Abstract
Background
Epidemiological evidence suggests that there is an association between rheumatoid arthritis (RA) and Alzheimer’s disease (AD). However, the causal relationship between RA and AD remains unclear. Therefore, this study aimed to investigate the causal relationship between RA and AD.
Methods
Using publicly available genome-wide association study datasets, bidirectional two-sample Mendelian randomization (TSMR) was performed using the inverse-variance weighted (IVW), weighted median, MR‒Egger regression, simple mode, and weighted mode methods.
Results
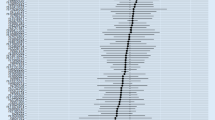
The results of MR for the causal effect of RA on AD (IVW, odds ratio [OR] = 0.959, 95% confidence interval [CI]: 0.941–0.978, P = 2.752E-05; weighted median, OR = 0.960, 95% CI: 0.937–0.984, P = 0.001) revealed a causal association between genetic susceptibility to RA and an increased risk of AD. The results of MR for the causal effect of AD on RA (IVW, OR = 0.978, 95% CI: 0.906–1.056, P = 0.576; weighted median, OR = 0.966, 95% CI: 0.894–1.043, P = 0.382) indicated that there was no causal association between genetic susceptibility to AD and an increased risk of RA.
Conclusions
The results of this two-way two-sample Mendelian randomization analysis revealed a causal association between genetic susceptibility to RA and a reduced risk of AD but did not reveal a causal association between genetic susceptibility to AD and an increased or reduced risk of RA.


Similar content being viewed by others
Data availability
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Code availability
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
References
Alzheimer’s Disease Anti-inflammatory Prevention Trial Research Group (2013) Results of a follow-up study to the randomized Alzheimer’s disease anti-inflammatory prevention trial (ADAPT). Alzheimers Dement 9:714–723. https://doi.org/10.1016/j.jalz.2012.11.012
Atri A (2019) The Alzheimer’s disease clinical spectrum: diagnosis and management. Med Clin North Am 103:263–293. https://doi.org/10.1016/j.mcna.2018.10.009
Bae SC, Lee YH (2018a) Coffee consumption and the risk of rheumatoid arthritis and systemic lupus erythematosus: a Mendelian randomization study. Clin Rheumatol 37:2875–2879. https://doi.org/10.1007/s10067-018-4278-9
Bae SC, Lee YH (2018b) Vitamin D level and risk of systemic lupus erythematosus and rheumatoid arthritis: a Mendelian randomization. Clin Rheumatol 37:2415–2421. https://doi.org/10.1007/s10067-018-4152-9
Bae SC, Lee YH (2019) Causal association between rheumatoid arthritis and a decreased risk of Alzheimer’s disease: a Mendelian randomization study. Z Rheumatol 78:359–364. https://doi.org/10.1007/s00393-018-0504-8
Bowden J, Smith GD, Haycock PC, Burgess S (2016) Consistent estimation in Mendelian randomization with some invalid instruments using a weighted median estimator. Genet Epidemiol 40:304–314. https://doi.org/10.1002/gepi.21965
Boyd TD, Bennett SP, Mori T, Governatori N, Runfeldt M, Norden M, Padmanabhan J, Neame P, Wefes I, Sanchez-Ramos J, Arendash GW, Potter H (2010) GM-CSF upregulated in rheumatoid arthritis reverses cognitive impairment and amyloidosis in Alzheimer mice. J Alzheimers Dis 21:507–518. https://doi.org/10.3233/jad-2010-091471
Breitner JC, Gau BA, Welsh KA, Plassman BL, McDonald WM, Helms MJ, Anthony JC (1994) Inverse association of anti-inflammatory treatments and Alzheimer’s disease: initial results of a co-twin control study. Neurology 44:227–232. https://doi.org/10.1212/wnl.44.2.227
Burgess S, Thompson SG (2011) Avoiding bias from weak instruments in Mendelian randomization studies. Int J Epidemiol 40:755–764. https://doi.org/10.1093/ije/dyr036
Burgess S, Thompson SG (2017) Interpreting findings from Mendelian randomization using the MR-Egger method. Eur J Epidemiol 32:377–389. https://doi.org/10.1007/s10654-017-0255-x
