Abstract
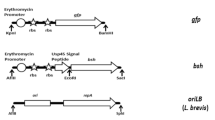
The genetic engineering of bacteria for food applications has biosafety requirements, including the use of non-antibiotic selectable markers. These can be gene-encoding bacteriocin immunity proteins, such as nisI and pedB, which require the use of promoters to ensure optimal expression. Our aim was to search for promoters for the expression of pediocin (pedB) and nisin (nisI) immunity genes, which could allow the selection of a wide variety of transformed lactic acid bacteria (LAB) and bifidobacteria strains. Eight promoters from LAB or bifidobacteria were initially studied using evoglow-Pp1 as the reporter gene in Lactococcus lactis NZ9000, resulting in the selection of P32, P3N, PTuR and PEF-P, which exhibited a strong constitutive expression. These promoters were further tested for the expression of the food-grade selectable markers pedB and nisI in agar diffusion assays with pediocin and nisin, respectively. The results obtained demonstrated that both the PTuR and PEF-P promoters allowed a good level of expression of nisI and pedB in the LAB and bifidobacteria strains tested. A suitable concentration of nisin or pediocin could be established for the selection of the strains transformed with vectors harbouring the combination of the selected promoters and markers nisI and pedB, and this was successfully applied to different strains of LAB and bifidobacteria. Therefore, PTuR and PEF-P promoters are excellent candidates for the expression of nisI and/or pedB as selectable markers in LAB and bifidobacteria, and they are suitable for use in food grade vectors to allow the selection of genetically engineered strains.
Key points
• Food-grade vectors require non-antibiotic selectable markers such as pedB and nisI.
• Eight promoters from LAB or bifidobacteria were initially tested in L. lactis NZ9000.
• PTuR and PEF-P efficiently drove the expression of pedB and nisI in LAB and bifidobacteria.



Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Allison GE, Klaenhammer TR (1996) Functional analysis of the gene encoding immunity to lactacin F, lafI, and its use as a Lactobacillus-specific, food-grade genetic marker. Appl Environ Microbiol 62:4450–4460. https://doi.org/10.1128/aem.62.12.4450-4460.1996
Arqués JL, Rodríguez JM, Gasson MJ, Horn N (2008) Immunity gene pedB enhances production of pediocin PA-1 in naturally resistant Lactococcus lactis strains. J Dairy Sci 91:2591–2594. https://doi.org/10.3168/jds.2007-0759
Capurso L (2019) Thirty years of Lactobacillus rhamnosus GG. J Clinl Gastroenterol 53:S1–S41. https://doi.org/10.1097/MCG.0000000000001170
de Andrés J, Jiménez E, Chico-Calero I, Fresno M, Fernández L, Rodríguez JM (2018) Physiological translocation of lactic acid bacteria during pregnancy contributes to the composition of the milk microbiota in mice. Nutrients 10:14. https://doi.org/10.3390/nu10010014
de Ruyter PG, Kuipers OP, de Vos WM (1996) Controlled gene expression systems for Lactococcus lactis with the food-grade inducer nisin. Appl Environ Microbiol 62:3662–3667. https://doi.org/10.1128/aem.62.10.3662-3667.1996
Fijan S (2014) Microorganisms with claimed probiotic properties: an overview of recent literature. Int J Environ Res Public Health 11:4745–4767. https://doi.org/10.3390/ijerph110504745
Garde S, Tomillo J, Gaya P, Medina M, Nuñez M (2002) Proteolysis in hispanico cheese manufactured using a mesophilic starter, a thermophilic starter, and a bacteriocin-producing Lactococcus lactis subsp. lactis INIA 415 adjunct culture. J Agric Food Chem 50:3479–3485. https://doi.org/10.1021/jf011291d
