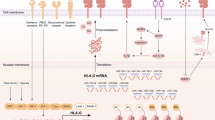
Abstract
Purpose
As molecules responsible for presenting antigens to T lymphocytes, leukocytes antigens (HLAs) play a vital role in cancer immunology. This review aims to provide current understanding of HLAs in tumour immunology.
Methods
Perspectives on how HLA alterations may contribute to the immune escape of cancer cells and resistance to immunotherapy, and potential methods to overcome HLA defects were summarized. In addition, we discussed the potential association between HLA and immune-related adverse events (irAEs), which has not been reviewed elsewhere.
Results
Downregulation, loss of heterogeneity and entire loss of HLAs are responsible for the immune escape of tumour cells. The strategies to overcome the HLA defects can be effective therapies of cancer. Compared with classical HLA-I, non-classical HLA-I molecules, such as HLA-E and HLA-G, appear to be more reliable predictors of prognosis, as they tend to play immunosuppressive roles in antitumor response. Relative diversified or high expression of classical HLA-I are potential predictors of favourable response of immunotherapy. Certain HLA types may be associated to enhanced affinity to self-antigen-mimicked tumour-antigens, thus may positively correlated to irAEs triggered by checkpoint inhibitors.
Conclusions
Further studies exploring the relationship between HLAs and cancer may not only lead to the development of novel therapies but also bring about better management of irAEs.


Similar content being viewed by others
Data availability
Not applicable.
References
Ali OH, Berner F, Bomze D, Fässler M, Diem S, Cozzio A, Jörger M, Früh M, Driessen C, Lenz TL, Flatz L (2019) Human leukocyte antigen variation is associated with adverse events of checkpoint inhibitors. Eur J Cancer 107:8–14. https://doi.org/10.1016/j.ejca.2018.11.009
Arnaiz-Villena A, Juarez I, Suarez-Trujillo F, López-Nares A, Vaquero C, Palacio-Gruber J, Martin-Villa JM (2021) HLA-G: function, polymorphisms and pathology. Int J Immunogenet 48:172–192. https://doi.org/10.1111/iji.12513
Boesen M, Svane IM, Engel AM, Rygaard J, Thomsen AR, Werdelin O (2000) CD8+ T cells are crucial for the ability of congenic normal mice to reject highly immunogenic sarcomas induced in nude mice with 3-methylcholanthrene. Clin Exp Immunol 121:210–215. https://doi.org/10.1046/j.1365-2249.2000.01292.x
Borst L, van der Burg SH, van Hall T (2020) The NKG2A-HLA-E axis as a novel checkpoint in the tumor microenvironment. Clin Cancer Res 26:5549–5556. https://doi.org/10.1158/1078-0432.CCR-19-2095
Byrne EH, Fisher DE (2017) Immune and molecular correlates in melanoma treated with immune checkpoint blockade. Cancer 123:2143–2153. https://doi.org/10.1002/cncr.30444
Cai L, Michelakos T, Yamada T, Fan S, Wang X, Schwab JH, Ferrone CR, Ferrone S (2018) Defective HLA class I antigen processing machinery in cancer. Cancer Immunol Immunother 67:999–1009. https://doi.org/10.1007/s00262-018-2131-2
Cai H, Feng Y, Fan P, Guo Y, Kuerban G, Chang C, Yao X, Peng Y, Wang R (2021) HPV16 E6-specific T cell response and HLA-A alleles are related to the prognosis of patients with cervical cancer. Infect Agent Cancer 16:61. https://doi.org/10.1186/s13027-021-00395-y