Burgess S, Butterworth A, Thompson SG (2013) Mendelian randomization analysis with multiple genetic variants using summarized data. Genet Epidemiol 37:658–665. https://doi.org/10.1002/gepi.21758
Butchart J, Brook L, Hopkins V et al (2015) Etanercept in Alzheimer disease: a randomized, placebo-controlled, double-blind, phase 2 trial. Neurology 84:2161–2168. https://doi.org/10.1212/wnl.0000000000001617
Cai Q, Xin Z, Zuo L, Li F, Liu B (2018) Alzheimer’s disease and rheumatoid arthritis: a Mendelian randomization study. Front Neurosci 12:627. https://doi.org/10.3389/fnins.2018.00627
Castellani G, Schwartz M (2020) Immunological features of non-neuronal brain cells: implications for Alzheimer’s disease immunotherapy. Trends Immunol 41:794–804. https://doi.org/10.1016/j.it.2020.07.005
Chen H, Ye R, Guo X (2022) Lack of causal association between heart failure and osteoporosis: a Mendelian randomization study. BMC Med Genomics 15:232. https://doi.org/10.1186/s12920-022-01385-8
Chou RC, Kane M, Ghimire S, Gautam S, Gui J (2016) Treatment for rheumatoid arthritis and risk of Alzheimer’s disease: a nested case-control analysis. CNS Drugs 30:1111–1120. https://doi.org/10.1007/s40263-016-0374-z
Darlington D, Li S, Hou H, Habib A, Tian J, Gao Y, Ehrhart J, Sanberg PR, Sawmiller D, Giunta B, Mori T, Tan J (2015) Human umbilical cord blood-derived monocytes improve cognitive deficits and reduce amyloid-β pathology in PSAPP mice. Cell Transplant 24:2237–2250. https://doi.org/10.3727/096368915x688894
Evans DM, Smith GD (2015) Mendelian randomization: new applications in the coming age of hypothesis-free causality. Annu Rev Genomics Hum Genet 16:327–350. https://doi.org/10.1146/annurev-genom-090314-050016
Fakhoury M (2018) Microglia and astrocytes in Alzheimer’s disease: implications for therapy. Curr Neuropharmacol 16:508–518. https://doi.org/10.2174/1570159x15666170720095240
Ferraccioli G, Carbonella A, Gremese E, Alivernini S (2012) Rheumatoid arthritis and Alzheimer’s disease: genetic and epigenetic links in inflammatory regulation. Discov Med 14:379–388
Fu Y, Hsiao JH, Paxinos G, Halliday GM, Kim WS (2016) ABCA7 mediates phagocytic clearance of amyloid-β in the brain. J Alzheimers Dis 54:569–584. https://doi.org/10.3233/jad-160456
Hansen DV, Hanson JE, Sheng M (2018) Microglia in Alzheimer’s disease. J Cell Biol 217:459–472. https://doi.org/10.1083/jcb.201709069
Hua L, Xiang S, Xu R, Xu X, Liu T, Shi Y, Wu L, Wang R, Sun Q (2022) Causal association between rheumatoid arthritis and celiac disease: a bidirectional two-sample Mendelian randomization study. Front Genet 13:976579. https://doi.org/10.3389/fgene.2022.976579
Huang S, Tian F, Yang X, Fang S, Fan Y, Bao J (2021) Physical activity and systemic lupus erythematosus among European populations: a two-sample Mendelian randomization study. Front Genet 12:784922. https://doi.org/10.3389/fgene.2021.784922
Huang J, Su B, Karhunen V et al (2023) Inflammatory diseases, inflammatory biomarkers, and Alzheimer disease: an observational analysis and Mendelian randomization. Neurology 100:e568–e581. https://doi.org/10.1212/wnl.0000000000201489
Jack CR, Knopman DS, Jagust WJ et al (2013) Tracking pathophysiological processes in Alzheimer’s disease: an updated hypothetical model of dynamic biomarkers. Lancet Neurol 12:207–216. https://doi.org/10.1016/s1474-4422(12)70291-0
Kiyota T, Machhi J, Lu Y, Dyavarshetty B, Nemati M, Yokoyama I, Mosley RL, Gendelman HE (2018) Granulocyte-macrophage colony-stimulating factor neuroprotective activities in Alzheimer’s disease mice. J Neuroimmunol 319:80–92. https://doi.org/10.1016/j.jneuroim.2018.03.009
Krieger M, Both M, Kranig SA, Pitzer C, Klugmann M, Vogt G, Draguhn A, Schneider A (2012) The hematopoietic cytokine granulocyte-macrophage colony stimulating factor is important for cognitive functions. Sci Rep 2:697. https://doi.org/10.1038/srep00697
