Abstract
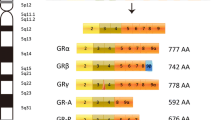
Glucocorticoid receptor (GR) has been implicated in prostate carcinoma growth and progression. Glucocorticoid receptor beta (GRβ) acts as an inhibitor of GR; however, its function is not well understood. Serum- and glucocorticoid-regulated kinase 1 (SGK1) is a GR-responsive gene that phosphorylates N-myc downstream-regulated gene 1 (NDRG1) and is involved in cancer growth and invasion. However, the expression of GR, GRβ, SGK1, and NDRG1 in prostate cancer and their relationship with clinicopathological and functional significance remain unknown. The association between the status of GR, GRβ, SGK1, and NDRG1 immunoreactivity and clinicopathological variables was analyzed in patients with prostate carcinoma to explore their clinical significance. In prostate carcinoma cases, the relative abundance of GR and NDRG1 immunoreactivity was inversely and significantly associated with the primary tumor stage (pT), while GR immunoreactivity was inversely and significantly associated with the Ki-67 score. The relative expression status of NDRG1 was significantly associated with that of GR. However, no significant correlation was observed between any of the clinicopathological parameters and GRβ and SGK1 expression. Our findings indicate that GR and NDRG1 expression status is correlated with clinicopathological features in patients with prostate cancer.



Similar content being viewed by others
References
Kach J, Conzen SD, Szmulewitz RZ (2015) Targeting the glucocorticoid receptor in breast and prostate cancers. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aac7531
Abduljabbar R, Negm OH, Lai CF, Jerjees DA, Al-Kaabi M, Hamed MR, Tighe PJ, Buluwela L, Mukherjee A, Green AR, Ali S, Rakha EA, Ellis IO (2015) Clinical and biological significance of glucocorticoid receptor (GR) expression in breast cancer. Breast Cancer Res Treat 150:335–346. https://doi.org/10.1007/s10549-015-3335-1
Chen X, Chen F, Ren Y, Weng G, Keng PC, Chen Y, Lee SO (2019) Glucocorticoid receptor upregulation increases radioresistance and triggers androgen independence of prostate cancer. Prostate 79:1386–1398. https://doi.org/10.1002/pros.23861
Shah N, Wang P, Wongvipat J, Karthaus WR, Abida W, Rockowitz AJ, S, Drier Y, Bernstein BE, Long HW, Freedman ML, Arora VK, Zheng D, Sawyers CL. (2017) Regulation of the glucocorticoid receptor via a BET-dependent enhancer drives antiandrogen resistance in prostate cancer. Elife 6:1–19
Puhr M, Hoefer J, Eigentler A, Ploner C, Handle F, Schaefer G, Kroon J, Leo A, Heidegger I, Eder I, Culig Z, Van der Pluijm G, Klocker H (2018) The glucocorticoid receptor is a key player for prostate cancer cell survival and a target for improved antiandrogen therapy. Clin Cancer Res 24:927–938. https://doi.org/10.1158/1078-0432.CCR-17-0989
McBeth L, Grabnar M, Selman S, Hinds TD (2015) Involvement of the androgen and glucocorticoid receptors in bladder cancer. Int J Endocrinol 2015:384860. https://doi.org/10.1155/2015/384860
Hinds TDJ, Ramakrishnan S, Cash HA, Stechschulte LA, Heinrich G, Najjar SM, Sanchez ER (2010) Discovery of glucocorticoid receptor-beta in mice with a role in metabolism. Mol Endocrinol 24:1715–1727. https://doi.org/10.1210/me.2009-0411
Kino T, Su YA, Chrousos GP (2009) Human glucocorticoid receptor isoform beta: recent understanding of its potential implications in physiology and pathophysiology. Cell Mol Life Sci 66:3435–3448. https://doi.org/10.1007/s00018-009-0098-z
McBeth L, Nwaneri AC, Grabnar M, Demeter J, Nestor-Kalinoski A, Hinds TD (2016) Glucocorticoid receptor beta increases migration of human bladder cancer cells. Oncotarget 7:27313–27324
Lin KT, Wang LH (2016) New dimension of glucocorticoids in cancer treatment. Steroids 111:84–88. https://doi.org/10.1016/j.steroids.2016.02.019
Mikosz CA, Brickley DR, Sharkey MS, Moran TW, Conzen SD (2001) Glucocorticoid receptor-mediated protection from apoptosis is associated with induction of the serine/threonine survival kinase gene, sgk-1. J Biol Chem 276:16649–16654. https://doi.org/10.1074/jbc.M010842200
