Abstract
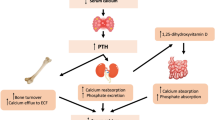
Bone turnover markers (BTMs) are released during the bone remodelling cycle and are measurable in blood or urine, reflecting bone remodelling rate. They have been useful in elucidating the pharmacodynamics and effectiveness of osteoporosis medication in clinical trials and are increasingly used in routine clinical management of osteoporosis, especially for monitoring therapy, in addition to their use in other metabolic bone disease such as Paget’s disease of bone and osteomalacia. Serum β isomerised C-terminal telopeptide of type I collagen and pro-collagen I N-terminal propeptide have been designated as reference BTMs for use in osteoporosis. In addition, bone-specific isoenzyme of alkaline phosphatase (B-ALP) secreted by osteoblasts and tartrate-resistant acid phosphatase 5b (TRACP-5b) secreted by osteoclasts are also found to be specific markers of bone formation and resorption, respectively. The concentrations of the latter enzymes in blood measured by immunoassay provide reliable measures of bone turnover even in the presence of renal failure. B-ALP is recommended for use in the assessment of renal bone disease of chronic kidney disease, and TRACP-5b shows promise as a marker of bone resorption in that condition. BTMs in blood do not suffer from biological variation to the same extent as the older BTMs that were measured in urine. Appropriate patient preparation and sample handling are important in obtaining accurate measures of BTMs for clinical use. Reference change values and treatment targets have been determined for the reference BTMs for their use in monitoring osteoporosis treatment. Further ongoing studies will enhance their clinical applications.
Similar content being viewed by others
References
Delmas PD, Eastell R, Garnero P, Seibel MJ, Stepan J (2000) The use of biochemical markers of bone turnover in osteoporosis. Osteoporos Int 11:S2–S17
Vasikaran S, Cooper C, Eastell R et al (2011) International Osteoporosis Foundation and International Federation of Clinical Chemistry and Laboratory Medicine position on bone marker standards in osteoporosis. Clin Chem Lab Med 49:1271–1274
Ralston SH, Corral-Gudino L, Cooper C et al (2019) Diagnosis and management of Paget’s disease of bone in adults: a clinical guideline. J Bone Miner Res 34:579–604
Peach H, Compston JE, Vedi S, Horton LWJ (1982) Value of plasma calcium, phosphate, and alkaline phosphatase measurements in the diagnosis of histological osteomalacia. Clin Pathol 35:625–630. https://doi.org/10.1210/jcem.77.4.8104954
Nizet A, Cavalier E, Stenvinkel P, Haarhaus M, Magnusson P (2020) Bone alkaline phosphatase: an important biomarker in chronic kidney disease—mineral and bone disorder. Clin Chim Acta 501:198–206. https://doi.org/10.1016/j.cca.2019.11.012
Li CY, Yam LT, Lam KW (1970) Acid phosphatase isoenzyme in human leukocytes in normal and pathologic conditions. J Histochem Cytochem 18:473–481. https://doi.org/10.1177/18.7.473
Halleen JM, Ylipahkala H, Alatalo SL et al (2002) Serum tartrate-resistant acid phosphatase 5b, but not 5a, correlates with other markers of bone turnover and bone mineral density. Calcif Tissue Int 71(1):20–25. https://doi.org/10.1007/s00223-001-2122-7
Janckila AJ, Yam LT (2009) Biology and clinical significance of tartrate-resistant acid phosphatases: new perspectives on an old enzyme. Calcif Tissue Int 85:465–483. https://doi.org/10.1007/s00223-009-9309-8
Vääräniemi J, Halleen JM, Kaarlonen K et al (2004) Intracellular machinery for matrix degradation in bone-resorbing osteoclasts. J Bone Miner Res 19:1432–1440. https://doi.org/10.1359/JBMR.040603
Power MJ, Fottrell PF (1991) Osteocalcin: diagnostic methods and clinical applications. Crit Rev Clin Lab Sci 28(4):287–335. https://doi.org/10.3109/10408369109106867
Wei J, Karsenty G (2015) An overview of the metabolic functions of osteocalcin. Rev Endocr Metab Disord 16:93–98. https://doi.org/10.1007/s11154-014-9307-7
Siebel MJ (2005) Biochemical markers of bone turnover part I: biochemistry and variability. Clin Biochem Rev 26:97–122
Koivula MK, Risteli L, Risteli J (2012) Measurement of aminoterminal propeptide of type I procollagen (PINP) in serum. Clin Biochem 45:920–927. https://doi.org/10.1016/j.clinbiochem.2012.03.023
Melkko J, Hellevik T, Risteli L, Risteli J, Smedsrød B (1994) Clearance of NH2-terminal propeptides of types I and III procollagen is a physiological function of the scavenger receptor in liver endothelial cells. J Exp Med 179:405–412. https://doi.org/10.1084/jem.179.2.405
Saunders PTK, Renegar RH, Raub TJ et al (1985) The carbohydrate structure of porcine uteroferrin and the role of the high mannose chains in promoting uptake by the reticuloendothelial cells of the fetal liver. J Biol Chem 260:3658–3665
Halleen J, Hentunen TA, Hellman J, Väänänen HK (1996) Tartrate-resistant acid phosphatase from human bone: purification and development of an immunoassay. J Bone Miner Res 11:1444–1452. https://doi.org/10.1002/jbmr.5650111011
