Abstract
Background
Endometriosis is a complex gynaecological disorder that contributes to infertility, dysmenorrhea, dyspareunia, and other chronic issues. It is a multifactorial disease involving genetic, hormonal, immunological and environmental components. Endometriosis’s pathogenesis remains unclear.
Aim of the study
was to analyse the polymorphisms in Interleukin 4, Interleukin 18, FCRL3 and sPLA2IIa genes to identify any significant association with the risk of endometriosis.
Material and methods
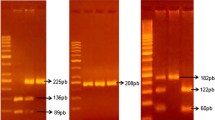
This study evaluated the polymorphism of -590 C/T in interleukin- 4(IL-4) gene, C607A in Interleukin − 18(IL-18) gene, -169T > C in FCRL3 gene and 763 C > G in sPLA2IIa gene in women with endometriosis. The case-control study included 150 women with endometriosis and 150 apparently healthy women as control subjects. DNA was extracted from peripheral blood leukocytes and endometriotic tissue of cases and blood samples for controls and further analysed by PCR amplification and then sequencing was carried out to find the allele and genotypes of the subjects and then to analyse the relationship between the gene polymorphisms and endometriosis. To evaluate the association of the different genotypes, 95% confidence intervals (CI) were calculated.
Results
Interleukin − 18 and FCRL3 gene polymorphisms of endometriotic tissue and blood samples of endometriosis (cases) showed significantly associated (OR = 4.88 [95% CI = 2.31–10.30], P > 0.0001) and (OR = 4.00 [95% CI = 2.2–7.33], P > 0.0001) when compared with normal blood samples. However, there was no significant difference in Interleukin − 4 and sPLA2IIa gene polymorphisms between control women and patients with endometriosis.
Conclusions
The present study suggests that the IL-18 and FCRL3 gene polymorphisms are associated with a higher risk for endometriosis, which delivers valuable knowledge of endometriosis’s pathogenesis. However, a larger sample size of patients from various ethnic backgrounds is necessary to evaluate whether these alleles have a direct effect on disease susceptibility.





Similar content being viewed by others
References
Barbosa CP, Souza ÂM, Bianco B, Christofolini D, Bach FA, Lima GR (2009) Frequency of endometriotic lesions in peritoneum samples from asymptomatic fertile women and correlation with CA125 values. Sao Paulo Med J 127:342–345. https://doi.org/10.1590/S1516-31802009000600004
Barbosa CP, de Souza ÂMB, Bianco B, Christofolini DM, Mafra FA, de Lima GR (2010) OC-125 immunostaining in endometriotic lesion samples. Arch Gynecol Obstet 281(1):43–47. https://doi.org/10.1007/s00404-009-1055-7
Sampson JA (1918) The escape of foreign material from the uterine cavity into the uterine veins. William Wood & Company
Levander G (1941) Über die Pathogenese bei Endometriose. Archiv für Klinische Chirurgie 202:497–515
Vinatier D, Dufour P, Oosterlynck D (1996) Immunological aspects of endometriosis. Hum Reprod Update 2(5):371–284. https://doi.org/10.1093/humupd/2.5.371
Nothnick WB (2001) Treating endometriosis as an autoimmune disease. Fertil Steril 76(2):223–231. https://doi.org/10.1016/S0015-0282(01)01878-7
Matarese G, De Placido G, Nikas Y, Alviggi C (2003) Pathogenesis of endometriosis: natural immunity dysfunction or autoimmune disease? Trends Mol Med 9(5):223–228. https://doi.org/10.1016/S1471-4914(03)00051-0
