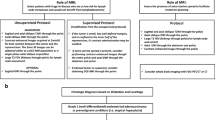
Abstract
Introduction
Cross-sectional imaging plays an integral role in the management of upper gastrointestinal (UGI) cancer, from initial diagnosis and staging to determining appropriate treatment strategies. Subjective imaging interpretation has known limitations. The field of radiomics has evolved to extract quantitative data from medical imaging and relate these to biological processes. The key concept behind radiomics is that the high-throughput analysis of quantitative imaging features can provide predictive or prognostic information, with the goal of providing individualised care.
Objective
Radiomic studies have shown promising utility in upper gastrointestinal oncology, highlighting a potential role in determining stage of disease and degree of tumour differentiation and predicting recurrence-free survival. This narrative review aims to provide an insight into the concepts underpinning radiomics, as well as its potential applications for guiding treatment and surgical decision-making in upper gastrointestinal malignancy.
Conclusion
Outcomes from studies to date have been promising; however, further standardisation and collaboration are required. Large prospective studies with external validation and evaluation of radiomic integration into clinical pathways are needed. Future research should now focus on translating the promising utility of radiomics into meaningful patient outcomes.


Similar content being viewed by others
Abbreviations
- AI:
-
Artificial intelligence
- ALBI:
-
Albumin-bilirubin score
- AUC:
-
Area under the receiver operating characteristic curve
- CT:
-
Computed tomography
- FDG:
-
Fludeoxyglucose
- FLRV:
-
Future liver remnant volume
- Gd-EOB-DTPA:
-
Gadolinium-ethoxybenzyl-diethylenetriamine
- GLCM:
-
Grey-level co-occurrence matrix
- GLDM:
-
Grey-level dependence matrix
- GLDZM:
-
Grey-level distance zone matrix
- GLRLM:
-
Grey-level run-length matrix
- GLSZM:
-
Grey-level size zone matrix
- GOJ:
-
Gastro-oesophageal junction
- HCC:
-
Hepatocellular carcinoma
- IBSI:
-
Imaging Biomarker Standardisation Initiative
- ICC:
-
Intra-class correlation coefficient
- ICG:
-
Indocyanine green
- LASSO:
-
Least absolute shrinkage and selection operator
- MELD:
-
Model for end-stage liver disease
- MRI:
-
Magnetic resonance imaging
- mRMR:
-
Minimum redundancy maximum relevance
- nCRT:
-
Neoadjuvant chemoradiotherapy
- NGLDM:
-
Neighbourhood grey-level dependence matrix
- NGTDM:
-
Neighbourhood grey-tone difference matrix
- pCR:
-
Complete pathological response
- PDAC:
-
Pancreatic ductal adenocarcinoma
- PET:
-
Positron emission tomography
- PHLF:
-
Post-operative liver failure
- PHM:
-
Pancreatic head malignancy
- ROI:
-
Region of interest
- SCC:
-
Squamous cell carcinoma
- SMA:
-
Superior mesenteric artery
- SMV:
-
Superior mesenteric vein
- TRG:
-
Tumour regression grade
- UGI:
-
Upper gastrointestinal
References
Stanzione A, Verde F, Romeo V, Boccadifuoco F, Mainenti PP, Maurea S (2021) Radiomics and machine learning applications in rectal cancer: current update and future perspectives. WJG 27(32):5306–5321
Lambin P, Rios-Velazquez E, Leijenaar R, Carvalho S, van Stiphout RGPM, Granton P et al (2012) Radiomics: extracting more information from medical images using advanced feature analysis. Eur J Cancer 48(4):441–446
Alderson PO, Summers RM (2020) The evolving status of radiomics. JNCI: J Natl Cancer Inst 112(9):869–70
Yip SSF, Aerts HJWL (2016) Applications and limitations of radiomics. Phys Med Biol 61(13):R150–R166
Wang Y, Jin ZY (2019) Radiomics approaches in gastric cancer: a frontier in clinical decision making. Chin Med J 132(16):1983–1989