Chang H, Shin YW, Keam B, Kim M, Im SA, Lee ST (2020) HLA-B27 association of autoimmune encephalitis induced by PD-L1 inhibitor. Ann Clin Transl Neurol 7:2243–2250. https://doi.org/10.1002/acn3.51213
Chen XY, Yan WH, Lin A, Xu HH, Zhang JG, Wang XX (2008) The 14 bp deletion polymorphisms in HLA-G gene play an important role in the expression of soluble HLA-G in plasma. Tissue Antigens 72:335–341. https://doi.org/10.1111/j.1399-0039.2008.01107.x
Chowell D, Morris LGT, Grigg CM et al (2018) Patient HLA class I genotype influences cancer response to checkpoint blockade immunotherapy. Science 359:582–587. https://doi.org/10.1126/science.aao4572
Christopher MJ, Petti AA, Rettig MP et al (2018) Immune Escape of Relapsed AML Cells after Allogeneic Transplantation. N Engl J Med 379:2330–2341. https://doi.org/10.1056/NEJMoa1808777
Contini P, Ghio M, Poggi A, Filaci G, Indiveri F, Ferrone S, Puppo F (2003) Soluble HLA-A,-B,-C and -G molecules induce apoptosis in T and NK CD8+ cells and inhibit cytotoxic T cell activity through CD8 ligation. Eur J Immunol 33:125–134. https://doi.org/10.1002/immu.200390015
De Velasco G, Je Y, Bossé D, Awad MM, Ott PA, Moreira RB, Schutz F, Bellmunt J, Sonpavde GP, Hodi FS, Choueiri TK (2017) Comprehensive Meta-analysis of Key Immune-Related Adverse Events from CTLA-4 and PD-1/PD-L1 Inhibitors in Cancer Patients. Cancer Immunol Res 5:312–318. https://doi.org/10.1158/2326-6066.Cir-16-0237
Del Campo AB, Carretero J, Munoz JA, Zinchenko S, Ruiz-Cabello F, Gonzalez-Aseguinolaza G, Garrido F, Aptsiauri N (2014) Adenovirus expressing beta2-microglobulin recovers HLA class I expression and antitumor immunity by increasing T-cell recognition. Cancer Gene Ther 21:317–332. https://doi.org/10.1038/cgt.2014.32
Dendrou CA, Petersen J, Rossjohn J, Fugger L (2018) HLA variation and disease. Nat Rev Immunol 18:325–339. https://doi.org/10.1038/nri.2017.143
Dhouioui S, Laaribi AB, Boujelbene N, Jelassi R, Ben Salah H, Bellali H, Ouzari HI, Mezlini A, Zemni I, Chelbi H, Zidi I (2021) Association of HLA-G 3’UTR polymorphisms and haplotypes with colorectal cancer susceptibility and prognosis. Hum Immunol 83:39–46. https://doi.org/10.1016/j.humimm.2021.10.003
Dimou A, Grewe P, Sidney J, Sette A, Norman PJ, Doebele RC (2021) HLA Class I Binding of Mutant EGFR Peptides in NSCLC Is Associated With Improved Survival. J Thorac Oncol 16:104–112. https://doi.org/10.1016/j.jtho.2020.08.023
Djajadiningrat RS, Horenblas S, Heideman DA, Sanders J, de Jong J, Jordanova ES (2015) Classic and nonclassic HLA class I expression in penile cancer and relation to HPV status and clinical outcome. J Urol 193:1245–1251. https://doi.org/10.1016/j.juro.2014.11.057
Donadi EA, Castelli EC, Arnaiz-Villena A, Roger M, Rey D, Moreau P (2011) Implications of the polymorphism of HLA-G on its function, regulation, evolution and disease association. Cell Mol Life Sci 68:369–395. https://doi.org/10.1007/s00018-010-0580-7
Esfahani K, Miller WH Jr (2017) Reversal of Autoimmune Toxicity and Loss of Tumor Response by Interleukin-17 Blockade. N Engl J Med 376:1989–1991. https://doi.org/10.1056/NEJMc1703047
Ferns DM, Heeren AM, Samuels S, Bleeker MCG, de Gruijl TD, Kenter GG, Jordanova ES (2016) Classical and non-classical HLA class I aberrations in primary cervical squamous- and adenocarcinomas and paired lymph node metastases. J Immunother Cancer 4:78. https://doi.org/10.1186/s40425-016-0184-3