Berbic M, Hey-Cunningham AJ, Ng C, Tokushige N, Ganewatta S, Markham R, …, Fraser IS (2010) The role of Foxp3 + regulatory T-cells in endometriosis: a potential controlling mechanism for a complex, chronic immunological condition. Hum Reprod 25(4):900–907. https://doi.org/10.1093/humrep/deq020
Harada T, Iwabe T, Terakawa N (2001) Role of cytokines in endometriosis. Fertil Steril 76(1):1–10. https://doi.org/10.1016/S0015-0282(01)01816-7
Wu MY, Ho HN (2003) The role of cytokines in endometriosis. Am J Reprod Immunol 49(5):285–296. https://doi.org/10.1034/j.1600-0897.2003.01207.x
Chun S, Kim H, Ku SY, Suh CS, Kim SH, Kim JG (2012) The Association between endometriosis and polymorphisms in the Interleukin-1 family genes in K orean women. Am J Reprod Immunol 68(2):154–163. https://doi.org/10.1034/j.1600-0897.2003.01207.x
Gallegos-Arreola MP, Figuera-Villanueva LE, Puebla-Pérez AM, Montoya-Fuentes H, Suarez-Rincon AE, Zúñiga-González GM (2012) Association of TP53 gene codon 72 polymorphism with endometriosis in mexican women. Genet Mol Res 11(2):1401–1408. https://doi.org/10.4238/2012.May.15.10
Luisi S, Galleri L, Marini F, Ambrosini G, Brandi ML, Petraglia F (2006) Estrogen receptor gene polymorphisms are associated with recurrence of endometriosis. Fertil Steril 85(3):764–766. https://doi.org/10.1016/j.fertnstert.2005.08.038
Yang Y, Li D, He C, Peng L, Xing S, Bai M, …, Jin T (2021) Fc receptor-like 1, 3, and 6 variants are associated with rheumatoid arthritis risk in the chinese Han population. Genes and Environment 43(1):1–8. https://doi.org/10.1186/s41021-021-00213-2
Podgaec S, Abrao MS, Dias Jr JA, Rizzo LV, De Oliveira RM, Baracat EC (2007) Endometriosis: an inflammatory disease with a Th2 immune response component. Hum Reprod 22(5):1373–1379. https://doi.org/10.1093/humrep/del516
Fan W, Li S, Chen Q, Huang Z, Ma Q, Xiao Z (2013) Association between interleukin-10 promoter polymorphisms and endometriosis: a meta-analysis. Gene 515(1):49–55. https://doi.org/10.1016/j.gene.2012.11.037
Ghayur T, Banerjee S, Hugunin M, Butler D, Herzog L, Carter A, Alien H (1997) Caspase-1 processes IFN-γ-inducing factor and regulates LPS-induced IFN-γ production. Nature 386(6625):619–623. https://doi.org/10.1038/386619a0
Gu Y, Kuida K, Tsutsui H, Ku G, Hsiao K, Fleming MA, …, Su MSS (1997) Activation of interferon-γ inducing factor mediated by interleukin-1β converting enzyme. Science 275(5297):206–209. https://doi.org/10.1126/science.275.5297.206
Giedraitis V, He B, Huang WX, Hillert J (2001) Cloning and mutation analysis of the human IL-18 promoter: a possible role of polymorphisms in expression regulation. J Neuroimmunol 112(1–2):146–152. https://doi.org/10.1016/s0165-5728(00)00407-0
Yoshino O, Osuga Y, Koga K, Tsutsumi O, Yano T, Fujii T, Taketani Y (2001) Evidence for the expression of interleukin (IL)-18, IL-18 receptor and IL-18 binding protein in the human endometrium. Mol Hum Reprod 7(7):649–654. https://doi.org/10.1093/molehr/7.7.649
Samimi M, Pourhanifeh MH, Mehdizadehkashi A, Eftekhar T, Asemi Z (2019) The role of inflammation, oxidative stress, angiogenesis, and apoptosis in the pathophysiology of endometriosis: Basic science and new insights based on gene expression. J Cell Physiol 234(11):19384–19392. https://doi.org/10.1002/jcp.28666
