Abstract
A novel bacterial strain, TLK-CK17T, was isolated from cow dung compost sample. The strain was Gram-staining negative, non-gliding rods, aerobic, and displayed growth at 15–40 °C (optimally, 35 °C), with 0–5.0% (w/v) NaCl (optimally, 0.5) and at pH 6.5–8.5 (optimally, 7.0–7.5). The assembled genome of strain TLK-CK17T has a total length of 4.3 Mb with a G + C content of 68.2%. According to the genome analysis, strain TLK-CK17T encodes quite a few glycoside hydrolases that may play a role in the degradation of accumulated plant biomass in compost. On the basis 16S rRNA gene sequence analysis, strain TLK-CK17T showed the highest sequence similarity (98.9%) with L. penaei GDMCC 1.1817 T, followed by L. maris KCTC 42381 T (98.3%). Cells contained iso-C16:0, iso-C15:0, and summed feature 9 (comprising C17:1 ω9c and/or 10-methyl C16:0), as its major cellular fatty acids (> 10.0%) and ubiquinone-8 as the exclusively respiratory quinone. Diphosphatidylglycerol, phosphatidylethanolamine, and phosphatidylglycerol prevailed among phospholipids. Based on the phenotypic, genomic and phylogenetic data, strain TLK-CK17T represents a novel species of the genus Lysobacter, for which the name Lysobacter chinensis sp. nov. is proposed, and the type strain is TLK-CK17T (= CCTCC AB2021257T = KCTC 92122 T).


Similar content being viewed by others
Data availability
The genome and 16S rRNA gene sequence are available from GenBank under the accession numbers provided in the manuscript.
References
Angiuoli SV, Gussman A, Klimke W, Cochrane G, Field D et al (2008) Toward an online repository of standard operating procedures (SOPs) for (meta) genomic annotation. OMICS 12:137–141. https://doi.org/10.1089/omi.2008.0017
Bowman JP (2000) Description of Cellulophaga algicola sp. nov., isolated from the surfaces of Antarctic algae, and reclassification of Cytophaga uliginosa (ZoBell and Upham 1944) Reichenbach 1989 as Cellulophaga uliginosa comb. nov. Int J Syst Evol Microbiol 50:1861–1868. https://doi.org/10.1099/00207713-50-5-1861
Chen W, Zhao YL, Cheng J, Zhou XK, Salam N et al (2016) Lysobacter cavernae sp. nov., a novel bacterium isolated from a cave sample. Antonie Van Leeuwenhoek 109:1047–1053. https://doi.org/10.1007/s10482-016-0704-7
Christensen P, Cook FD (1978) Lysobacter, a new genus of nonfruiting, gliding bacteria with a high base ratio. Int J Syst Bacteriol 28:367–393
Christensen P (2005) Genus IV Lysobacter Christensen and Cook 1978 372AL. In: Brenner DJ, Krieg NR, Staley JT, Garrity GM (eds) Bergey’s manual of systematic bacteriology, 2nd edn. Springer, New York, pp 95–101
CLSI (2012) Performance standards for antimicrobial susceptibility Testing, Twenty-second informational supplement CLSI document M100–S22. Clinical and Laboratory Standards Institute.