Gambacorta V, Beretta S, Ciccimarra M et al (2022) Integrated Multiomic Profiling Identifies the Epigenetic Regulator PRC2 as a Therapeutic Target to Counteract Leukemia Immune Escape and Relapse. Cancer Discov 12:1449–1461. https://doi.org/10.1158/2159-8290.Cd-21-0980
Garrido F (2019) HLA class-I expression and cancer immunotherapy. Adv Exp Med Biol 1151:79–90. https://doi.org/10.1007/978-3-030-17864-2_3
Goeppert B, Frauenschuh L, Zucknick M, Roessler S, Mehrabi A, Hafezi M, Stenzinger A, Warth A, Pathil A, Renner M, Schirmacher P, Weichert W (2015) Major histocompatibility complex class I expression impacts on patient survival and type and density of immune cells in biliary tract cancer. Br J Cancer 113:1343–1349. https://doi.org/10.1038/bjc.2015.337
Gooden M, Lampen M, Jordanova ES, Leffers N, Trimbos JB, van der Burg SH, Nijman H, van Hall T (2011) HLA-E expression by gynecological cancers restrains tumor-infiltrating CD8+ T lymphocytes. Proc Natl Acad Sci U S A 108:10656–10661
Hiraoka N, Ino Y, Hori S et al (2020) Expression of classical human leukocyte antigen class I antigens, HLA-E and HLA-G, is adversely prognostic in pancreatic cancer patients. Cancer Sci 111:3057–3070. https://doi.org/10.1111/cas.14514
Ho J, Schmidt D, Lowinus T et al (2022) Targeting MDM2 enhances antileukemia immunity after allogeneic transplantation via MHC-II and TRAIL-R1/2 upregulation. Blood 140:1167–1181. https://doi.org/10.1182/blood.2022016082
Imai D, Yoshizumi T, Okano S, Uchiyama H, Ikegami T, Harimoto N, Itoh S, Soejima Y, Aishima S, Oda Y, Maehara Y (2017) The prognostic impact of programmed cell death ligand 1 and human leukocyte antigen class I in pancreatic cancer. Cancer Med 6:1614–1626. https://doi.org/10.1002/cam4.1087
Imani R, Seyedmajidi M, Ghasemi N, Moslemi D, Shafaee S, Bijani A (2018) HLA-G expression is associated with an unfavorable prognosis of oral squamous cell carcinoma. Asian Pac J Cancer Prev 19:2527–2533
Ishigami S, Arigami T, Okumura H, Uchikado Y, Kita Y, Kurahara H, Maemura K, Kijima Y, Ishihara Y, Sasaki K, Uenosono Y, Natsugoe S (2015) Human leukocyte antigen (HLA)-E and HLA-F expression in gastric cancer. Anticancer Res 35:2279–2285
Jongsma MLM, Guarda G, Spaapen RM (2019) The regulatory network behind MHC class I expression. Mol Immunol 113:16–21. https://doi.org/10.1016/j.molimm.2017.12.005
Kennedy LB, Salama AKS (2020) A review of cancer immunotherapy toxicity. CA Cancer J Clin 70:86–104. https://doi.org/10.3322/caac.21596
King A, Allan DS, Bowen M, Powis SJ, Joseph S, Verma S, Hiby SE, McMichael AJ, Loke YW, Braud VM (2000) HLA-E is expressed on trophoblast and interacts with CD94/NKG2 receptors on decidual NK cells. Eur J Immunol 30:1623–1631. https://doi.org/10.1002/1521-4141(200006)30:6%3c1623::aid-immu1623%3e3.0.co;2-m
Köstlin N, Ostermeir AL, Spring B, Schwarz J, Marmé A, Walter CB, Poets CF, Gille C (2017) HLA-G promotes myeloid-derived suppressor cell accumulation and suppressive activity during human pregnancy through engagement of the receptor ILT4. Eur J Immunol 47:374–384. https://doi.org/10.1002/eji.201646564
Krijgsman D, Roelands J, Hendrickx W, Bedognetti D, Kuppen PJK (2020) HLA-G: a new immune checkpoint in cancer? Int J Mol Sci 21:4528. https://doi.org/10.3390/ijms21124528