Gaya P, Peirotén A, Landete JM (2020) Expression of a β-glucosidase in bacteria with biotechnological interest confers them the ability to deglycosylate lignans and flavonoids in vegetal foods. Appl Microbiol Biotechnol 104:4903–4913. https://doi.org/10.1007/s00253-020-10588-x
Gosalbes MJ, Esteban CD, Galán JL, Pérez-Martinez G (2000) Integrative food-grade expression system based on the lactose regulon of Lactobacillus casei. Appl Environ Microbiol 66:4822–4828. https://doi.org/10.1128/aem.66.11.4822-4828.2000
Israr B, Kim J, Anam S, Anjum FR (2018) Lactic acid bacteria as vectors: a novel approach for mucosal vaccine delivery. J Clin Cell Immunol 9:548. https://doi.org/10.4172/2155-9899.1000548
Joosten HM, Nuñez M, Devreese B, van Beeumen J, Marugg JD (1996) Purification and characterization of enterocin 4, a bacteriocin produced by Enterococcus faecalis INIA 4. Appl Environ Microbiol 62:4220–4223. https://doi.org/10.1128/aem.62.11.4220-4223.1996
Kleerebezem M, Boekhorst J, van Kranenburg R, Molenaar D, Kuipers OP, Leer R, Tarchini R, Peters SA, Sandbrink HM, Fiers MW, Stiekema W, Lankhorst RM, Bron PA, Hoffer SM, Groot MN, Kerkhoven R, de Vries M, Ursing B, de Vos WM, Siezen RJ (2003) Complete genome sequence of Lactobacillus plantarum WCFS1. Proc Natl Acad Sci USA 100:1990–1995. https://doi.org/10.1073/pnas.0337704100
Koivula T, Sibakov M, Palva I (1991) Isolation and characterization of Lactococcus lactis subsp. lactis promoters. Appl Environ Microbiol 57:333–340. https://doi.org/10.1128/aem.57.2.333-340.1991
Kuipers OP, Beerthuyzen MM, de Ruyter PG, Luesink EJ, de Vos WM (1995) Autoregulation of nisin biosynthesis in Lactococcus lactis by signal transduction. J Biol Chem 270:27299–27304. https://doi.org/10.1074/jbc.270.45.27299
Kuipers OP, de Ruyter PGGA, Kleerebeze M, de Vos WM (1998) Quorum sensing-controlled gene expression in lactic acid bacteria. J Biotech 64:15–21. https://doi.org/10.1016/S0168-1656(98)00100-X
Landete JM (2017) A review of food-grade vectors in lactic acid bacteria: from the laboratory to their application. Crit Rev Biotechnol 37:296–308. https://doi.org/10.3109/07388551.2016.1144044
Landete JM, Arqués JL, Peirotén A, Langa S, Medina M (2014) An improved method for the electrotransformation of lactic acid bacteria: a comparative survey. J Microbiol Meth 105:130–133. https://doi.org/10.1016/j.mimet.2014.07.022
Landete JM, Langa S, Escudero C, Peirotén A, Arqués JL (2020) Fluorescent detection of nisin by genetically modified Lactococcus lactis strains in milk and a colonic model: application of whole-cell nisin biosensors. J Biosci Bioeng 129:435–440. https://doi.org/10.1016/j.jbiosc.2019.10.011
Landete JM, Langa J, Revilla C, Margolles A, Medina M, Arqués J (2015) Use of anaerobic green fluorescent protein versus green fluorescent protein as reporter in lactic acid bacteria. Appl Microbiol Biotechnol 99:6865–6877. https://doi.org/10.1007/s00253-015-6770-3
Landete JM, Peirotén A, Medina M, Arqués JL (2017) Labeling Listeria with anaerobic fluorescent protein for food safety studies. J Dairy Sci 100:113–117. https://doi.org/10.3168/jds.2016-11226
Landete JM, Peirotén A, Rodríguez E, Margolles A, Medina M, Arqués JL (2014) Anaerobic green fluorescent protein as a marker of Bifidobacterium strains. Int J Food Microbiol 175:6–13. https://doi.org/10.1016/j.ijfoodmicro.2014.01.008