Cowan ST, Steel KJ (1974) Bacterial characters and characterization, 2nd edn. Cambridge University Press, Cambridge
de Bruijn I, Cheng X, de Jager V, Expósito RG (2015) Comparative genomics and metabolic profiling of the genus Lysobacter. BMC Genomics 16:991. https://doi.org/10.1186/s12864-015-2191-z
Dong XZ, Cai MY (2001) Determination of biochemical characteristics. In: Dong XZ, Cai MY (eds) Manual for the systematic identification of general bacteria. Science Press, Beijing, pp 370–398
Fang DB, Han JR, Liu Y, Du ZJ (2017) Seonamhaeicola marinus sp. nov., isolated from marine algae. Int J Syst Evol Microbiol 67:4857–4861. https://doi.org/10.1099/ijsem.0.002396
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17(6):368–376. https://doi.org/10.1007/BF01734359
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolut Int J Org Evolut 39(4):783–791. https://doi.org/10.1111/j.1558-5646.1985.tb00420.x
Fitch WM (1971) Towards defining the course of evolution: minimal change for a specified tree topology. Syst Zool 20:406–416. https://doi.org/10.2307/2412116
Foster PL (2005) Stress responses and genetic variation in bacteria. Mutat Res 569(1–2):3–11. https://doi.org/10.1016/j.mrfmmm.2004.07.017
Goris J, Konstantinidis KT, Klappenbach JA, Coenye T, Vandamme P et al (2007) DNA-DNA hybridization values and their relationship to whole-genome sequence similarities. Int J Syst Evol Microbiol 57:81–91. https://doi.org/10.1099/ijs.0.64483-0
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120. https://doi.org/10.1007/BF01731581
Kumar S, Stecher G, Tamura K (2016) MEGA 7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Lee D, Jang JH, Cha S, Seo T (2017) Lysobacter humi sp. nov., isolated from soil. Int J Syst Evol Microbiol 67:951–955. https://doi.org/10.1099/ijsem.0.001722
Li R, Li Y, Kristiansen K, Wang J (2008) SOAP: short oligonucleotide alignment program. Bioinformatics 24:713–714. https://doi.org/10.1093/bioinformatics/btn025
Li R, Zhu H, Ruan J, Qian W, Fang X (2010) De novo assembly of human genomes with massively parallel short read sequencing. Genome Res 20:265–272. https://doi.org/10.1101/gr.097261.109
Li J, Han Y, Guo W, Wang Q, Wang G (2018) Lysobacter tongrenensis sp. nov., isolated from soil of a manganese factory. Arch Microbiol 200:439–444. https://doi.org/10.1007/s00203-017-1457-z
Liu M, Liu Y, Wang Y, Luo X, Dai J, Fang C (2011) Lysobacter xinjiangensis sp. nov., a moderately thermotolerant and alkalitolerant bacterium isolated from a gamma-irradiated sand soil sample. Int J Syst Evol Microbiol 61:433–437. https://doi.org/10.1099/ijs.0.016931-0
Liu QQ, Li XL, Rooney AP, Du ZJ, Chen GJ (2014) Tangfeifania diversioriginum gen. nov., sp. nov., a representative of the family Draconibacteriaceae. Int J Syst Evol Microbiol 64:3473–3477. https://doi.org/10.1099/ijs.0.066902-0
Margesin R, Zhang DC, Albuquerque L, Froufe HJ, Egas C (2018) Lysobacter silvestris sp. nov., isolated from alpine forest soil, and reclassifcation of Luteimonas tolerans as Lysobacter tolerans comb. nov. Int J Syst Evol Microbiol 68:1571–1577. https://doi.org/10.1099/ijsem.0.002710
Meier-Kolthoff JP, Auch AF, Klenk HP, Göker M (2013) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinform 14:60. https://doi.org/10.1186/1471-2105-14-60
Minnikin DE, O’Donnell AG, Goodfellow M, Alderson G, Athalye M et al (1984) An integrated procedure for the extraction of bacterial isoprenoid quinones and polar lipids. J Microbiol Methods 2:233–241. https://doi.org/10.1016/0167-7012(84)90018-6
Na SI, Kim YO, Yoon SH, Ha S, Baek I et al (2018) UBCG: Up-to-date bacterial core gene set and pipeline for phylogenomic tree reconstruction. J Microbiol 56:280–285. https://doi.org/10.1007/s12275-018-8014-6
Palumbo JD, Yuen GY, Jochum CC, Tatum K, Kobayashi DY (2005) Mutagenesis of beta-1, 3-glucanase genes in Lysobacter enzymogenes strain c3 results in reduced biological control activity toward bipolaris leaf spot of tall fescue and pythium damping-off of sugar beet. Phytopathology 95:701–707. https://doi.org/10.1094/PHYTO-95-0701