Chistiakov DA, Chistiakov AP (2007) Is FCRL3 a new general autoimmunity gene? Hum Immunol 68(5):375–383. https://doi.org/10.1016/j.humimm.2007.01.013
Swainson LA, Mold JE, Bajpai UD, McCune JM (2010) Expression of the autoimmune susceptibility gene FcRL3 on human regulatory T cells is associated with dysfunction and high levels of programmed cell death-1. J Immunol 184(7):3639–3647. https://doi.org/10.4049/jimmunol.0903943
Nagata S, Ise T, Pastan I Fc receptor-like 3 protein expressed on IL-2 nonresponsive subset of human regulatory T cells.The Journal of Immunology. 2009 Jun15;182(12):7518–26. https://doi.org/10.4049/jimmunol.0802230
Kochi Y, Yamada R, Suzuki A, Harley JB, Shirasawa S, Sawada T, Yamamoto K (2005) A functional variant in FCRL3, encoding fc receptor-like 3, is associated with rheumatoid arthritis and several autoimmunities. Nat Genet 37(5):478–485. https://doi.org/10.1038/ng1540
Sanchez E, Callejas JL, Sabio JM, de Haro M, Camps M, de Ramon E, Gonzalez-Escribano MF (2006) Polymorphisms of the FCRL3 gene in a spanish population of systemic lupus erythematosus patients. Rheumatology (Oxford) 45(8):1044–1046. https://doi.org/10.1093/rheumatology/kel160
Inoue N, Watanabe M, Yamada H, Takemura K, Hayashi F, Yamakawa N, …, Iwatani Y (2012) Associations between autoimmune thyroid disease prognosis and functional polymorphisms of susceptibility genes, CTLA4, PTPN22, CD40, FCRL3, and ZFAT, previously revealed in genome-wide association studies. J Clin Immunol 32(6):1243–1252. https://doi.org/10.1007/s10875-012-9721-0
Siminovitch KA (2004) PTPN22 and autoimmune disease. Nat Genet 36(12):1248–1249. https://doi.org/10.1038/ng1204-1248
Szczepańska M, Wirstlein P, Hołysz H, Skrzypczak J, Jagodziński PP (2013) The FCRL3 – 169T > C polymorphism and the risk of endometriosis-related infertility in a polish population. Arch Gynecol Obstet 288(4):799–804. https://doi.org/10.1007/s00404-013-2829-5
Keskinen LA, Annous BA (2011) Efficacy of adding detergents to sanitizer solutions for inactivation of Escherichia coli O157: H7 on romaine lettuce. Int J Food Microbiol 147(3):157–161. https://doi.org/10.1016/j.ijfoodmicro.2011.04.002
Bianco B, Teles JS, Lerner TG, Vilarino FL, Christofolini DM, Barbosa CP (2011) Association of FCRL3 – 169T/C polymorphism with endometriosis and identification of a protective haplotype against the development of the disease in brazilian population. Hum Immunol 72(9):774–778. https://doi.org/10.1016/j.humimm.2011.05.005
Linton MF, Fazio S (2001) Class A scavenger receptors, macrophages, and atherosclerosis. Curr Opin Lipidol 12(5):489–495
De Villiers WJ, Smart EJ (1999) Macrophage scavenger receptors and foam cell formation. J Leukoc Biol 66(5):740–746. https://doi.org/10.1002/jlb.66.5.740
Melo AS, Rosa-e-Silva JC, de Sá Rosa ACJ, Poli-Neto OB, Ferriani RA, Vieira CS (2010) Unfavorable lipid profile in women with endometriosis. Fertil Steril 93(7):2433–2436. https://doi.org/10.1016/j.fertnstert.2009.08.043
Six DA, Dennis EA (2000) The expanding superfamily of phospholipase A2 enzymes: classification and characterization. Biochim et Biophys Acta (BBA)-Molecular Cell Biology Lipids 1488(1–2):1–19. https://doi.org/10.1016/S1388-1981(00)00105-0