Latorre D, Kallweit U, Armentani E et al (2018) T cells in patients with narcolepsy target self-antigens of hypocretin neurons. Nature 562:63–68. https://doi.org/10.1038/s41586-018-0540-1
Lee JH, Shklovskaya E, Lim SY et al (2020) Transcriptional downregulation of MHC class I and melanoma de- differentiation in resistance to PD-1 inhibition. Nat Commun 11:1897. https://doi.org/10.1038/s41467-020-15726-7
LeMaoult J, Krawice-Radanne I, Dausset J, Carosella ED (2004) HLA-G1-expressing antigen-presenting cells induce immunosuppressive CD4+ T cells. Proc Natl Acad Sci U S A 101:7064–7069. https://doi.org/10.1073/pnas.0401922101
Leo PJ, Madeleine MM, Wang S et al (2017) Defining the genetic susceptibility to cervical neoplasia-A genome-wide association study. PLoS Genet 13:e1006866. https://doi.org/10.1371/journal.pgen.1006866
Lin A, Yan WH (2021) HLA-G/ILTs targeted solid cancer immunotherapy: opportunities and challenges. Front Immunol 12:698677. https://doi.org/10.3389/fimmu.2021.698677
Maggs L, Sadagopan A, Moghaddam AS, Ferrone S (2021) HLA class I antigen processing machinery defects in antitumor immunity and immunotherapy. Trends Cancer 7:1089–1101. https://doi.org/10.1016/j.trecan.2021.07.006
Marty R, Kaabinejadian S, Rossell D, Slifker MJ, van de Haar J, Engin HB, de Prisco N, Ideker T, Hildebrand WH, Font-Burgada J, Carter H (2017) MHC-I Genotype Restricts the Oncogenic Mutational Landscape. Cell 171:1272-1283.e1215. https://doi.org/10.1016/j.cell.2017.09.050
Marty Pyke R, Thompson WK, Salem RM, Font-Burgada J, Zanetti M, Carter H (2018) Evolutionary Pressure against MHC Class II Binding Cancer Mutations. Cell 175:416-428.e413. https://doi.org/10.1016/j.cell.2018.08.048
Masuda T, Ito H, Hirata J, Sakaue S, Ueda Y, Kimura T, Takeuchi F, Murakami Y, Matsuda K, Matsuo K, Okada Y (2020) Fine Mapping of the Major Histocompatibility Complex Region and Association of the HLA-B*52:01 Allele With Cervical Cancer in Japanese Women. JAMA Netw Open 3:e2023248. https://doi.org/10.1001/jamanetworkopen.2020.23248
Mints M, Landin D, Nasman A, Mirzaie L, Ursu RG, Zupancic M, Marklund L, Dalianis T, Munck-Wikland E, Ramqvist T (2021) Tumour inflammation signature and expression of S100A12 and HLA class I improve survival in HPV-negative hypopharyngeal cancer. Sci Rep 11:1782. https://doi.org/10.1038/s41598-020-80226-z
Moretta A, Bottino C, Pende D, Tripodi G, Tambussi G, Viale O, Orengo A, Barbaresi M, Merli A, Ciccone E et al (1990) Identification of four subsets of human CD3-CD16+ natural killer (NK) cells by the expression of clonally distributed functional surface molecules: correlation between subset assignment of NK clones and ability to mediate specific alloantigen recognition. J Exp Med 172:1589–1598. https://doi.org/10.1084/jem.172.6.1589
Murdaca G, Calamaro P, Lantieri F, Pigozzi S, Mastracci L, Grillo F, Magnani O, Ceppa P, Puppo F, Fiocca R (2018) HLA-G expression in gastric carcinoma: clinicopathological correlations and prognostic impact. Virchows Arch 473:425–433. https://doi.org/10.1007/s00428-018-2379-0
Naji A, Menier C, Morandi F, Agaugué S, Maki G, Ferretti E, Bruel S, Pistoia V, Carosella ED, Rouas-Freiss N (2014) Binding of HLA-G to ITIM-bearing Ig-like transcript 2 receptor suppresses B cell responses. J Immunol 192:1536–1546. https://doi.org/10.4049/jimmunol.1300438
