Abstract
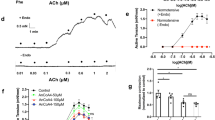
Angiotensin II (Ang II), a mitogen-activated peptide, exerts numerous effects on the cardiovascular system including the regulation of blood pressure. The current study focused on the potential mechanisms that seem to be involved in Ang II vasodilation using bovine aortic endothelial cells (BAE-1) cell lines. Expression of the Ang II receptor (AT2) in BAE-1 was checked by western blots in the presence of valsartan (AT1 inhibitor). To check if Ang II’s vasodilator impact was mediated by the nitric oxide (NO) pathway, the Griess reagent was used. Furthermore, cell-attached patch-clamp and fire-polished borosilicate electrodes with a resistance of 3–5 MΩ in the working solutions was used to record membrane currents from treated BAE-1. BEA-1 revealed 50 kDa immunoreactive bands that matched AT2. The concentration of AT2 was elevated in valsartan-treated cells in comparison to control cells. The biochemical experimental data indicated that the NO level increased in a concentration-dependent manner. Meanwhile, Ang II at a concentration of 1 µM, the level of NO increased more than at 100 µM. In patch-clamp experiments, K current and chord conductance were enhanced after incubation of Ang II with valsartan. When 100 µM Ang II was added, the current peaked rapidly and after 15 min of incubation, the maximum value was obtained, as opposed to 10 min and control (110.9 ± 13.3 pA control, 141.4 ± 30.4 pA after 10 min and 174.4 ± 49.3 pA after 15 min). Ang II type two receptor inhibitor (PD1231777) reduced the current and conductance induced by Ang II. The presented data revealed that Ang II released NO via the activation of AT2. K currents were stimulated by Ang II and evoked mainly a current consistent with the activation of K channels.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Asada H, Inoue A, Ngako Kadji FM, Hirata K, Shiimura Y, Im D, Shimamura T, Nomura N, Iwanari H, Hamakubo T, Kusano-Arai O, Hisano H, Uemura T, Suno C, Aoki J, Iwata S (2020) The crystal structure of angiotensin II type 2 receptor with endogenous peptide hormone. Structure 28:418–425. https://doi.org/10.1016/j.str.2019.12.003
Batenburg W (2005) Angiotensin II-induced vasodilation. Role of bradykinin, NO, and endothelium-derived hyperpolarizing factors. PhD Thesis, Erasmus University, Rotterdam.
Gallinat S, Busche S, Raizada MK, Sumners C (2000) The angiotensin II type 2 receptor: an enigma with multiple variations. Am J Physiol Endocrinol Metab 278:E357–E374. https://doi.org/10.1152/ajpendo.2000.278.3.E357
Gao L, Zucker IH (2011) AT2 receptor signaling and sympathetic regulation. Curr Opin Pharmacol 11:124–130. https://doi.org/10.1016/j.coph.2010.11.004
Gao J, Chao J, Parbhu KJ, Yu L, Xiao L, Gao F, Gao L (2012) Ontogeny of angiotensin type 2 and type 1 receptor expression in mice. J Renin Angiotensin Aldosterone Syst 13:341–352. https://doi.org/10.1177/1470320312443720
Gauthier KM, Zhang DX, Edwards EM, Holmes B, Campbell WB (2005) Angiotensin II dilates bovine adrenal cortical arterioles: role of endothelial nitric oxide. Endocrinology 146:3319–3324. https://doi.org/10.1210/en.2005-0129
Guimond MO, Wallinder C, Alterman M, Hallberg A, Gallo-Payet N (2013) Comparative functional properties of two structurally similar selective nonpeptide drug-like ligands for the angiotensin II type-2 (AT(2)) receptor. Effects on neurite outgrowth in NG108-15 cells. Eur J Pharmacol 699:160–171. https://doi.org/10.1016/j.ejphar.2012.11.032