Pidot SJ, Coyne S, Kloss F, Hertweck C (2014) Antibiotics from neglected bacterial sources. Int J Med Microbiol 304:14–22. https://doi.org/10.1016/j.ijmm.2013.08.011
Price MN, Dehal PS, Arkin AP (2010) FastTree 2 – approximately maximum-likelihood trees for large alignments. PLoS ONE 2010(5):e9490. https://doi.org/10.1371/journal.pone.0009490
Saitou N, Nei M (1987) The neighbour-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a0
Sasser M (1990) Identification of bacteria by gas chromatography of cellular fatty acids, MIDI technical note 101. Microbial ID Inc, Newark, DE
Siddiqi MZ, Im WT (2016) Lysobacter pocheonensis sp. nov., isolated from soil of a ginseng feld. Arch Microbiol 198:551–557. https://doi.org/10.1007/s00203-016-1214-8
Spudich JL, Yang CS, Jung KH, Spudich EN (2000) Retinylidene proteins: structures and functions from archaea to humans. Annu Rev Cell Dev Biol 16:365–392. https://doi.org/10.1146/annurev.cellbio.16.1.365
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882. https://doi.org/10.1093/nar/25.24.4876
Thompson CC, Chimetto L, Edwards RA, Swings J, Stackebrandt E et al (2013) Microbial genomic taxonomy. BMC Genomics 14:913. https://doi.org/10.1186/1471-2164-14-913
Tindall BJ, Sikorski J, Smibert RM, Krieg NR (2007) Phenotypic characterization and the principles of comparative systematics. In: Reddy CA, Beveridge TJ, Breznak JA, Marzluf G, Schmidt TM et al (eds) Methods for general and molecular microbiology, 3rd edn. ASM Press, Washington, DC, pp 330–393
Trifinopoulos J, Nguyen LT, von Haeseler A, Minh BQ (2016) W-IQ-TREE: a fast online phylogenetic tool for maximum likelihood analysis. Nucleic Acids Res 44:W232–W235. https://doi.org/10.1093/nar/gkw256
Xie Y, Wright S, Shen Y, Du L (2012) Bioactive natural products from Lysobacter. Nat Prod Rep 29:1277–1287. https://doi.org/10.1039/c2np20064c
Zhang Z, Yuen GY, Sarath G, Penheiter AR (2001) Chitinases from the plant disease biocontrol agent, Stenotrophomonas maltophilia C3. Phytopathology 91(2):204–211. https://doi.org/10.1094/PHYTO.2001.91.2.204
Kroppenstedt RM (1982) Separation of bacterial menaquinones by HPLC using reverse phase (RP18) and a silver loaded ion exchanger as stationary phases. J Liq Chromatogr aphy 5:2359–2367. https://doi.org/10.1080/01483918208067640
Wayne LG (1988) International committee on systematic bacteriology: announcement of the report of the ad hoc committee on reconciliation of Approaches to bacterial systematics. Syst Appl Microbiol 10(2):99–100. https://doi.org/10.1016/S0723-2020(88)80020-1
Funding
This work was supported by the Xinjiang Academy of Agricultural Sciences Young Science and Technology Backbone Innovation Ability Training Project (xjnkq-2022019), Regional Collaborative Innovation Special Project of Xinjiang Uygur Autonomous Region (Nos. 2021E02022) and Forestry Development Subsidy Fund Project of Xinjiang Uygur Autonomous Region (Nos. XJLYKJ-2021–15).
Author information
Authors and Affiliations
Contributions
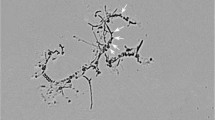
All authors contributed to the study conception and design. YYL wrote the manuscript and analysed the cultivation data. LYZ performed the genomic and phylogenetic analysis. YXP and PBL isolated the strain and performed the initial cultivation and strain deposition. YXX, JPD and ZQS contributed to text preparation and revised the manuscript. LF performed the electron microscopic analysis and prepared the SEM pictures. XWW and ZFW took the samples. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethics approval
This article does not contain any studies with animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, Y., Zhou, L., Yang, X. et al. Lysobacter chinensis sp. nov., a cellulose-degrading strain isolated from cow dung compost. Antonie van Leeuwenhoek 115, 1031–1040 (2022). https://doi.org/10.1007/s10482-022-01755-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-022-01755-3