Osorio JC, Ni A, Chaft JE et al (2017) Antibody-mediated thyroid dysfunction during T-cell checkpoint blockade in patients with non-small-cell lung cancer. Ann Oncol 28:583–589. https://doi.org/10.1093/annonc/mdw640
Park HS, Cho U, Im SY, Yoo CY, Jung JH, Suh YJ, Choi HJ (2019a) Loss of human leukocyte antigen class I expression is associated with poor prognosis in patients with advanced breast cancer. J Pathol Transl Med 53:75–85. https://doi.org/10.4132/jptm.2018.10.11
Park Y, Koh J, Kwak Y, Ahn SH, Park DJ, Kim HH, Kim WH, Lee HS (2019b) Clinicopathologic significance of human leukocyte antigen class I expression in patients with stage II and III gastric cancer. Cancer Immunol Immunother 68:1779–1790. https://doi.org/10.1007/s00262-019-02410-z
Pathiraja V, Kuehlich JP, Campbell PD et al (2015) Proinsulin-specific, HLA-DQ8, and HLA-DQ8-transdimer-restricted CD4+ T cells infiltrate islets in type 1 diabetes. Diabetes 64:172–182. https://doi.org/10.2337/db14-0858
Postow MA, Sidlow R, Hellmann MD (2018) Immune-Related Adverse Events Associated with Immune Checkpoint Blockade. N Engl J Med 378:158–168. https://doi.org/10.1056/NEJMra1703481
Prinz JC (2017) Melanocytes: Target Cells of an HLA-C*06:02-Restricted Autoimmune Response in Psoriasis. J Invest Dermatol 137:2053–2058. https://doi.org/10.1016/j.jid.2017.05.023
Ritter C, Fan K, Paschen A, Reker Hardrup S, Ferrone S, Nghiem P, Ugurel S, Schrama D, Becker JC (2017) Epigenetic priming restores the HLA class-I antigen processing machinery expression in Merkel cell carcinoma. Sci Rep 7:2290. https://doi.org/10.1038/s41598-017-02608-0
Robinson J, Barker DJ, Georgiou X, Cooper MA, Flicek P, Marsh SGE (2020) IPD-IMGT/HLA Database. Nucleic Acids Res 48:D948-d955. https://doi.org/10.1093/nar/gkz950
Rousseau P, Le Discorde M, Mouillot G, Marcou C, Carosella ED, Moreau P (2003) The 14 bp deletion-insertion polymorphism in the 3’ UT region of the HLA-G gene influences HLA-G mRNA stability. Hum Immunol 64:1005–1010. https://doi.org/10.1016/j.humimm.2003.08.347
Schaafsma E, Fugle CM, Wang X, Cheng C (2021) Pan-cancer association of HLA gene expression with cancer prognosis and immunotherapy efficacy. Br J Cancer 125:422–432. https://doi.org/10.1038/s41416-021-01400-2
Schreiber RD, Old LJ, Smyth MJ (2011) Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science 331:1565–1570. https://doi.org/10.1126/science.1203486
Seliger B, Kloor M, Ferrone S (2017) HLA class II antigen-processing pathway in tumors: Molecular defects and clinical relevance. Oncoimmunology 6:e1171447. https://doi.org/10.1080/2162402x.2016.1171447
Shete S, Liu H, Wang J, Yu R, Sturgis EM, Li G, Dahlstrom KR, Liu Z, Amos CI, Wei Q (2020) A Genome-Wide Association Study Identifies Two Novel Susceptible Regions for Squamous Cell Carcinoma of the Head and Neck. Cancer Res 80:2451–2460. https://doi.org/10.1158/0008-5472.Can-19-2360
Shukla SA, Rooney MS, Rajasagi M et al (2015) Comprehensive analysis of cancer-associated somatic mutations in class I HLA genes. Nat Biotechnol 33:1152–1158. https://doi.org/10.1038/nbt.3344
