Abstract
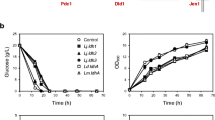
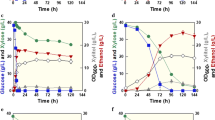
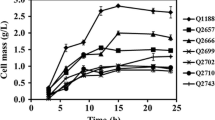
Saccharomyces cerevisiae is the workhorse of fermentation industry. Upon engineering for D-lactate production by a series of gene deletions, this yeast had deficiencies in cell growth and D-lactate production at high substrate concentrations. Complex nutrients or high cell density were thus required to support growth and D-lactate production with a potential to increase medium and process cost of industrial-scale D-lactate production. As an alternative microbial biocatalyst, a Crabtree-negative and thermotolerant yeast Kluyveromyces marxianus was engineered in this study to produce high titer and yield of D-lactate at a lower pH without growth defects. Only pyruvate decarboxylase 1 (PDC1) gene was replaced by a codon-optimized bacterial D-lactate dehydrogenase (ldhA). Ethanol, glycerol, or acetic acid was not produced by the resulting strain, KMΔpdc1::ldhA. Aeration rate at 1.5 vvm and culture pH 5.0 at 30 °C provided the highest D-lactate titer of 42.97 ± 0.48 g/L from glucose. Yield and productivity of D-lactate, and glucose-consumption rate were 0.85 ± 0.01 g/g, 0.90 ± 0.01 g/(L·h), and 1.06 ± 0.00 g/(L·h), respectively. Surprisingly, D-lactate titer, productivity, and glucose-consumption rate of 52.29 ± 0.68 g/L, 1.38 ± 0.05 g/(L·h), and 1.22 ± 0.00 g/(L·h), respectively, were higher at 42 °C compared to 30 °C. Sugarcane molasses, a low-value carbon, led to the highest D-lactate titer and yield of 66.26 ± 0.81 g/L and 0.91 ± 0.01 g/g, respectively, in a medium without additional nutrients. This study is a pioneer work of engineering K. marxianus to produce D-lactate at the yield approaching theoretical maximum using simple batch process. Our results support the potential of an engineered K. marxianus for D-lactate production on an industrial scale.
Key points
• K. marxianus was engineered by deleting PDC1 and expressing codon-optimized D-ldhA.
• The strain allowed high D-lactate titer and yield under pH ranging from 3.5 to 5.0.
• The strain produced 66 g/L D-lactate at 30 °C from molasses without any additional nutrients.





Similar content being viewed by others
Data availability
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
References
Awasthi D, Wang L, Rhee MS, Wang Q, Chauliac D, Ingram LO, Shanmugam KT (2018) Metabolic engineering of Bacillus subtilis for production of D-lactic acid. Biotechnol Bioengineer 115:453–463. https://doi.org/10.1002/bit.2647
Baek SH, Kwon E, Kim YH, Hahn JS (2016) Metabolic engineering and adaptive evolution for efficient production of D-lactic acid in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 100(6):2737–2748. https://doi.org/10.1007/s00253-015-7174-0
Choo JH, Han C, Lee DW, Sim GH, Moon HY, Kim JY, Song JY, Kang HA (2018) Molecular and functional characterization of two pyruvate decarboxylase genes, PDC1 and PDC5, in the thermotolerant yeast Kluyveromyces marxianus. Appl Microbiol Biotechnol 102(8):3723–3737. https://doi.org/10.1007/s00253-018-8862-3
Concordet JP, Haeussler M (2018) CRISPOR: intuitive guide selection for CRISPR/Cas9 genome editing experiments and screens. Nucleic Acids Res 46(W1):W242–W245. https://doi.org/10.1093/nar/gky354
Fu X, Wang Y, Wang J, Garza E, Manow R, Zhou S (2017) Semi-industrial scale (30 m3) fed-batch fermentation for the production of D-lactate by Escherichia coli strain HBUT-D15. J Ind Microbiol Biotechnol 44(2):221–228. https://doi.org/10.1007/s10295-016-1877-9
Gambacorta FV, Dietrich J, Yan Q, Pfleger BF (2020) Rewiring yeast metabolism to synthesize products beyond ethanol. Curr Opin Chem Biol 59:182–192. https://doi.org/10.1016/j.cbpa.2020.08.005
Gao C, Yang X, Wang H, Rivero CP, Li C, Cui Z, Qi Q, Lin CSK (2016) Robust succinic acid production from crude glycerol using engineered Yarrowia lipolytica. Biotechnol Biofuels 9:1–11. https://doi.org/10.1186/s13068-016-0597-8
Hama S, Mizuno S, Kihara M, Tanaka T, Ogino C, Noda H, Kondo A (2015) Production of D-lactic acid from hardwood pulp by mechanical milling followed by simultaneous saccharification and fermentation using metabolically engineered Lactobacillus plantarum. Bioresour Technol 187:167–172. https://doi.org/10.1016/j.biortech.2015.03.106