Vassilopoulou L, Matalliotakis M, Zervou MI, Matalliotaki C, Krithinakis K, Matalliotakis I, …, Goulielmos GN (2019) Defining the genetic profile of endometriosis. Experimental and therapeutic medicine 17(5):3267–3281. https://doi.org/10.3892/etm.2019.7346
Rosenson RS, Gelb MH (2009) Secretory phospholipase A2: a multifaceted family of proatherogenic enzymes. Curr Cardiol Rep 11(6):445–451. https://doi.org/10.1007/s11886-009-0064-2
Leonarduzzi G, Gamba P, Gargiulo S, Biasi F, Poli G (2012) Inflammation-related gene expression by lipid oxidation-derived products in the progression of atherosclerosis. Free Radic Biol Med 52(1):19–34. https://doi.org/10.1016/j.freeradbiomed.2011.09.031
Lambeau G, Gelb MH (2008) Biochemistry and physiology of mammalian secreted phospholipases A2. Annu Rev Biochem 77(1):495–520. https://doi.org/10.1146/annurev.biochem.76.062405.154007
Bostrom MA, Boyanovsky BB, Jordan CT, Wadsworth MP, Taatjes DJ, de Beer FC, Webb NR (2007) Group V secretory phospholipase A2 promotes atherosclerosis: evidence from genetically altered mice. Arterioscler Thromb Vasc Biol 27(3):600–606. https://doi.org/10.1161/01.ATV.0000257133.60884.44
Van Langendonckt A, Casanas-Roux F, Dolmans MM, Donnez J (2002) Potential involvement of hemoglobin and heme in the pathogenesis of peritoneal endometriosis. Fertil Steril 77(3):561–570. https://doi.org/10.1016/s0015-0282(01)03211-3
Hahn LW, Ritchie MD, Moore JH (2003) Multifactor dimensionality reduction software for detecting gene–gene and gene–environment interactions. Bioinformatics 19(3):376–382. https://doi.org/10.1093/bioinformatics/btf869
Cattaert T, Calle ML, Dudek SM, John JMM, van Lishout F, Urrea V, …, van Steen K (2011) A detailed view on model-based Multifactor Dimensionality reduction for detecting gene-gene interactions in case-control data in the absence and presence of noise. Ann Hum Genet 75(1):78. https://doi.org/10.1111/j.1469-1809.2010.00604.x
Ritchie MD, Hahn LW, Moore JH (2003) Power of multifactor dimensionality reduction for detecting gene-gene interactions in the presence of genotyping error, missing data, phenocopy, and genetic heterogeneity. Genetic Epidemiology: The Official Publication of the International Genetic Epidemiology Society 24(2):150–157. https://doi.org/10.1002/gepi.10218
Trovó de Marqui AB (2012) Genetic polymorphisms and endometriosis: contribution of genes that regulate vascular function and tissue remodeling. Revista da Associação Médica Brasileira 58:620–632. https://doi.org/10.1590/S0104-42302012000500022
Andrei M, Costin N, Diculescu D, Ciortea R, Gaia O, Bucuri C, Mihu D (2014) Gene polymorphism involment in endometriosis. Gineco.eu[10]158–161
Falconer H, D’Hooghe T, Fried G (2007) Endometriosis and genetic polymorphisms. Obstet Gynecol Surv 62(9):616–628
Oku H, Tsuji Y, Kashiwamura SI, Adachi S, Kubota A, Okamura H, Koyama K (2004) Role of IL-18 in pathogenesis of endometriosis. Hum Reprod 19(3):709–714. https://doi.org/10.1093/humrep/deh108
Hsu CC, Yang BC, Wu MH, Huang KE (1997) Enhanced interleukin-4 expression in patients with endometriosis. Fertil Steril 67(6):1059–1064. https://doi.org/10.1016/S0015-0282(97)81439-2