Langa S, Landete JM, Martín-Cabrejas I, Rodríguez E, Arqués JL, Medina M (2013) In situ reuterin production by Lactobacillus reuteri in dairy products. Food Control 33:200–206. https://doi.org/10.1016/j.foodcont.2013.02.035
Langa S, Arqués JL, Medina M, Landete JM (2017) Coproduction of colicin V and lactic acid bacteria bacteriocins in lactococci and enterococci strains of biotechnological interest. J Appl Microbiol 122:1159–1167. https://doi.org/10.1111/jam.13439
Leroy F, De Vuyst L (2004) Lactic acid bacteria as functional starter cultures for the food fermentation industry. Trends Food Sci Technol 2:67–78. https://doi.org/10.1016/j.tifs.2003.09.004
Li R, Takala TM, Qiao M, Xu H, Saris PEJ (2011) Nisin-selectable food-grade secretion vector for Lactococcus lactis. Biotechnol Lett 33:797–803. https://doi.org/10.1007/s10529-010-0503-6
Lu W, Kong J, Kong W (2013) Construction and application of a food-grade expression system for Lactococcus lactis. Mol Biotechnol 54:170–176. https://doi.org/10.1007/s12033-012-9558-z
MacCormick CA, Griffin HG, Gasson MJ (1995) Construction of a food-grade host/vector system for Lactococcus lactis based on the lactose operon. FEMS Microbiol Lett 127:105–109. https://doi.org/10.1111/j.1574-6968.1995.tb07457.x
Mazé A, Boël G, Zúñiga M, Bourand A, Loux V, Yebra MJ, Monedero V, Correia K, Jacques N, Beaufils S, Poncet S, Joyet P, Milohanic E, Casaregola S, Auffray Y, Pérez-Martínez G, Gibrat JF, Zagorec M, Francke C, Hartke A, Deutscher J (2010) Complete genome sequence of the probiotic Lactobacillus casei strain BL23. J Bacteriol 192:2647–2648. https://doi.org/10.1128/JB.00076-10
McAuliffe O, Hill C, Ross RP (2000) Identification and overexpression of ltnl, a novel gene which confers immunity to the two-component lantibiotic lacticin 3147. Microbiology (reading) 146:129–138. https://doi.org/10.1099/00221287-146-1-129
Mierau I, Kleerebezem M (2005) 10 years of the nisin-controlled gene expression system (NICE) in Lactococcus lactis. Appl Microbiol Biotechnol 68:705–717. https://doi.org/10.1007/s00253-005-0107-6
Montenegro-Rodríguez C, Peirotén A, Sanchez-Jimenez A, Arqués JL, Landete JM (2015) Analysis of gene expression of bifidobacteria using as the reporter an anaerobic fluorescent protein. Biotechnol Lett 37:1405–1413. https://doi.org/10.1007/s10529-015-1802-8
Morales-Contreras JA, Rodríguez-Pérez JE, Álvarez-González CA, Martínez-López MC, Juárez-Rojop IE, Ávila-Fernández A (2021) Potential applications of recombinant bifidobacterial proteins in the food industry, biomedicine, process innovation and glycobiology. Food Sci Biotechnol 30:1277–1291. https://doi.org/10.1007/s10068-021-00957-1
Peirotén A, Arqués JL, Medina M, Rodríguez-Mínguez E (2018) Bifidobacterial strains shared by mother and child as source of probiotics. Benef Microbes 9:231–238. https://doi.org/10.3920/BM2017.0133
Peirotén A, Gaya P, Álvarez I, Landete JM (2020) Production of O-desmethylangolensin, tetrahydrodaidzein, 6’-hydroxy-O-desmethylangolensin and 2-(4-hydroxyphenyl)-propionic acid in fermented soy beverage by lactic acid bacteria and Bifidobacterium strains. Food Chem 318:126521. https://doi.org/10.1016/j.foodchem.2020.126521
Peirotén A, Gaya P, Landete JM (2020) Application of recombinant lactic acid bacteria and bifidobacteria able to enrich soy beverage in dihydrodaidzein and dihydrogenistein. Food Res Int 134:109257. https://doi.org/10.1016/j.foodres.2020.109257
Plavec TV, Berlec A (2020) Safety aspects of genetically modified lactic acid bacteria. Microorganisms 8:297. https://doi.org/10.3390/microorganisms8020297