Stokidis S, Fortis SP, Kogionou P, Anagnostou T, Perez SA, Baxevanis CN (2020) HLA class I allele expression and clinical outcome in de novo metastatic prostate cancer. Cancers (basel) 12:1623. https://doi.org/10.3390/cancers12061623
Tinker AV, Hirte HW, Provencher D, Butler M, Ritter H, Tu D, Azim HA Jr, Paralejas P, Grenier N, Hahn SA, Ramsahai J, Seymour L (2019) Dose-ranging and cohort-expansion study of monalizumab (IPH2201) in patients with advanced gynecologic malignancies: a trial of the Canadian cancer trials group (CCTG): IND221. Clin Cancer Res 25:6052–6060. https://doi.org/10.1158/1078-0432.CCR-19-0298
Toffalori C, Zito L, Gambacorta V et al (2019) Immune signature drives leukemia escape and relapse after hematopoietic cell transplantation. Nat Med 25:603–611. https://doi.org/10.1038/s41591-019-0400-z
Trowsdale J, Knight JC (2013) Major histocompatibility complex genomics and human disease. Annu Rev Genomics Hum Genet 14:301–323. https://doi.org/10.1146/annurev-genom-091212-153455
Vaquero-Yuste C, Juarez I, Molina-Alejandre M et al (2021) HLA-G 3’UTR polymorphisms are linked to susceptibility and survival in Spanish gastric adenocarcinoma patients. Front Immunol 12:698438. https://doi.org/10.3389/fimmu.2021.698438
von Boehmer H (1991) Positive and negative selection of the alpha beta T-cell repertoire in vivo. Curr Opin Immunol 3:210–215. https://doi.org/10.1016/0952-7915(91)90052-3
Wan R, Wang ZW, Li H, Peng XD, Liu GY, Ou JM, Cheng AQ (2017) Human leukocyte antigen-G inhibits the anti-tumor effect of natural killer cells via immunoglobulin-like transcript 2 in gastric cancer. Cell Physiol Biochem 44:1828–1841. https://doi.org/10.1159/000485819
Watkins TBK, Lim EL, Petkovic M et al (2020) Pervasive chromosomal instability and karyotype order in tumour evolution. Nature 587:126–132. https://doi.org/10.1038/s41586-020-2698-6
Watson NF, Ramage JM, Madjd Z, Spendlove I, Ellis IO, Scholefield JH, Durrant LG (2006) Immunosurveillance is active in colorectal cancer as downregulation but not complete loss of MHC class I expression correlates with a poor prognosis. Int J Cancer 118:6–10. https://doi.org/10.1002/ijc.21303
Wu Z, Liang J, Wang Z, Li A, Fan X, Jiang T (2020) HLA-E expression in diffuse glioma: relationship with clinicopathological features and patient survival. BMC Neurol 20:59. https://doi.org/10.1186/s12883-020-01640-4
Xu X, Zhou Y, Wei H (2020) Roles of HLA-G in the maternal-fetal immune microenvironment. Front Immunol 11:592010. https://doi.org/10.3389/fimmu.2020.592010
Yahia HB, Boujelbene N, Babay W et al (2020) Expression analysis of immune-regulatory molecules HLA-G, HLA-E and IDO in endometrial cancer. Hum Immunol 81:305–313. https://doi.org/10.1016/j.humimm.2020.03.008
Yano S, Ashida K, Sakamoto R et al (2020) Human leucocyte antigen DR15, a possible predictive marker for immune checkpoint inhibitor-induced secondary adrenal insufficiency. Eur J Cancer 130:198–203. https://doi.org/10.1016/j.ejca.2020.02.049
Yarmarkovich M, Farrel A, Sison A 3rd, di Marco M, Raman P, Parris JL, Monos D, Lee H, Stevanovic S, Maris JM (2020) Immunogenicity and Immune Silence in Human Cancer. Front Immunol 11:69. https://doi.org/10.3389/fimmu.2020.00069