Lai PH, Wang TH, Zhang NY, Wu KC, Yao CJ, Lin CJ (2021) Changes of blood-brain-barrier function and transfer of amyloid beta in rats with collagen-induced arthritis. J Neuroinflammation 18:35. https://doi.org/10.1186/s12974-021-02086-2
Lane CA, Hardy J, Schott JM (2018) Alzheimer’s disease. Eur J Neurol 25:59–70. https://doi.org/10.1111/ene.13439
Li Q, Wu Y, Chen J, Xuan A, Wang X (2022) Microglia and immunotherapy in Alzheimer’s disease. Acta Neurol Scand 145:273–278. https://doi.org/10.1111/ane.13551
Lin TM, Chen WS, Sheu JJ, Chen YH, Chen JH, Chang CC (2018) Autoimmune rheumatic diseases increase dementia risk in middle-aged patients: a nationwide cohort study. PLoS ONE 13:e0186475. https://doi.org/10.1371/journal.pone.0186475
Lin L, Luo P, Yang M, Wang J, Hou W, Xu P (2022) Causal relationship between osteoporosis and osteoarthritis: a two-sample Mendelian randomized study. Front Endocrinol (lausanne) 13:1011246. https://doi.org/10.3389/fendo.2022.1011246
Linnerbauer M, Wheeler MA, Quintana FJ (2020) Astrocyte crosstalk in CNS inflammation. Neuron 108:608–622. https://doi.org/10.1016/j.neuron.2020.08.012
Long JM, Holtzman DM (2019) Alzheimer disease: an update on pathobiology and treatment strategies. Cell 179:312–339. https://doi.org/10.1016/j.cell.2019.09.001
McAlpine CS, Park J, Griciuc A et al (2021) Astrocytic interleukin-3 programs microglia and limits Alzheimer’s disease. Nature 595:701–706. https://doi.org/10.1038/s41586-021-03734-6
McGeer PL, McGeer E, Rogers J, Sibley J (1990) Anti-inflammatory drugs and Alzheimer disease. Lancet 335:1037. https://doi.org/10.1016/0140-6736(90)91101-f
Morris GP, Clark IA, Zinn R, Vissel B (2013) Microglia: a new frontier for synaptic plasticity, learning and memory, and neurodegenerative disease research. Neurobiol Learn Mem 105:40–53. https://doi.org/10.1016/j.nlm.2013.07.002
Okada Y, Wu D, Trynka G et al (2014) Genetics of rheumatoid arthritis contributes to biology and drug discovery. Nature 506:376–381. https://doi.org/10.1038/nature12873
Policicchio S, Ahmad AN, Powell JF, Proitsi P (2017) Rheumatoid arthritis and risk for Alzheimer’s disease: a systematic review and meta-analysis and a Mendelian randomization study. Sci Rep 7:12861. https://doi.org/10.1038/s41598-017-13168-8
Price D (2003) Effect of NSAIDs on risk of Alzheimer’s disease: conclusions on NSAIDs and Alzheimer’s disease were overstated. BMJ 327:752. https://doi.org/10.1136/bmj.327.7417.752
Riaz H, Khan MS, Siddiqi TJ, Usman MS, Shah N, Goyal A, Khan SS, Mookadam F, Krasuski RA, Ahmed H (2018) Association between obesity and cardiovascular outcomes: a systematic review and meta-analysis of Mendelian randomization studies. JAMA Netw Open 1:e183788. https://doi.org/10.1001/jamanetworkopen.2018.3788
Schäbitz WR, Krüger C, Pitzer C et al (2008) A neuroprotective function for the hematopoietic protein granulocyte-macrophage colony stimulating factor (GM-CSF). J Cereb Blood Flow Metab 28:29–43. https://doi.org/10.1038/sj.jcbfm.9600496
Scheltens P, Blennow K, Breteler MM, De Strooper B, Frisoni GB, Salloway S, Van Der Flier WM (2016) Alzheimer’s disease. Lancet 388:505–517. https://doi.org/10.1016/s0140-6736(15)01124-1
Schwartzentruber J, Cooper S, Liu JZ et al (2021) Genome-wide meta-analysis, fine-mapping and integrative prioritization implicate new Alzheimer’s disease risk genes. Nat Genet 53:392–402. https://doi.org/10.1038/s41588-020-00776-w
Shultz SR, Tan XL, Wright DK, Liu SJ, Semple BD, Johnston L, Jones NC, Cook AD, Hamilton JA, O’Brien TJ (2014) Granulocyte-macrophage colony-stimulating factor is neuroprotective in experimental traumatic brain injury. J Neurotrauma 31:976–983. https://doi.org/10.1089/neu.2013.3106
Sipe GO, Lowery RL, Tremblay M, Kelly EA, Lamantia CE, Majewska AK (2016) Microglial P2Y12 is necessary for synaptic plasticity in mouse visual cortex. Nat Commun 7:10905. https://doi.org/10.1038/ncomms10905
Smolen JS, Aletaha D, McInnes IB (2016) Rheumatoid arthritis. Lancet 388:2023–2038. https://doi.org/10.1016/s0140-6736(16)30173-8