Shur JD, Doran SJ, Kumar S, apDafydd D, Downey K, O’Connor JPB et al (2021) Radiomics in oncology: a practical guide. RadioGraphics 41(6):1717–32
Mayerhoefer ME, Materka A, Langs G, Häggström I, Szczypiński P, Gibbs P et al (2020) Introduction to radiomics. J Nucl Med 61(4):488–495
Rizzo S, Botta F, Raimondi S, Origgi D, Fanciullo C, Morganti AG et al (2018) Radiomics: the facts and the challenges of image analysis. Eur Radiol Exp 2(1):36
van Timmeren JE, Cester D, Tanadini-Lang S, Alkadhi H, Baessler B (2020) Radiomics in medical imaging—“how-to” guide and critical reflection. Insights Imaging 11(1):91
Shaikh FA, Kolowitz BJ, Awan O, Aerts HJ, von Reden A, Halabi S et al (2017) Technical challenges in the clinical application of radiomics. JCO Clin Cancer Inf 1:1–8
Mu W, Schabath MB, Gillies RJ (2022) Images are data: challenges and opportunities in the clinical translation of radiomics. Can Res 82(11):2066–2068
Avanzo M, Stancanello J, El Naqa I (2017) Beyond imaging: the promise of radiomics. Physica Med 38:122–139
Alderson PO (2020) The quest for generalizability in radiomics. Radiology: Artif Intell 2(3):e200068
van Griethuysen JJM, Fedorov A, Parmar C, Hosny A, Aucoin N, Narayan V et al (2017) Computational radiomics system to decode the radiographic phenotype. Can Res 77(21):e104–e107
Bibault JE, Xing L, Giraud P, El Ayachy R, Giraud N, Decazes P et al (2020) Radiomics: a primer for the radiation oncologist. Cancer/Radiothérapie 24(5):403–410
Bodalal Z, Trebeschi S, Nguyen-Kim TDL, Schats W, Beets-Tan R (2019) Radiogenomics: bridging imaging and genomics. Abdom Radiol 44(6):1960–1984
Vickers AJ, Elkin EB (2006) Decision curve analysis: a novel method for evaluating prediction models. Med Decis Making 26(6):565–574
Wu L, Wang C, Tan X, Cheng Z, Zhao K, Yan L et al (2018) Radiomics approach for preoperative identification of stages I−II and III−IV of esophageal cancer. Chin J Cancer Res 30(4):396–405
Kawahara D, Murakami Y, Tani S, Nagata Y (2021) A prediction model for degree of differentiation for resectable locally advanced esophageal squamous cell carcinoma based on CT images using radiomics and machine-learning. BJR 94(1124):20210525
Gao X, Ma T, Cui J, Zhang Y, Wang L, Li H et al (2021) A CT-based radiomics model for prediction of lymph node metastasis in early stage gastric cancer. Acad Radiol 28(6):e155–e164
Wang Y, Liu W, Yu Y, Liu JJ, Xue HD, Qi YF et al (2020) CT radiomics nomogram for the preoperative prediction of lymph node metastasis in gastric cancer. Eur Radiol. 30(2):976–86
Sun Z, Jiang Y, Chen C, Zheng H, Huang W, Xu B et al (2021) Radiomics signature based on computed tomography images for the preoperative prediction of lymph node metastasis at individual stations in gastric cancer: a multicenter study. Radiother Oncol 165:179–190
Dong D, Fang MJ, Tang L, Shan XH, Gao JB, Giganti F et al (2020) Deep learning radiomic nomogram can predict the number of lymph node metastasis in locally advanced gastric cancer: an international multicenter study. Ann Oncol 31(7):912–920
Zhao B, Zhu HT, Li XT, Shi YJ, Cao K, Sun YS (2021) Predicting lymph node metastasis using computed tomography radiomics analysis in patients with resectable esophageal squamous cell carcinoma. J Comput Assist Tomogr 45(2):323–329
Shen C, Liu Z, Wang Z, Guo J, Zhang H, Wang Y et al (2018) Building CT radiomics based nomogram for preoperative esophageal cancer patients lymph node metastasis prediction. Translational Oncology 11(3):815–824