Yazdi MT, van Riet S, van Schadewijk A, Fiocco M, van Hall T, Taube C, Hiemstra PS, van der Burg SH (2016) The positive prognostic effect of stromal CD8+ tumor-infiltrating T cells is restrained by the expression of HLA-E in non-small cell lung carcinoma. Oncotarget 7:3477–3488
Yie SM, Yang H, Ye SR, Li K, Dong DD, Lin XM (2007) Expression of human leucocyte antigen G (HLA-G) is associated with prognosis in non-small cell lung cancer. Lung Cancer 58:267–274. https://doi.org/10.1016/j.lungcan.2007.06.011
Zeestraten EC, Reimers MS, Saadatmand S, Goossens-Beumer IJ, Dekker JW, Liefers GJ, van den Elsen PJ, van de Velde CJ, Kuppen PJ (2014) Combined analysis of HLA class I, HLA-E and HLA-G predicts prognosis in colon cancer patients. Br J Cancer 110:459–468. https://doi.org/10.1038/bjc.2013.696
Zhang P, Guo X, Li J, Yu S, Wang L, Jiang G, Yang D, Wei Z, Zhang N, Liu J, Sun Y (2015a) Immunoglobulin-like transcript 4 promotes tumor progression and metastasis and up-regulates VEGF-C expression via ERK signaling pathway in non-small cell lung cancer. Oncotarget 6:13550–13563
Zhang X, Lv Z, Yu H, Wang F, Zhu J (2015b) The HLA-DQB1 gene polymorphisms associated with cervical cancer risk: A meta-analysis. Biomed Pharmacother 73:58–64. https://doi.org/10.1016/j.biopha.2015.06.002
Zhang RL, Zhang X, Dong SS, Hu B, Han QY, Zhang JG, Zhou WJ, Lin A, Yan WH (2017) Predictive value of different proportion of lesion HLA-G expression in colorectal cancer. Oncotarget 8:107441–107451
Zhang S, Kohli K, Black RG et al (2019) Systemic interferon-gamma increases MHC class I expression and T-cell infiltration in cold tumors: results of a phase 0 clinical trial. Cancer Immunol Res 7:1237–1243. https://doi.org/10.1158/2326-6066.CIR-18-0940
Zhong S, Malecek K, Johnson LA et al (2013) T-cell receptor affinity and avidity defines antitumor response and autoimmunity in T-cell immunotherapy. Proc Natl Acad Sci U S A 110:6973–6978. https://doi.org/10.1073/pnas.1221609110
Zhou F (2009) Molecular mechanisms of IFN-gamma to up-regulate MHC class I antigen processing and presentation. Int Rev Immunol 28:239–260. https://doi.org/10.1080/08830180902978120
Funding
Funding program: Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences (Construction and Application of Clinical Trial Institution Evaluation System 2021-I2M-1-045); Beijing Hope Run Special Fund of Cancer Foundation of China (LC2021B11).
Author information
Authors and Affiliations
Contributions
NJ and YY drafted the manuscript. DW, SW, YF, HM, PM and HH provided perspectives on HLA. MZ and YZ prepared the tables and figures. TY and NL supervised the study and reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Conflict of interests
Min Zhang is employed by Novogene Co., Ltd, Beijing, China. Yu Zhang is employed by Renke Beijing Biotechnology Co., Ltd, Beijing, China. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical approval
This review involved no human or animal subject. Ethics approval is exempted.
Consent to participate
Not applicable.
Consent to publish
NO human subjects were involved in this review.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jiang, N., Yu, Y., Wu, D. et al. HLA and tumour immunology: immune escape, immunotherapy and immune-related adverse events. J Cancer Res Clin Oncol 149, 737–747 (2023). https://doi.org/10.1007/s00432-022-04493-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04493-1