Kitawaki J, Koshiba H, Kitaoka Y, Teramoto M, Hasegawa G, Nakamura N, Honjo H (2004) Interferon-γ gene dinucleotide (CA) repeat and interleukin-4 promoter region (– 590 C/T) polymorphisms in japanese patients with endometriosis. Hum Reprod 19(8):1765–1769. https://doi.org/10.1093/humrep/deh337
Luo Q, Ning W, Wu Y, Zhu X, Jin F, Sheng J, Huang H (2006) Altered expression of interleukin-18 in the ectopic and eutopic endometrium of women with endometriosis. J Reprod Immunol 72(1–2):108–117. https://doi.org/10.1016/j.jri.2006.03.003
Ning W, Huang H, Jin F (2004), September Expression of IL-18 in endometriosis. In International Congress Series (Vol. 1271, pp. 244–247). Elsevier. https://doi.org/10.1016/j.ics.2004.05.089
Giedraitis V, He B, Huang WX, Hillert J (2001) Cloning and mutation analysis of the human IL-18 promoter: a possible role of polymorphisms in expression regulation. J Neuroimmunol 112(1–2):146–152. https://doi.org/10.1016/S0165-5728(00)00407-0
Akira S (2000) The role of IL-18 in innate immunity. Curr Opin Immunol 12(1):59–63. https://doi.org/10.1016/S0952-7915(99)00051-5
Okamura H, Tsutsui H, Komatsu T, Yutsudo M, Hakura A, Tanimoto T, Kurimoto M (1995) Cloning of a new cytokine that induces IFN-γ production by T cells. Nature 378(6552):88–91. https://doi.org/10.1038/378088a0
Guo SW (2007) Nuclear factor-κB (NF-κB): an unsuspected major culprit in the pathogenesis of endometriosis that is still at large? Gynecol Obstet Invest 63(2):71–97. https://doi.org/10.1159/000096047
González-Ramos R, Van Langendonckt A, Defrère S, Lousse JC, Colette S, Devoto L, Donnez J (2010) Involvement of the nuclear factor-κB pathway in the pathogenesis of endometriosis. Fertil Steril 94(6):1985–1994. https://doi.org/10.1016/j.fertnstert.2010.01.013
Osuga Y, Koga K, Hirota Y, Hirata T, Yoshino O, Taketani Y (2011) Lymphocytes in endometriosis. Am J Reprod Immunol 65(1):1–10. https://doi.org/10.1111/j.1600-0897.2010.00887.x
Polson AG, Zheng B, Elkins K, Chang W, Du C, Dowd P, Ebens A (2006) Expression pattern of the human FcRH/IRTA receptors in normal tissue and in B-chronic lymphocytic leukemia. Int Immunol 18(9):1363–1373. https://doi.org/10.1093/intimm/dxl069
Nagata S, Ise T, Pastan I (2009) Fc receptor-like 3 protein expressed on IL-2 nonresponsive subset of human regulatory T cells. J Immunol 182(12):7518–7526. https://doi.org/10.4049/jimmunol.0802230
Swainson LA, Mold JE, Bajpai UD, McCune JM (2010) Expression of the autoimmune susceptibility gene FcRL3 on human regulatory T cells is associated with dysfunction and high levels of programmed cell death-1. J Immunol (Baltimore Md : 1950) 184(7):3639–3647. https://doi.org/10.4049/jimmunol.0903943
Berbic M, Hey-Cunningham AJ, Ng C, Tokushige N, Ganewatta S, Markham R, Fraser IS (2010) The role of Foxp3 + regulatory T-cells in endometriosis: a potential controlling mechanism for a complex, chronic immunological condition. Hum Reprod 25(4):900–907. https://doi.org/10.1093/humrep/deq020
Berbic M, Fraser IS (2011) Regulatory T cells and other leukocytes in the pathogenesis of endometriosis. J Reprod Immunol 88(2):149–155. https://doi.org/10.1016/j.jri.2010.11.004
Szmitko PE, Wang CH, Weisel RD, Jeffries GA, Anderson TJ, Verma S (2003) Biomarkers of vascular disease linking inflammation to endothelial activation: part II. Circulation 108(17):2041–2048