Sahoo S, Brickley DR, Kocherginsky M, Conzen SD (2005) Coordinate expression of the PI3-kinase downstream effectors serum and glucocorticoid-induced kinase (SGK-1) and Akt-1 in human breast cancer. Eur J Cancer 41:2754–2759. https://doi.org/10.1016/j.ejca.2005.07.018
Yao Y, Jiang Q, Jiang L, Wu J, Zhang Q, Wang J, Feng H, Zang P (2016) Lnc-SGK1 induced by helicobacter pylori infection and highsalt diet promote Th2 and Th17 differentiation in human gastric cancer by SGK1/Jun B signaling. Oncotarget 7:20549–20560
Xiaobo Y, Qiang L, Xiong Q, Zheng R, Jianhua Z, Zhifeng L, Yijiang S, Zheng J (2016) Serum and glucocorticoid kinase 1 promoted the growth and migration of non-small cell lung cancer cells. Gene 576:339–346. https://doi.org/10.1016/j.gene.2015.10.072
Brunet A, Park J, Tran H, Hu LS, Hemmings BA, Greenberg ME (2001) Protein kinase SGK mediates survival signals by phosphorylating the forkhead transcription factor FKHRL1 (FOXO3a). Mol Cell Biol 21:952–965. https://doi.org/10.1128/MCB.21.3.952-965.2001
Murray JT, Campbell DG, Morrice N, Auld GC, Shpiro N, Marquez R, Peggie M, Bain J, Bloomberg GB, Grahammer F, Lang F, Wulff P, Kuhl D, Cohen P (2004) Exploitation of Kestrel to identify NDRG family members as physiological substrates for SGK1 and GSK3. Biochem J 384:477–488. https://doi.org/10.1042/BJ20041057
Melotte V, Qu X, Ongenaert M, van Criekinge W, de Bruïne AP, Baldwin HS, van Engeland M (2010) The N-myc downstream regulated gene (NDRG) family: diverse functions, multiple applications. FASEB J 24:4153–4166. https://doi.org/10.1096/fj.09-151464
Hata S, Ise K, Azmahani A, Konosu-Fukaya S, McNamara KM, Fujishima F, Shimada K, Mitsuzuka K, Arai Y, Sasano H, Nakamura Y (2017) Expression of AR, 5αR1 and 5αR2 in bladder urothelial carcinoma and relationship to clinicopathological factors. Life Sci 190:15–20. https://doi.org/10.1016/j.lfs.2017.09.029
Nakamura Y, Suzuki T, Fukuda T, Ito A, Endo M, Moriya T, Arai Y, Sasano H (2006) Steroid sulfatase and estrogen sulfotransferase in human prostate cancer. Prostate 66:1005–1012. https://doi.org/10.1002/pros.20426
Ueki S, Fujishima F, Kumagai T, Ishida H, Okamoto H, Takaya K, Sato C, Taniyma Y, Kamei T, Sasano H (2020) GR, Sgk1, and NDRG1 in esophageal squamous cell carcinoma: Their correlation with therapeutic outcome of neoadjuvant chemotherapy. BMC Cancer 20:161. https://doi.org/10.1186/s12885-020-6652-7
Gokon Y, Fujishima F, Taniyama Y, Ishida H, Yamagata T, Sawai T, Uzuki M, Ichikawa H, Itakura Y, Takahashi K, Yajima N, Hagiwara M, Nishida A, Ozawa Y, Sakuma T, Sakamoto K, Zuguchi M, Saito M, Kamei T, Sasano H (2020) Glucocorticoid receptor and serum- and glucocorticoid-induced kinase-1 in esophageal adenocarcinoma and adjacent Barrett’s esophagus. Pathol Int 70:355–363. https://doi.org/10.1111/pin.12922
Zhao XY, Malloy PJ, Krishnan AV, Swami S, Navone NM, Peehl DM, Feldman D (2000) Glucocorticoids can promote androgen-independent growth of prostate cancer cells through a mutated androgen receptor. Nat Med 6:703–706. https://doi.org/10.1038/76287
Isikbay M, Otto K, Kregel S, Kach J, Cai Y, Vander Griend DJ, Conzen SD, Szmulewitz RZ (2014) Glucocorticoid receptor activity contributes to resistance to androgen-targeted therapy in prostate cancer. Horm Cancer 5:72–89. https://doi.org/10.1007/s12672-014-0173-2
Nakamura Y, Suzuki T, Nakabayashi M, Endoh M, Sakamoto K, Mikami Y, Moriya T, Ito A, Takahashi S, Yamada S, Arai Y, Sasano H (2005) In situ androgen producing enzymes in human prostate cancer. Endocr Relat Cancer 12:101–107. https://doi.org/10.1677/erc.1.00914
Lewis-Tuffin LJ, Cidlowski JA (2006) The physiology of human glucocorticoid receptor β (hGRbeta) and glucocorticoid resistance. Ann N Y Acad Sci 1069:1–9. https://doi.org/10.1196/annals.1351.001
Di Cristofano A (2017) SGK1: The dark side of PI3K signaling. Curr Top Dev Biol 123:49–71. https://doi.org/10.1016/bs.ctdb.2016.11.006
Liang X, Lan C, Zhou J, Fu W, Long X, An Y, Jiao G, Wang K, Li Y, Xu J, Huang Q, Xu B, Xiao J (2017) Development of a new analog of SGK1 inhibitor and its evaluation as a therapeutic molecule of colorectal cancer. J Cancer 8:2256–2262. https://doi.org/10.7150/jca.19566