Hayabuchi Y, Nakaya Y, Yasui S, Mawatari K, Mori K, Suzuki M, Kagami S (2006) Angiotensin II activates intermediate-conductance Ca2+-activated K + channels in arterial smooth muscle cells. J Mol Cell Cardiol 41:972–979. https://doi.org/10.1016/j.yjmcc.2006.07.010
Ingraham NE, Barakat AG, Reilkoff R, Bezdicek T, Schacker T, Chipman JG, Tignanelli CJ, Puskarich MA (2020) Understanding the renin-angiotensin-aldosterone-SARS-CoV axis: a comprehensive review. Eur Respir J 56:2000912. https://doi.org/10.1183/13993003.00912-2020
Iqbal H, Verma AK, Yadav P, Alam S, Shafiq M, Mishra D, Khan F, Hanif K, Negi AS, Chanda D (2021) Antihypertensive Effect of a Novel angiotensin II receptor blocker Fluorophenyl Benzimidazole: contribution of cGMP, voltage-dependent Calcium channels, and BKCa channels to Vasorelaxant mechanisms. Front Pharmacol 12:611109. https://doi.org/10.3389/fphar.2021.611109
Kaschina E, Namsolleck P, Unger T (2017) AT2 receptors in cardiovascular and renal diseases. Pharmacol Res 125:39–47. https://doi.org/10.1016/j.phrs.2017.07.008
Kemp BA, Howell NL, Keller SR, Gildea JJ, Padia SH, Carey RM (2016) AT2 receptor activation prevents Sodium Retention and reduces blood pressure in angiotensin II-Dependent Hypertension. Circul Res 119:532–543. https://doi.org/10.1161/CIRCRESAHA.116.308384
Kizub IV, Lakhkar A, Dhagia V, Joshi SR, Jiang H, Wolin MS, Falck JR, Koduru SR, Errabelli R, Jacobs ER, Schwartzman ML, Gupte SA (2016) Involvement of gap junctions between smooth muscle cells in sustained hypoxic pulmonary vasoconstriction development: a potential role for 15-HETE and 20-HETE. Am J Physiol Lung Cell Mol Physiol 310:L772–L783. https://doi.org/10.1152/ajplung.00377.2015
Li J, Zhao X, Li X, Lee KM, Olson SC (2007) Angiotensin II type 2 receptor-dependent increases in nitric oxide synthase expression in the pulmonary endothelium are mediated via a G alpha i3/Ras/Raf/MAPK pathway. Am J Physiol Cell Physiol 292:C2185–C2196. https://doi.org/10.1152/ajpcell.00204.2006
Li Y, Li XH, Yuan H (2012) Angiotensin II type-2 receptor-specific effects on the cardiovascular system. Cardiovasc Diagn Ther 2:56–62. https://doi.org/10.3978/j.issn.2223-3652.2012.02.02
Li XC, Zhang J, Zhuo JL (2017) The vasoprotective axes of the renin-angiotensin system: physiological relevance and therapeutic implications in cardiovascular, hypertensive and kidney Diseases. Pharmacol Res 125:21–38. https://doi.org/10.1016/j.phrs.2017.06.005
Loiola RA, Reis FC, Kawamoto EM, Scavone C, Abdalla DS, Fernandes L, Pesquero JB (2011) Role of vascular kinin B1 and B2 receptors in endothelial nitric oxide metabolism. Peptides 32:1700–1705. https://doi.org/10.1016/j.peptides.2011.06.010
Ni W, Yang X, Yang D, Bao J, Li R, Xiao Y, Hou C, Wang H, Liu J, Yang D, Xu Y, Cao Z, Gao Z (2020) Role of angiotensin-converting enzyme 2 (ACE2) in COVID-19. Crit Care 24:422. https://doi.org/10.1186/s13054-020-03120-0
Pueyo ME, Gonzalez W, Nicoletti A, Savoie F, Arnal JF, Michel JB (2000) Angiotensin II stimulates endothelial vascular cell adhesion molecule-1 via nuclear factor-kappab activation induced by intracellular oxidative stress. Arterioscler Thromb Vasc Biol 20:645–651. https://doi.org/10.1161/01.atv.20.3.645