Gazvani R, Templeton A (2002) Peritoneal environment, cytokines and angiogenesis in the pathophysiology of endometriosis. Reproduction 123(2):217–226. https://doi.org/10.1530/rep.0.1230217
Lebovic DI, Mueller MD, Taylor RN (2001) Immunobiology of endometriosis. Fertil Steril 75(1):1–10. https://doi.org/10.1016/s0015-0282(00)01630-7
Lousse JC, Van Langendonckt A, González-Ramos R, Defrère S, Renkin E, Donnez J (2008) Increased activation of nuclear factor-kappa B (NF-kappaB) in isolated peritoneal macrophages of patients with endometriosis. Fertil Steril 90(1):217–220. https://doi.org/10.1016/j.fertnstert.2007.06.015
Ann M, Paul H (2001) Oxidized LDL and HDL: antagonists in atherothrombosis. FASEB J 15(12):2073–2084. https://doi.org/10.1096/fj.01-0273rev
Kougias P, Chai H, Lin PH, Lumsden AB, Yao Q, Chen C (2005) Lysophosphatidylcholine and secretory phospholipase A2 in vascular disease: mediators of endothelial dysfunction and atherosclerosis. Medical science monitor: international medical journal of experimental and clinical research 12(1):RA5-16. PMID: 16369478
Wootton PT, Drenos F, Cooper JA, Thompson SR, Stephens JW, Hurt-Camejo E, Talmud PJ (2006) Tagging-SNP haplotype analysis of the secretory PLA2IIa gene PLA2G2A shows strong association with serum levels of sPLA2IIa: results from the UDACS study. Hum Mol Genet 15(2):355–361. https://doi.org/10.1093/hmg/ddi453
Flood C, Gustafsson M, Pitas RE, Arnaboldi L, Walzem RL, Borén J (2004) Molecular mechanism for changes in proteoglycan binding on compositional changes of the core and the surface of low-density lipoprotein–containing human apolipoprotein B100. Arterioscler Thromb Vasc Biol 24(3):564–570. https://doi.org/10.1161/01.ATV.0000117174.19078.85
Hurt-Camejo E, Camejo G, Sartipy P (2000) Phospholipase A2 and small, dense low-density lipoprotein. Curr Opin Lipidol 11(5):465–471
Sartipy P, Camejo G, Svensson L, Hurt-Camejo E (1999) Phospholipase A2 modification of low density lipoproteins forms small high density particles with increased affinity for proteoglycans and glycosaminoglycans. J Biol Chem 274(36):25913–25920. https://doi.org/10.1074/jbc.274.36.25913
Polak G, Mazurek D, Rogala E, Nowicka A, Derewianka-Polak M, Kotarski J (2011) Increased oxidized LDL cholesterol levels in peritoneal fluid of women with advanced-stage endometriosis. Ginekologia Polska 82(3)
Hahn LW, Ritchie MD, Moore JH (2003) Multifactor dimensionality reduction software for detecting gene–gene and gene–environment interactions. Bioinformatics 19(3):376–382. https://doi.org/10.1093/bioinformatics/btf869
Funding
This research did not receive any specifc grant from funding agencies.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no confict of interest.
Ethical approval
This study was approved by the ethical committee of the Sri Ramachandra Institute of Higher Education and Research (DU) [Ref No.: IEC-NI/17/APR/59/38].
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Balunathan, N., Rani G, U., Perumal, V. et al. Single nucleotide polymorphisms of Interleukin − 4, Interleukin-18, FCRL3 and sPLA2IIa genes and their association in pathogenesis of endometriosis. Mol Biol Rep 50, 4239–4252 (2023). https://doi.org/10.1007/s11033-023-08316-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-023-08316-5