Ha-Tran DM, Nguyen TTM, Huang CC (2020) Kluyveromyces marxianus: current state of omics studies, strain improvement strategy and potential industrial implementation. Fermentation 6(124):1–19. https://doi.org/10.3390/fermentation6040124
Ilmén M, Koivuranta K, Ruohonen L, Rajgarhia V, Suominen P, Penttilä M (2013) Production of L-lactic acid by the yeast Candida sonorensis expressing heterologous bacterial and fungal lactate dehydrogenases. Microb Cell Factories 12(53):1–15. https://doi.org/10.1186/1475-2859-12-53
In S, Khunnonkwao P, Wong N, Phosiran C, Jantama SS, Jantama K (2020) Combining metabolic engineering and evolutionary adaptation in Klebsiella oxytoca KMS004 to significant improve optically pure D-(-)-lactic acid yield and specific productivity in low nutrient medium. Biotechnol Prod Proc Eng 104(22):9565–9579. https://doi.org/10.1007/s00253-020-10933-0
Ishida N, Suzuki T, Tokuhiro K, Nagamori E, Onishi T, Saitoh S, Kitamoto K, Takahashi H (2006) D-lactic acid production by metabolically engineered Saccharomyces cerevisiae. J Biosci Bioeng 101(2):172–177. https://doi.org/10.1263/jbb.101.172
Kong X, Zhang B, Hua Y, Zhu Y, Li W, Wang D, Hong J (2019) Efficient L-lactic acid production from corncob residue using metabolically engineered thermo-tolerant yeast. Bioresour Technol 273:220–230. https://doi.org/10.1016/j.biortech.2018.11.018
Lee JW, In JH, Park JB, Shin J, Park JH, Sung BH, Sohn JH, Seo JH, Park JB, Kim SR, Kweon DH (2017) Co-expression of two heterologous lactate dehydrogenases genes in Kluyveromyces marxianus for L-lactic acid production. J Biotechnol 241:81–86. https://doi.org/10.1016/j.jbiotec.2016.11.015
Li C, Tao F, Xu P (2016) Carbon flux trapping: highly efficient production of polymer-grade d-lactic acid with a thermophilic d-lactate dehydrogenase. ChemBioChem 17:1482–1578. https://doi.org/10.1002/cbic.201600288
Liu Y, Gao W, Zhao Z, Wang J, Garza E, Manow R, Zhou S (2014) Pilot scale demonstration of d-lactic acid fermentation facilitated by Ca(OH)2 using a metabolically engineered Escherichia coli. Bioresour Technol 169:559–565. https://doi.org/10.1016/j.biortech.2014.06.056
Lo SC, Yang CY, Mathew DC, Huang CC (2021) Growth and autolysis of the kefir yeast Kluyveromyces marxianus in lactate culture. Sci Rep 11:1–11. https://doi.org/10.1038/s41598-021-94101-y
Merico A, Galafassi S, Piškur J, Compagno C (2009) The oxygen level determines the fermentation pattern in Kluyveromyces lactis. FEMS Yeast Res 9:749–756. https://doi.org/10.1111/j.1567-1264.2009.00528.x
Narendranath NV, Thomas KC, Ingledew WM (2018) Acetic acid and lactic acid inhibition of growth of Saccharomyces cerevisiae by different mechanisms. J Am Soc Brew Chem 59(4):187–194. https://doi.org/10.1094/ASBCJ-59-0187
Okano K, Yoshida S, Yamada R, Tanaka T, Ogino C, Fukuda H, Kondo A (2009) Improved production of homo-D-lactic acid via xylose fermentation by introduction of xylose assimilation genes and redirection of the phosphoketolase pathway to the pentose phosphate pathway in L-lactate dehydrogenase gene-deficient Lactobacillus plantarum. Appl Enviro Microbiol 75:7858–7861. https://doi.org/10.1128/AEM.01692-09
Peetermans A, Foulquié-Moreno MR, Thevelein JM (2021) Mechanisms underlying lactic acid tolerance and its influence on lactic acid production in Saccharomyces cerevisiae. Microbial Cell 8(6):111–130. https://doi.org/10.15698/mic2021.06.751
Raab AM, Gebhardt G, Bolotina N, Weuster-Botz D, Lang C (2010) Metabolic engineering of Saccharomyces cerevisiae for biotechnological production of succinic acid. Metab Eng 12(6):518–525. https://doi.org/10.1016/j.ymben.2010.08.005
Rajkumar AS, Varela JA, Juergens H, Daran JMG, Morrissey JP (2019) Biological parts for Kluyveromyces marxianus synthetic biology. Front Bioeng Biotechnol 7(97):1–15. https://doi.org/10.3389/fbioe.2019.00097
Rugthaworn P, Murata Y, Machida M, Apiwatanapiwat W, Hirooka A, Thanapase W, Dangjarean H, Ushiwaka S, Morimitsu K, Kosugi A, Arai T, Vaithanomsat P (2014) Growth inhibition of thermotolerant yeast, Kluyveromyces marxianus, in hydrolysates from cassava pulp. Appl Biochem Biotechnol 173:1–15. https://doi.org/10.1007/s12010-014-0906.2