Liang X, Lan C, Jiao G, Fu W, Long X, An Y, Wang K, Zhou J, Chen T, Li Y, Xu J, Huang Q, Xu B, Xiao J (2017) Therapeutic inhibition of SGK1 suppresses colorectal cancer. Exp Mol Med 49:e399. https://doi.org/10.1038/emm.2017.184
Panettieri RA, Schaafsma D, Amrani Y, Koziol-White C, Ostrom R, Tliba O (2019) Non-genomic effects of glucocorticoids: An updated view. Trends Pharmacol Sci 40:38–49. https://doi.org/10.1016/j.tips.2018.11.002
Nuñez FJ, Johnstone TB, Corpuz ML, Kazarian AG, Mohajer NN, Tliba O, Panettieri RA Jr, Koziol White C, Roosan MR, Ostrom RS (2020) Glucocorticoids rapidly activate cAMP production via Gαs to initiate non-genomic signaling that contributes to one-third of their canonical genomic effects. FASEB J 34:2882–2895. https://doi.org/10.1096/fj.201902521R
Bandyopadhyay S, Pai SK, Gross SC, Hirota S, Hosobe S, Miura K, Saito K, Commes T, Hayashi S, Watabe M, Watabe K (2003) The Drg-1 gene suppresses tumor metastasis in prostate cancer. Cancer Res 63:1731–1736
Bandyopadhyay S, Pai SK, Hirota S, Hosobe S, Tsukada T, Miura K, Takano Y, Saito K, Commes T, Piquemal D, Watabe M, Gross S, Wang Y, Huggenvik J, Watabe K (2004) PTEN up-regulates the tumor metastasis suppressor gene Drg-1 in prostate and breast cancer. Cancer Res 64:7655–7660. https://doi.org/10.1158/0008-5472.CAN-04-1623
Fang BA, Kovačević Ž, Park KC, Kalinowski DS, Jansson PJ, Lane DJR, Sahni S, Richardson DR (2014) Molecular functions of the iron-regulated metastasis suppressor, NDRG1, and its potential as a molecular target for cancer therapy. Biochim Biophys Acta 1845:1–19. https://doi.org/10.1016/j.bbcan.2013.11.002
Lim SC, Geleta B, Maleki S, Richardson DR, Kovačević Ž (2021) The metastasis suppressor NDRG1 directly regulates androgen receptor signaling in prostate cancer. J Biol Chem 297:101414. https://doi.org/10.1016/j.jbc.2021.101414
Bae DH, Jansson PJ, Huang ML, Kovacevic Z, Kalinowski D, Lee CS, Sahni S, Richardson DR (2013) The role of NDRG1 in the pathology and potential treatment of human cancers. J Clin Pathol 66:911–917. https://doi.org/10.1136/jclinpath-2013-201692
Ellen TP, Ke Q, Zhang P, Costa M (2008) NDRG1, a growth and cancer related gene: regulation of gene expression and function in normal and disease states. Carcinogenesis 29:2–8. https://doi.org/10.1093/carcin/bgm200
Chen Z, Zhang D, Yue F, Zheng M, Kovacevic Z, Richardson DR (2012) The iron chelators Dp44mT and DFO inhibit TGF-β-induced epithelial-mesenchymal transition via up-regulation of N-Myc downstream-regulated gene 1 (NDRG1). J Biol Chem 287:17016–17028. https://doi.org/10.1074/jbc.M112.350470
Murakami Y, Hosoi F, Izumi H, Maruyama Y, Ureshino H, Watari K, Kohno K, Kuwano M, Ono M (2010) Identification of sites subjected to serine/threonine phosphorylation by SGK1 affecting N-myc downstream-regulated gene 1 (NDRG1)/Cap43-dependent suppression of angiogenic CXC chemokine expression in human pancreatic cancer cells. Biochem Biophys Res Commun 396:376–381. https://doi.org/10.1016/j.bbrc.2010.04.100
Zhou R, Snyder PM (2005) Nedd4–2 phosphorylation induces serum and glucocorticoid-regulated kinase (SGK) ubiquitination and degradation. J Biol Chem 280:4518–4523. https://doi.org/10.1074/jbc.M411053200
Acknowledgements
This study was conducted in cooperation with the common equipment room of Tohoku Medical and Pharmaceutical University Faculty of Medicine. We thank the members of the Department of Pathology, Tohoku University Graduate School of Medicine for their support. We thank Editage (www.editage.com) for English language editing.
Funding
This work was supported by JSPS KAKENHI Grant Number JP18K06995.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hata, S., Shimada, H., Sato, N. et al. Expression and clinicopathological significance of glucocorticoid receptor, SGK1, and NDRG1 in hormone-naïve prostate carcinoma. Med Mol Morphol 55, 283–291 (2022). https://doi.org/10.1007/s00795-022-00332-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00795-022-00332-x