Abstract
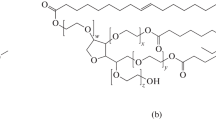
A series of the solid emulsion gels with the oil volume fraction in the range of 0–50% were synthesized through a polycondensation reaction between activated p-nitrophenyl carbonate poly(ethylene glycol) and protein-stabilized oil-in-water emulsions. The resultant structures were investigated in terms of swelling behavior, composition, morphology, mechanical and skin hydration properties. Solid emulsions gels share the properties of both hydrogel and emulsion. Similar to the classical hydrogel, the SEG swells in water up to equilibrium swelling degree, which decreases as the oil volume fraction increases, and comprises immobilized drops of protein-stabilized oil. The impregnation of the oil phase is found to reduce tensile stiffness of the material, but improves material’s extensibility. The mechanical properties of the constructs (Young moduli in the range of 9–15 kPa and the elongation at break of 120–220%) are interpreted according to the “rule of elasticity mixture” that considers the elasticity of the composite material to be a sum of the contributions from individual components, i.e. hydrogel and dispersed oil drops. An idealized model that takes into account the history of the material preparation has been proposed to explain the improved extensibility of the constructs. The results of the mechanical tests, equilibrium swelling, and the skin hydration effect of the solid emulsion gels in vivo are discussed from the perspective of the biomedical applications of the solid emulsion gels, in particular, for the transdermal delivery of hydrophilic and lipophilic drugs.






Similar content being viewed by others
References
M.J. Lawrence, G.D. Rees, Adv. Drug Deliv. Rev. 45, 89–121 (2000). doi:10.1016/S0169-409X(00)00103-4
M. Kreilgaard, Adv. Drug Deliv. Rev. 54, S77–S98 (2002). doi:10.1016/S0169-409X(02)00116-3
A. Spernath, A. Aserin, Adv. Colloid Interface Sci. 128–130, 47–64 (2006). doi:10.1016/j.cis.2006.11.016
M. Graziacascone, Z. Zhu, F. Borselli, L. Lazzeri, J. Mater. Sci.: Mater. Med. 13, 29–32 (2002). doi:10.1023/A:1013674200141
D. Gulsen, A. Chauhan, Int. J. Pharm. 292, 95–117 (2005). doi:10.1016/j.ijpharm.2004.11.033
C. Holtze, K. Landfester, M. Antonietti, Macromol. Mater. Eng. 290, 1025–1028 (2005). doi:10.1002/mame.200500241
H. Chen, D. Mou, D. Du, X. Chang, D. Zhu, J. Liu, H. Xu, X. Yang, Int. J. Pharm. 341, 78–84 (2007). doi:10.1016/j.ijpharm.2007.03.052
K.I. Shingel, M.P. Faure, L. Azoulay, C. Roberge, R.H. Deckelbaum, J. Tissue Eng. Regen. Med. 3, 383–393 (2008). doi:10.1002/term.101
K.S. Anseth, C.N. Bowman, L.B. Peppas, Biomaterials 17, 1647–1657 (1996). doi:10.1016/0142-9612(96)87644-7
Faure MP, Shingel KI (2008) US Patent No. 7,351,787
R. Snyders, K.I. Shingel, O. Zabeida, C. Roberge, M.F. Faure, L. Martinu, J.E. Klemberg-Sapieha, J. Biomed. Mater. Res. 83A, 88–97 (2007). doi:10.1002/jbm.a.31217
K.I. Shingel, M.P. Faure, Biomacromolecules 6, 1635–1641 (2005). doi:10.1021/bm0492475
S. Tcholakova, N.D. Denkov, D. Sidzhakova, I.B. Ivanov, B. Campbell, Langmuir 19, 5640–5649 (2003). doi:10.1021/la034411f
H.S. Kim, S.I. Hong, S.J. Kim, J. Mater. Process. Technol. 112, 109–113 (2001). doi:10.1016/S0924-0136(01)00565-9
T. van Vliet, Colloid Polym. Sci. 266, 518–524 (1988). doi:10.1007/BF01420762
N.K. Pandit, J. Kanjia, K. Patel, D.G. Pontikes, Int. J. Pharm. 122, 27–33 (1995). doi:10.1016/0378-5173(95)00032-E
C. van der Poel, Rheol. Acta 1, 198–205 (1958). doi:10.1007/BF01968867
C.M. Wijmans, E. Dickinson, J. Chem. Soc. Faraday Trans. 94, 129–137 (1998). doi:10.1039/a706632e
G. Sala, G.A. van Aken, M. Cohen Stuart, F. van de Velde, J. Texture Stud. 38, 511–535 (2007). doi:10.1111/j.1745-4603.2007.00110.x
F.E. Mitidieri, J.R. Wagner, Food Res. Int. 35, 547–557 (2002). doi:10.1016/S0963-9969(01)00155-7
C. Solans, J.G. Dominguez, J.L. Parra, J. Heuser, S.E. Friberg, Colloid Polym. Sci. 266, 570–574 (1988). doi:10.1007/BF01420770
N. Drelon, F. Leal-Calderon, Langmuir 23, 4792–4799 (2007). doi:10.1021/la070071c
R.O. Potts, M.L. Francoeur, J. Invest. Dermatol. 96, 495–499 (1991). doi:10.1111/1523-1747.ep12470197
V.H. Mak, R.O. Potts, R.H. Guy, Pharm. Res. 8, 1064–1065 (1991). doi:10.1023/A:1015873511692
H. Tang, D. Blankschtein, R. Langer, J. Pharm. Sci. 91, 1891–1907 (2002). doi:10.1002/jps.10177
T.M. Suhonen, J.A. Bouwstra, A. Urtti, J. Control Release 59, 149–161 (1999). doi:10.1016/S0168-3659(98)00187-4
Acknowledgements
The authors are thankful to Lucas Poncelet, Maxime Paquette (Department of Engineering Physics, Ecole Polytechnique, Montreal), and Ludmila Anohina and Henadz Isakau (Bioartificial Gel Technologies) for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shingel, K.I., Roberge, C., Zabeida, O. et al. Solid emulsion gel as a novel construct for topical applications: synthesis, morphology and mechanical properties. J Mater Sci: Mater Med 20, 681–689 (2009). https://doi.org/10.1007/s10856-008-3613-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10856-008-3613-0