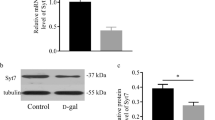
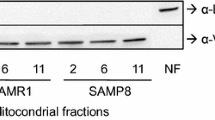
Senescence-accelerated mouse prone 8 (SAMP8) is considered as a useful animal model for age-related learning and memory impairments. Hippocampus, a critical brain region associated with cognitive decline during normal aging and various neurodegenerative diseases, appeared a series of abnormalities in SAMP8. To investigate the molecular mechanisms underlying age-related cognitive disorders, we used 2-DE coupled with MALDI TOF/TOF MS to analyze the differential protein expression of the hippocampus of SAMP8 at 6-month-old compared with the age-matched SAM/resistant 1 (SAMR1) which shows normal aging process. Two proteins were found to be markedly changed in SAMP8 as compared to SAMR1: ubiquitin carboxyl-terminal hydrolase L3 (Uchl3), implicating in cytosolic proteolysis of oxidatively damaged proteins, was down-regulated while mitofilin, a vital protein for normal mitochondria function, exhibited four isoforms with a consistent basic shift of isoelectric point among the soluble hippocampal proteins in SAMP8 compared with SAMR1. The alterations were confirmed by Western blotting analysis. The analysis of their expression changes may shed light on the mechanisms of learning and memory deficits and mitochondrial dysfunction as observed in SAMP8.




Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- CHAPS:
-
3-(3-(Cholamidopropyl) dimethylammonio) propanesulfonate
- DTT:
-
Dithiothreitol
- MALDI TOF/TOF:
-
Matrix-assisted laser desorption ionization time-of-flight/time-of-flight
- MS:
-
Mass spectrometry
- ROS:
-
Reactive oxygen species
- SOD1:
-
Cu, Zn-superoxide dismutase
- SOD2:
-
Manganese superoxide dismutase
- SAMP8:
-
Senescence-accelerated mouse prone 8
- Uchl3:
-
Ubiquitin carboxyl-terminal hydrolase L3
- PMF:
-
Peptide mapping fingerprinting
- 2-DE:
-
Two dimensional electrophoresis
References
Takeda T, Hosokawa M, Takeshita S et al (1981) A new murine model of accelerated senescence. Mech Ageing Dev 17(2):183–194
Flood JF, Morley JE (1998) Learning and memory in the SAMP8 mouse. Neurosci Biobehav Rev 22(1):1–20
Sureda FX, Gutierrez-Cuesta J, Romeu M et al (2006) Changes in oxidative stress parameters and neurodegeneration markers in the brain of the senescence-accelerated mice SAMP-8. Exp Gerontol 41(4):360–367
Tanaka J, Okuma Y, Tomobe K et al (2005) The age-related degeneration of oligodendrocytes in the hippocampus of the senescence-accelerated mouse (SAM) P8: a quantitative immunohistochemical study. Biol Pharm Bull 28(4):615–618
Wu Y, Zhang AQ, Yew DT (2005) Age related changes of various markers of astrocytes in senescence-accelerated mice hippocampus. Neurochem Int 46(7):565–574
Wei X, Zhang Y, Zhou J (1999) Alzheimer’s disease-related gene expression in the brain of senescence accelerated mouse. Neurosci Lett 268(3):139–142
Morley JE, Kumar VB, Bernardo AE et al (2000) Beta-amyloid precursor polypeptide in SAMP8 mice affects learning and memory. Peptides 21(12):1761–1767
Cheng XR, Zhou WX, Zhang YX et al (2007) Differential gene expression profiles in the hippocampus of senescence-accelerated mouse. Neurobiol Aging 28(4):497–506
Butterfield DA, Poon HF (2005) The senescence-accelerated prone mouse (SAMP8): a model of age-related cognitive decline with relevance to alterations of the gene expression and protein abnormalities in Alzheimer’s disease. Exp Gerontol 40(10):774–783
Tomobe K, Okuma Y, Nomura Y (2007) Impairment of CREB phosphorylation in the hippocampal CA1 region of the senescence-accelerated mouse (SAM) P8. Brain Res 1141:214–217
Okuma Y, Nomura Y (1998) Senescence-accelerated mouse (SAM) as an animal model of senile dementia: pharmacological, neurochemical and molecular biological approach. Jpn J Pharmacol 78(4):399–404
Kurokawa T, Asada S, Nishitani S et al (2001) Age-related changes in manganese superoxide dismutase activity in the cerebral cortex of senescence-accelerated prone and resistant mouse. Neurosci Lett 298(2):135–138
Fujibayashi Y, Yamamoto S, Waki A et al (1998) Increased mitochondrial DNA deletion in the brain of SAMP8, a mouse model for spontaneous oxidative stress brain. Neurosci Lett 254(2):109–112
Xu J, Shi C, Li Q et al (2007) Mitochondrial dysfunction in platelets and hippocampi of senescence-accelerated mice. J Bioenerg Biomembr 39(2):195–202