Chen Y, Xi W, Yao W, Wang L, Xu Z, Wels M et al (2021) Dual-energy computed tomography-based radiomics to predict peritoneal metastasis in gastric cancer. Front Oncol 14(11):659981
Xue B, Jiang J, Chen L, Wu S, Zheng X, Zheng X et al (2021) Development and validation of a radiomics model based on 18F-FDG PET of primary gastric cancer for predicting peritoneal metastasis. Front Oncol 26(11):740111
Peng H, Xue T, Chen Q, Li M, Ge Y, Feng F (2022) Computed tomography-based radiomics nomogram for predicting the postoperative prognosis of esophageal squamous cell carcinoma: a multicenter study. Acad Radiol S1076-6332(22)00070-8
Tang S, Ou J, Wu YP, Li R, Chen TW, Zhang XM (2021) Contrast-enhanced CT radiomics features to predict recurrence of locally advanced oesophageal squamous cell cancer within 2 years after trimodal therapy: a case-control study. Medicine 100(27):e26557
Tang S, Ou J, Liu J, Wu YP, Wu CQ, Chen TW et al (2021) Application of contrast-enhanced CT radiomics in prediction of early recurrence of locally advanced oesophageal squamous cell carcinoma after trimodal therapy. Cancer Imaging 21(1):38
Kong J, Zhu S, Shi G, Liu Z, Zhang J, Ren J (2021) Prediction of locoregional recurrence-free survival of oesophageal squamous cell carcinoma after chemoradiotherapy based on an enhanced CT-based radiomics model. Front Oncol 24(11):739933
Luo HS, Chen YY, Huang WZ, Wu SX, Huang SF, Xu HY et al (2021) Development and validation of a radiomics-based model to predict local progression-free survival after chemo-radiotherapy in patients with esophageal squamous cell cancer. Radiat Oncol 16(1):201
Deantonio L, Garo ML, Paone G, Valli MC, Cappio S, La Regina D et al (2022) 18F-FDG PET radiomics as predictor of treatment response in oesophageal cancer: a systematic review and meta-analysis. Front Oncol 15(12):861638
van Rossum PSN, Fried DV, Zhang L, Hofstetter WL, van Vulpen M, Meijer GJ et al (2016) The incremental value of subjective and quantitative assessment of 18 F-FDG PET for the prediction of pathologic complete response to preoperative chemoradiotherapy in esophageal cancer. J Nucl Med 57(5):691–700
Yip SSF, Coroller TP, Sanford NN, Mamon H, Aerts HJWL, Berbeco RI (2016) Relationship between the temporal changes in positron-emission-tomography-imaging-based textural features and pathologic response and survival in esophageal cancer patients. Front Oncol 6:72
Eyck BM, van der Wilk BJ, Lagarde SM, Wijnhoven BPL, Valkema R, Spaander MCW, Nuyttens JJME, van der Gaast A, van Lanschot JJB (2018) Neoadjuvant chemoradiotherapy for resectable oesophageal cancer. Best Pract Res Clin Gastroenterol 36–37:37–44
Beukinga RJ, Hulshoff JB, van Dijk LV, Muijs CT, Burgerhof JGM, Kats-Ugurlu G et al (2017) Predicting response to neoadjuvant chemoradiotherapy in esophageal cancer with textural features derived from pretreatment 18 F-FDG PET/CT imaging. J Nucl Med 58(5):723–729
Hirata A, Hayano K, Ohira G, Imanishi S, Hanaoka T, Murakami K et al (2020) Volumetric histogram analysis of apparent diffusion coefficient for predicting pathological complete response and survival in esophageal cancer patients treated with chemoradiotherapy. Am J Surg 219(6):1024–1029
Yang Z, He B, Zhuang X, Gao X, Wang D, Li M et al (2019) CT-based radiomic signatures for prediction of pathologic complete response in esophageal squamous cell carcinoma after neoadjuvant chemoradiotherapy. J Radiat Res 60(4):538–545
Rishi A, Zhang GG, Yuan Z, Sim AJ, Song EY, Moros EG et al (2021) Pretreatment CT and 18 F-FDG PET-based radiomic model predicting pathological complete response and loco-regional control following neoadjuvant chemoradiation in oesophageal cancer. J Med Imag Rad Onc 65(1):102–111