Reviriego C, Fernández L, Rodríguez JM (2007) A food-grade system for production of pediocin PA-1 in nisin-producing and non–nisin-producing Lactococcus lactis strains: application to inhibit Listeria growth in a cheese model system. J Food Prot 70:2512–2517
Rodríguez E, Arqués JL, Rodríguez R, Peirotén A, Landete JM, Medina M (2012) Antimicrobial properties of probiotic strains isolated from breast-fed infants. J Funct Foods 4:542–551. https://doi.org/10.1016/j.jff.2012.02.015
Rodrı́guez E, González B, Gaya P, Nuñez M, Medina M (2000) Diversity of bacteriocins produced by lactic acid bacteria isolated from raw milk. Int Dairy J 10:7–15. https://doi.org/10.1016/S0958-6946(00)00017-0
Sridhar VR, Smeianov VV, Steele JL (2006) Construction and evaluation of food-grade vectors for Lactococcus lactis using aspartate aminotransferase and alpha-galactosidase as selectable markers. J Appl Microbiol 101:161–171. https://doi.org/10.1111/j.1365-2672.2006.02898.x
Takala TM, Saris PE (2002) A food-grade cloning vector for lactic acid bacteria based on the nisin immunity gene nisI. Appl Microbiol Biotechnol 59:467–471. https://doi.org/10.1007/s00253-002-1034-4
Tavares LM, de Jesus LCL, da Silva TF, Barroso FAL, Batista VL, Coelho-Rocha ND, Azevedo V, Drumond MM, Mancha-Agresti P (2020) Novel strategies for efficient production and delivery of live biotherapeutics and biotechnological uses of Lactococcus lactis: the lactic acid bacterium model. Front Bioeng Biotechnol 8:517166. https://doi.org/10.3389/fbioe.2020.517166
van de Guchte M, Kodde J, van der Vossen JM, Kok J, Venema G (1990) Heterologous gene expression in Lactococcus lactis subsp. lactis: synthesis, secretion, and processing of the Bacillus subtilis neutral protease. Appl Environ Microbiol 56:2606–2611. https://doi.org/10.1128/aem.56.9.2606-2611.1990
Xiong Z-Q, Qiao-Hui Wang Q-H, Ling-Hui Kong L-H (2017) Improving the activity of bile salt hydrolases in Lactobacillus casei based on in silico molecular docking and heterologous expression. J Dairy Sci 100:975–980. https://doi.org/10.3168/jds.2016-11720
Zhao H, Yuan L, Hu K, Liu L, Peng S, Li H, Wang H (2019) Heterologous expression of ctsR from Oenococcus oeni enhances the acid-ethanol resistance of Lactobacillus plantarum. FEMS Microbiol Lett 366:fnz192. https://doi.org/10.1093/femsle/fnz192
Zhu D, Liu F, Xu H, Bai Y, Zhang X, Saris PEJ, Qiao M (2015) Isolation of strong constitutive promoters from Lactococcus lactis subsp. lactis N8. FEMS Microbiol Lett 362:107. https://doi.org/10.1093/femsle/fnv107
Zuo F, Chen S, Marcotte H (2020) Engineer probiotic bifidobacteria for food and biomedical applications – current status and future prospective. Biotechnol Adv 45:107654. https://doi.org/10.1016/j.biotechadv.2020.107654
Funding
This study was funded by the project PID2020-11960RB-I00 from the Spanish Ministry of Science, Innovation and Universities.
Author information
Authors and Affiliations
Contributions
JML conceived and designed research, and conducted experiments. AP and SL analyzed the data. JML, SL, AP, JAC, and JLA interpreted the data and wrote the manuscript. All authors read and approved the submitted manuscript.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Langa, S., Peirotén, Á., Curiel, J.A. et al. Promoters for the expression of food-grade selectable markers in lactic acid bacteria and bifidobacteria. Appl Microbiol Biotechnol 106, 7845–7856 (2022). https://doi.org/10.1007/s00253-022-12237-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-022-12237-x