Song X, Lin Q (2017) Genomics, transcriptomics and proteomics to elucidate the pathogenesis of rheumatoid arthritis. Rheumatol Int 37:1257–1265. https://doi.org/10.1007/s00296-017-3732-3
Trzeciak P, Herbet M, Dudka J (2021) Common factors of Alzheimer’s disease and rheumatoid arthritis-pathomechanism and treatment. Molecules 26:6038. https://doi.org/10.3390/molecules26196038
Vainchtein ID, Molofsky AV (2020) Astrocytes and microglia: in sickness and in health. Trends Neurosci 43:144–154. https://doi.org/10.1016/j.tins.2020.01.003
Van Der Woude D, Van Der Helm-Van Mil AHM (2018) Update on the epidemiology, risk factors, and disease outcomes of rheumatoid arthritis. Best Pract Res Clin Rheumatol 32:174–187. https://doi.org/10.1016/j.berh.2018.10.005
Venetsanopoulou AI, Alamanos Y, Voulgari PV, Drosos AA (2022) Epidemiology of rheumatoid arthritis: genetic and environmental influences. Expert Rev Clin Immunol 18:923–931. https://doi.org/10.1080/1744666x.2022.2106970
Wake H, Moorhouse AJ, Jinno S, Kohsaka S, Nabekura J (2009) Resting microglia directly monitor the functional state of synapses in vivo and determine the fate of ischemic terminals. J Neurosci 29:3974–3980. https://doi.org/10.1523/jneurosci.4363-08.2009
Wallin K, Solomon A, Kåreholt I, Tuomilehto J, Soininen H, Kivipelto M (2012) Midlife rheumatoid arthritis increases the risk of cognitive impairment two decades later: a population-based study. J Alzheimers Dis 31:669–676. https://doi.org/10.3233/jad-2012-111736
Xiang K, Wang P, Xu Z et al (2021) Causal effects of gut microbiome on systemic lupus erythematosus: a two-sample Mendelian randomization study. Front Immunol 12:667097. https://doi.org/10.3389/fimmu.2021.667097
Xu Y, Yan H, Zhang X, Zhuo J, Han Y, Zhang H, Xie D, Lan X, Cai W, Wang X, Wang S, Li X (2022) Roles of altered macrophages and cytokines: implications for pathological mechanisms of postmenopausal osteoporosis, rheumatoid arthritis, and Alzheimer’s disease. Front Endocrinol (lausanne) 13:876269. https://doi.org/10.3389/fendo.2022.876269
Yasuoka M, Tange C, Nishita Y, Tomida M, Watanabe R, Shimokata H, Otsuka R, Kojima M (2023) Longitudinal changes in physical and cognitive functions among participants with and without rheumatoid arthritis in community-dwelling middle-aged and older adults. Ann Geriatr Med Res 27:58–65. https://doi.org/10.4235/agmr.22.0142
Zandi PP, Breitner JC (2001) Do NSAIDs prevent Alzheimer’s disease? And, if so, why? The epidemiological evidence. Neurobiol Aging 22:811–817. https://doi.org/10.1016/s0197-4580(01)00297-4
Zuroff L, Daley D, Black KL, Koronyo-Hamaoui M (2017) Clearance of cerebral Aβ in Alzheimer’s disease: reassessing the role of microglia and monocytes. Cell Mol Life Sci 74:2167–2201. https://doi.org/10.1007/s00018-017-2463-7
Funding
This study is supported by the Natural Science Foundation of Gansu Province, China. (20JR10RA358).
Author information
Authors and Affiliations
Contributions
GSL, YZY, GRM and PFL: conceptualization, investigation, data curation, writing—original draft. ZZZ, FKZ, QHC and ARZ: conceptualization, investigation, writing—original draft. HF, XY: investigation, writing—review and editing. HZG: conceptualization, writing—review and editing. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval
No ethical approval or informed consent was needed, as this study was based on previously published articles and public databases.
Consent to participate
Not applicable to this article.
Consent to publish
Not applicable to this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, GS., Yang, YZ., Ma, GR. et al. Rheumatoid arthritis is a protective factor against Alzheimer’s disease: a bidirectional two-sample Mendelian randomization study. Inflammopharmacol 32, 863–871 (2024). https://doi.org/10.1007/s10787-023-01397-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-023-01397-5