Hu Y, Xie C, Yang H, Ho JWK, Wen J, Han L et al (2020) Assessment of intratumoral and peritumoral computed tomography radiomics for predicting pathological complete response to neoadjuvant chemoradiation in patients with esophageal squamous cell carcinoma. JAMA Netw Open 3(9):e2015927
Murakami Y, Kawahara D, Tani S, Kubo K, Katsuta T, Imano N et al (2021) Predicting the local response of esophageal squamous cell carcinoma to neoadjuvant chemoradiotherapy by radiomics with a machine learning method using 18F-FDG PET images. Diagnostics 11(6):1049
Zhu WS, Shi SY, Yang ZH, Song C, Shen J (2020) Radiomics model based on preoperative gadoxetic acid-enhanced MRI for predicting liver failure. WJG 26(11):1208–1220
Chen Y, Liu Z, Mo Y, Li B, Zhou Q, Peng S et al (2021) Prediction of post-hepatectomy liver failure in patients with hepatocellular carcinoma based on radiomics using Gd-EOB-DTPA-enhanced MRI: the liver failure model. Front Oncol 10(11):605296
Søreide JA, Deshpande R (2021) Post hepatectomy liver failure (PHLF) – recent advances in prevention and clinical management. Eur J Surg Oncol 47(2):216–224
Cai W, He B, Hu M, Zhang W, Xiao D, Yu H et al (2019) A radiomics-based nomogram for the preoperative prediction of posthepatectomy liver failure in patients with hepatocellular carcinoma. Surg Oncol 28:78–85
Xiang F, Liang X, Yang L, Liu X, Yan S (2021) CT radiomics nomogram for the preoperative prediction of severe post-hepatectomy liver failure in patients with huge (≥ 10 cm) hepatocellular carcinoma. World J Surg Onc 19(1):344
Hanafy AS (2021) Prediction and prevention of post-hepatectomy liver failure: where do we stand? J Clin Transl Hepatol 000(000):000–000
Versteijne E, Vogel JA, Besselink MG, Busch ORC, Wilmink JW, Daams JG et al (2018) Meta-analysis comparing upfront surgery with neoadjuvant treatment in patients with resectable or borderline resectable pancreatic cancer. Br J Surg 105(8):946–958
Maeda S, Moore AM, Yohanathan L, Hata T, Truty MJ, Smoot RL et al (2020) Impact of resection margin status on survival in pancreatic cancer patients after neoadjuvant treatment and pancreatoduodenectomy. Surgery 167(5):803–811
Fukuda Y, Yamada D, Eguchi H, Hata T, Iwagami Y, Noda T et al (2017) CT density in the pancreas is a promising imaging predictor for pancreatic ductal adenocarcinoma. Ann Surg Oncol 24(9):2762–2769
Weyhe D, Obonyo D, Uslar VN, Stricker I, Tannapfel A (2021) Predictive factors for long-term survival after surgery for pancreatic ductal adenocarcinoma: making a case for standardized reporting of the resection margin using certified cancer center data. Wellner U, editor. PLoS ONE 16(3):e0248633
Ocaña J, Sanjuanbenito A, García A, Molina JM, Lisa E, Mendía E et al (2020) Relevance of positive resection margins in ductal pancreatic adenocarcinoma and prognostic factors. Cirugía Española (English Edition) 98(2):85–91
Menon KV, Gomez D, Smith AM, Anthoney A, Verbeke CS (2009) Impact of margin status on survival following pancreatoduodenectomy for cancer: the Leeds Pathology Protocol (LEEPP). HPB 11(1):18–24
Liu KL, Wu T, Chen PT, Tsai YM, Roth H, Wu MS et al (2020) Deep learning to distinguish pancreatic cancer tissue from non-cancerous pancreatic tissue: a retrospective study with cross-racial external validation. Lancet Digital Health 2(6):e303–e313
Cassinotto C, Dohan A, Zogopoulos G, Chiche L, Laurent C, Sa-Cunha A et al (2017) Pancreatic adenocarcinoma: a simple CT score for predicting margin-positive resection in patients with resectable disease. Eur J Radiol 95:33–38