Senbel AM, Elmoneim A, Sharabi HM, Mohy El-Din MM (2017) Neuronal voltage-gated potassium channels may modulate nitric oxide synthesis in Corpus Cavernosum. Front Pharmacol 8:297. https://doi.org/10.3389/fphar.2017.00297
Singh KD, Karnik SS (2016) Angiotensin receptors: structure, function, signaling and clinical applications. J cell Signal 1:111. https://doi.org/10.4172/jcs.1000111
Sollini M, Frieden M, Bény JL (2002) Charybdotoxin-sensitive small conductance K(ca) channel activated by bradykinin and substance P in endothelial cells. Br J Pharmacol 136:1201–1209. https://doi.org/10.1038/sj.bjp.0704819
Sturrock BR, Milne KM, Chevassut TJ (2020) The renin-angiotensin system - a therapeutic target in COVID-19? Clin Med 20:e72–e75. https://doi.org/10.7861/clinmed.2020-0146
Taguchi K, Matsumoto T, Kamata K, Kobayashi T (2012) Angiotensin II type 2 receptor-dependent increase in nitric oxide synthase activity in the endothelium of db/db mice is mediated via an MEK pathway. Pharmacol Res 66:41–50. https://doi.org/10.1016/j.phrs.2012.02.010
Tsukamoto I, Inoue S, Teramura T, Takehara T, Ohtani K, Akagi M (2013) Activating types 1 and 2 angiotensin II receptors modulate the hypertrophic differentiation of chondrocytes. FEBS open bio 3:279–284. https://doi.org/10.1016/j.fob.2013.07.001
van Dalen JW, Marcum ZA, Gray SL, Barthold D, van Moll EP, van Gool WA, Crane PK, Larson EB, Richard E (2021) Association of Angiotensin II-Stimulating antihypertensive use and Dementia risk: Post Hoc Analysis of the PreDIVA Trial. Neurology 96:e67–e80. https://doi.org/10.1212/WNL.0000000000010996
van der Graaf AM, Wiegman MJ, Plösch T, Zeeman GG, van Buiten A, Henning RH, Buikema H, Faas MM (2013) Endothelium-dependent relaxation and angiotensin II sensitivity in experimental preeclampsia. PLoS ONE 8:e79884. https://doi.org/10.1371/journal.pone.0079884
Wingler LM, Elgeti M, Hilger D, Latorraca NR, Lerch MT, Staus DP, Dror RO, Kobilka BK, Hubbell WL, Lefkowitz RJ (2019a) Angiotensin analogs with Divergent Bias stabilize distinct receptor conformations. Cell 176:468-478e11. https://doi.org/10.1016/j.cell.2018.12.005
Wingler LM, McMahon C, Staus DP, Lefkowitz RJ, Kruse AC (2019b) Distinctive activation mechanism for angiotensin receptor revealed by a synthetic nanobody. Cell 176:479-490e12. https://doi.org/10.1016/j.cell.2018.12.006
Zhao X, Li X, Trusa S, Olson SC (2005) Angiotensin type 1 receptor is linked to inhibition of nitric oxide production in pulmonary endothelial cells. Regul Pept 132:113–122. https://doi.org/10.1016/j.regpep.2005.09.010
Acknowledgements
We’d like to express our gratitude to the Consiglio Nazionale delle Ricerche-Istituto di Biofisica in Genova/ Italy for providing permission to work in their labs.
Funding
This research did not receive any grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
CMM designed, coordinated, analyzed the data, and wrote the manuscript draft. OAMA supervised the project and revised the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare there is no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mohammed, C.M., Al-Habib, O.A. Nitric oxide-cyclic GMP role in Ang II induced hyperpolarization in bovine aortic endothelium cell line (BAE-1). Cytotechnology 76, 113–121 (2024). https://doi.org/10.1007/s10616-023-00602-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-023-00602-1