Alvarez-Garcia O, Vega-Naredo I, Sierra V et al (2006) Elevated oxidative stress in the brain of senescence-accelerated mice at 5 months of age. Biogerontology 7(1):43–52
Colas D, Gharib A, Bezin L et al (2006) Regional age-related changes in neuronal nitric oxide synthase (nNOS), messenger RNA levels and activity in SAMP8 brain. BMC Neurosci 7:81
Finkel T, Holbrook NJ (2000) Oxidants, oxidative stress and the biology of ageing. Nature 408(6809):239–247
Nabeshi H, Oikawa S, Inoue S et al (2006) Proteomic analysis for protein carbonyl as an indicator of oxidative damage in senescence-accelerated mice. Free Radic Res 40(11):1173–1181
Poon HF, Castegna A, Farr SA et al (2004) Quantitative proteomics analysis of specific protein expression and oxidative modification in aged senescence-accelerated-prone 8 mice brain. Neuroscience 126(4):915–926
Hegde AN, Inokuchi K, Pei W et al (1997) Ubiquitin C-terminal hydrolase is an immediate-early gene essential for long-term facilitation in Aplysia. Cell 89(1):115–126
Wood MA, Kaplan MP, Brensinger CM et al (2005) Ubiquitin C-terminal hydrolase L3 (Uchl3) is involved in working memory. Hippocampus 15(5):610–621
Barrachina M, Castano E, Dalfo E et al (2006) Reduced ubiquitin C-terminal hydrolase-1 expression levels in dementia with Lewy bodies. Neurobiol Dis 22(2):265–273
Armbrecht HJ, Boltz MA, Kumar VB et al (1999) Effect of age on calcium-dependent proteins in hippocampus of senescence-accelerated mice. Brain Res 842(2):287–293
Yang W, Liu P, Liu Y et al (2006) Proteomic analysis of rat pheochromocytoma PC12 cells. Proteomics 6(10):2982–2990
Candiano G, Bruschi M, Musante L et al (2004) Blue silver: a very sensitive colloidal Coomassie G-250 staining for proteome analysis. Electrophoresis 25(9):1327–1333
Larsen CN, Price JS, Wilkinson KD (1996) Substrate binding and catalysis by ubiquitin C-terminal hydrolases: identification of two active site residues. Biochemistry 35(21):6735–6744
Larsen CN, Krantz BA, Wilkinson KD (1998) Substrate specificity of deubiquitinating enzymes: ubiquitin C-terminal hydrolases. Biochemistry 37(10):3358–3368
Hershko A, Ciechanover A, Varshavsky A (2000) Basic Medical Research Award. The ubiquitin system. Nat Med 6(10):1073–1081
DiAntonio A, Haghighi AP, Portman SL et al (2001) Ubiquitination-dependent mechanisms regulate synaptic growth and function. Nature 412(6845):449–452
Gieffers C, Korioth F, Heimann P et al (1997) Mitofilin is a transmembrane protein of the inner mitochondrial membrane expressed as two isoforms. Exp Cell Res 232(2):395–399
Myung JK, Gulesserian T, Fountoulakis M et al (2003) Deranged hypothetical proteins Rik protein, Nit protein 2 and mitochondrial inner membrane protein, Mitofilin, in fetal Down syndrome brain. Cell Mol Biol (Noisy-le-grand) 49(5):739–746
Wishart TM, Paterson JM, Short DM et al (2007) Differential proteomics analysis of synaptic proteins identifies potential cellular targets and protein mediators of synaptic neuroprotection conferred by the slow Wallerian degeneration (Wlds) gene. Mol Cell Proteomics 6(8):1318–1330
Johnston SC, Larsen CN, Cook WJ et al (1997) Crystal structure of a deubiquitinating enzyme (human UCH-L3) at 1.8 A resolution. Embo J 16(13):3787–3796
Nishikawa T, Takahashi JA, Fujibayashi Y et al (1998) An early stage mechanism of the age-associated mitochondrial dysfunction in the brain of SAMP8 mice; an age-associated neurodegeneration animal model. Neurosci Lett 254(2):69–72
Omori A, Ichinose S, Kitajima S et al (2002) Gerbils of a seizure-sensitive strain have a mitochondrial inner membrane protein with different isoelectric points from those of a seizure-resistant strain. Electrophoresis 23(24):4167–4174
Lai Y, Chen Y, Watkins SC et al (2007) Identification of poly-ADP-ribosylated mitochondrial proteins after traumatic brain injury. J Neurochem
Acknowledgements
This work was supported by grants from National Basic Research Program of China (No. 2006CB910103).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Q., Liu, Y., Zou, X. et al. The Hippocampal Proteomic Analysis of Senescence-Accelerated Mouse: Implications of Uchl3 and Mitofilin in Cognitive Disorder and Mitochondria Dysfunction in SAMP8. Neurochem Res 33, 1776–1782 (2008). https://doi.org/10.1007/s11064-008-9628-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-008-9628-6