Isaji S, Mizuno S, Windsor JA, Bassi C, Fernández-del Castillo C, Hackert T et al (2018) International consensus on definition and criteria of borderline resectable pancreatic ductal adenocarcinoma 2017. Pancreatology 18(1):2–11
Kobi M, Veillette G, Narurkar R, Sadowsky D, Paroder V, Shilagani C et al (2020) Imaging and management of pancreatic cancer. Sem Ultrasound CT MRI 41(2):139–151
Lopez NE (2014) Borderline resectable pancreatic cancer: definitions and management. WJG 20(31):10740
Bian Y, Jiang H, Ma C, Cao K, Fang X, Li J et al (2020) Performance of CT-based radiomics in diagnosis of superior mesenteric vein resection margin in patients with pancreatic head cancer. Abdom Radiol 45(3):759–773
Rigiroli F, Hoye J, Lerebours R, Lafata KJ, Li C, Meyer M et al (2021) CT radiomic features of superior mesenteric artery involvement in pancreatic ductal adenocarcinoma: a pilot study. Radiology 7:210699
Lin Z, Tang B, Cai J, Wang X, Li C, Tian X, Yang Y, Wang X (2021) Preoperative prediction of clinically relevant postoperative pancreatic fistula after pancreaticoduodenectomy. Eur J Radiol 139:109693
Zhang W, Cai W, He B, Xiang N, Fang C, Jia F (2018) A radiomics-based formula for the preoperative prediction of postoperative pancreatic fistula in patients with pancreaticoduodenectomy. Cancer Manag Res 28(10):6469–6478
Skawran SM, Kambakamba P, Baessler B, von Spiczak J, Kupka M, Müller PC, Moeckli B, Linecker M, Petrowsky H, Reiner CS (2021) Can magnetic resonance imaging radiomics of the pancreas predict postoperative pancreatic fistula? Eur J Radiol 140:109733
Lee CH, Yoon HJ (2017) Medical big data: promise and challenges. Kidney Res Clin Pract 36(1):3–11
de la Pinta C (2021) Radiomics in pancreatic cancer for oncologist: present and future. Hepatobiliary Pancreat Dis Int 21(4):356–361
Chetan MR, Gleeson FV (2021) Radiomics in predicting treatment response in non-small-cell lung cancer: current status, challenges and future perspectives. Eur Radiol 31(2):1049–1058
Park JE, Kim D, Kim HS, Park SY, Kim JY, Cho SJ, Shin JH, Kim JH (2020) Quality of science and reporting of radiomics in oncologic studies: room for improvement according to radiomics quality score and TRIPOD statement. Eur Radiol 30(1):523–536
Lambin P. Radiomics quality score - RQS. Available from: https://www.radiomics.world (Accessed: May 2023)
Fanciullo C, Gitto S, Carlicchi E, Albano D, Messina C, Sconfienza LM (2022) Radiomics of musculoskeletal sarcomas: a narrative review. J Imaging 8(2):45
Chen B, Yang L, Zhang R, Luo W, Li W (2020) Radiomics: an overview in lung cancer management—a narrative review. Ann Transl Med 8(18):1191–1191
Author information
Authors and Affiliations
Contributions
SK and RB provided the concept for the paper. JD wrote the main manuscript, prepared tables and figures. All authors reviewed and critically revised the manuscript before final submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key points
• Radiomic studies to date have shown promising utility in upper gastrointestinal oncology.
• The powerful predictive capacity of radiomics has potential to guide surgical decision-making.
• Further prospective research is required to integrate radiomic modelling into clinical pathways in order to impact patient outcomes.
Sacheen Kumar and Ricky H. Bhogal are joint senior authors.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Doyle, J.P., Patel, P.H., Petrou, N. et al. Radiomic applications in upper gastrointestinal cancer surgery. Langenbecks Arch Surg 408, 226 (2023). https://doi.org/10.1007/s00423-023-02951-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00423-023-02951-z