Sakihama Y, Hidese R, Hasunuma T, Kondo A (2019) Increased flux in acetyl-CoA synthetic pathway and TCA cycle of Kluyveromyces marxianus under respiratory conditions. Sci Rep 9(5319):1–10. https://doi.org/10.1038/s41598-019-41863-1
Sangproo M, Polyiam P, Jantama SS, Kanchanatawee S, Jantama K (2012) Metabolic engineering of Klebsiella oxytoca M5a1 to produce optically pure D-lactate in mineral salts medium. Bioresour Technol 119:191–198. https://doi.org/10.1016/j.biortech.2012.05.114
Sauer M, Porro D, Mattanovich D, Branduardi P (2010) 16 years research on lactic acid production with yeast - ready for the market? Biotechnol Genet Eng Rev 27:229–256. https://doi.org/10.1080/02648725.2021.10648152
Sornlek W, Sae-Tang K, Watcharawipas A, Wongwisansri S, Tanapongpipat S, Eurwilaichtr L, Champreda V, Runguphan W, Schaap PJ, Santos VAPMd (2022) D-lactic acid production from sugarcane bagasse by genetically engineered Saccharomyces cerevisiae. J Fungi 8(816):1–14. https://doi.org/10.3390/jof8080816
Wang Q, Ingram LO, Shanmugam KT (2011) Evolution of D-lactate dehydrogenase activity from glycerol dehydrogenase and its utility for D-lactate production from lignocellulose. Proc Natl Acad Sci USA 108(47):18920–18925. https://doi.org/10.1073/pnas.1111085108
Wang Y, Tian T, Zhao J, Wang J, Yan T, Xu L, Liu Z, Garza E, Iverson A, Manow R, Finan C, Zhou S (2012) Homofermentative production of D-lactic acid from sucrose by a metabolically engineered Escherichia coli. Biotechnol Lett 34:2069–2075. https://doi.org/10.1007/s10529-012-1003-7
Watcharawipas A, Sae-tang K, Sansatchanon K, Sudying P, Boonchoo K, Tanapongpipat S, Kocharin K, Runguphan W (2021) Systematic engineering of Saccharomyces cerevisiae for D-lactic acid production with near theoretical yield. FEMS Yeast Res 21(4):1–15. https://doi.org/10.1093/femsyr/foab024
Wong N, Jantama K (2022) Engineering Escherichia coli for a high yield of 1,3-propanediol near the theoretical maximum through chromosomal integration and gene deletion. Appl Microbiol Biotechnol 106(8):2937–2951. https://doi.org/10.1007/s00253-022-11898-y
Yamada R, Wakita K, Mitsui R, Ogino H (2017) Enhanced D-lactic acid production by recombinant Saccharomyces cerevisiae following optimization of the global metabolic pathway. Biotechnol Bioeng 114(9):2075–2084. https://doi.org/10.1002/bit.26330
Yang C, Hu S, Zhu S, Wang D, Gao X, Hong J (2015) Characterizing yeast promoters used in Kluyveromyces marxianus. World J Microbiol Biotechnol 31:1641–1646. https://doi.org/10.1007/s11274-015-1899-x
Zhou S, Causey TB, Hasona A, Shanmugam KT, Ingram LO (2003) Production of optically pure d-lactic acid in mineral salts medium by metabolically engineered Escherichia coli W3110. Appl Environ Microbiol 69(1):399–407. https://doi.org/10.1128/AEM.69.1.399-407.2003
Acknowledgements
The authors thank Mitr Phol Sugar Corporation, Ltd. (Thailand) for the provision of sugarcane molasses used in this study.
Funding
This work was financially supported by (i) Suranaree University of Technology (SUT), (ii) Thailand Science Research and Innovation (TSRI), and (iii) National Science Research and Innovation Fund (NSRF) (project code 90464) under the full-time researcher fellowship (Grant No.61/10/2562).
Author information
Authors and Affiliations
Contributions
KJ conceived, designed, and suggested methodology and research plan, supervised, and performed project administration and funding acquisition. CG investigated experiments, provided methodology, and wrote an original draft of the manuscript. PK helped investigating experiments. KJ also analyzed and validated data and edited, reviewed, and finalized the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gosalawit, C., Khunnonkwao, P. & Jantama, K. Genome engineering of Kluyveromyces marxianus for high D-( −)-lactic acid production under low pH conditions. Appl Microbiol Biotechnol 107, 5095–5105 (2023). https://doi.org/10.1007/s00253-023-12658-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-023-12658-2