Chu P, Chao TY, Lin YF, Janckila AJ, Yam LT (2003) Correlation between histomorphometric parameters of bone resorption and serum type 5b tartrate-resistant acid phosphatase in uremic patients on maintenance hemodialysis. Am J Kidney Dis 41:1052–1059. https://doi.org/10.1016/S0272-6386(03)00203-8
Hannon RA, Clowes JA, Eagleton AC, Al Hadari A, Eastell R, Blumsohn A (2004) Clinical performance of immunoreactive tartrate-resistant acid phosphatase isoform 5b as a marker of bone resorption. Bone 34:187–194. https://doi.org/10.1016/j.bone.2003.04.002
Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group (2011) KDIGO 2017 clinical practice guideline update for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). Kidney Int Suppl 7(1):1–59. https://doi.org/10.1016/j.kisu.2017.04.001
Fukumoto S, Ozono K, Michigami T et al (2015) Pathogenesis and diagnostic criteria for rickets and osteomalacia—proposal by an expert panel supported by the Ministry of Health, Labour and Welfare, Japan, the Japanese Society for Bone and Mineral Research, and the Japan Endocrine Society. J Bone Miner Metab 33(5):467–473. https://doi.org/10.1007/s00774-015-0698-7
Camacho PM, Petak SM, Binkley N et al (2020) American Association of Clinical Endocrinologists/American College of Endocrinology clinical practice guidelines for the diagnosis and treatment of postmenopausal osteoporosis—2020 update. Endocr Pract 26(Suppl 1):1–46
National Kidney Foundation (2007) KDOQI clinical practice guidelines and clinical practice recommendations for diabetes and chronic kidney disease. Am J Kidney Dis 49(Suppl 2):S12-154
Vasikaran SD, McCloskey EV, Kahn S, Kanis JA (1994) Intra-individual variation in fasting urinary calcium- and hydroxyproline-creatinine ratios measured in metabolic bone clinic patients as both outpatients and inpatients. Ann Clin Biochem 31:272–276. https://doi.org/10.1177/000456329403100310
Beck-Jensen JE, Kollerup G, Sørensen HA, Pors Nielsen S, Sørensen OH (1997) A single measurement of biochemical markers of bone turnover has limited utility in the individual person. Scand J Clin Lab Invest 57(4):351–359. https://doi.org/10.3109/00365519709099408
Cavalier E, Lukas P, Bottani M, European Federation of Clinical Chemistry and Laboratory Medicine Working Group on Biological Variation and IOF-IFCC Committee on Bone Metabolism et al (2020) European Biological Variation Study (EuBIVAS): within- and between-subject biological variation estimates of β-isomerized C-terminal telopeptide of type I collagen (β-CTX), N-terminal propeptide of type I collagen (PINP), osteocalcin, intact fibroblast growth factor 23 and uncarboxylated-unphosphorylated matrix-Gla protein—a cooperation between the EFLM Working Group on Biological Variation and the International Osteoporosis Foundation-International Federation of Clinical Chemistry Committee on Bone Metabolism. Osteoporos Int 31:1461–1470. https://doi.org/10.1007/s00198-020-05362-8
Szulc P, Naylor K, Hoyle NR et al (2017) Use of CTX-I and PINP as bone turnover markers: National Bone Health Alliance recommendations to standardize sample handling and patient preparation to reduce pre-analytical variability. Osteoporos Int 28:2541–2556
Bhattoa HP, Cavalier E, Eastell R, IFCC-IOF Committee for Bone Metabolism et al (2021) Analytical considerations and plans to standardize or harmonize assays for the reference bone turnover markers PINP and β-CTX in blood. Clin Chim Acta 515:16–20. https://doi.org/10.1016/j.cca.2020.12.023
Ohashi T, Igarashi Y, Mochizuki Y et al (2007) Development of a novel fragments absorbed immunocapture enzyme assay system for tartrate-resistant acid phosphatase 5b. Clin Chim Acta 376(1–2):205–20512. https://doi.org/10.1016/j.cca.2006.08.021
Nakanishi M, Yoh K, Miura T, Ohasi T, Rai SK, Uchida K (2000) Development of a kinetic assay for band 5b tartrate-resistant acid phosphatase activity in serum. Clin Chem 46(4):469–473
Halleen JM, Alatalo SL, Suominen H, Cheng S, Janckila AJ, Väänänen HK (2000) Tartrate-resistant acid phosphatase 5b: a novel serum marker of bone resorption. J Bone Miner Res 15:1337–1345. https://doi.org/10.1359/jbmr.2000.15.7.1337
Ylipahkala H, Fagerlund KM, Janckila AJ, Houston B, Laurie D, Halleen JM (2009) Specificity and clinical performance of two commercial TRACP 5b immunoassays. Clin Lab 55:223–228
Morris HA, Eastell R, Jorgensen NR, IFCC-IOF Working Group for Standardisation of Bone Marker Assays (WG-BMA), et al (2017) Clinical usefulness of bone turnover marker concentrations in osteoporosis. Clin Chim Acta 467:34–41. https://doi.org/10.1016/j.cca.2016.06.036
Jørgensen NR, Møllehave LT, Hansen YBL, Quardon N, Lylloff L, Linneberg A (2017) Comparison of two automated assays of BTM (CTX and P1NP) and reference intervals in a Danish population. Osteoporos Int 28:2103–2113
Tokida R, Uehara M, Nakano M et al (2021) Reference values for bone metabolism in a Japanese cohort survey randomly sampled from a basic elderly resident registry. Sci Rep 11(1):7822. https://doi.org/10.1038/s41598-021-87393-7
Kikuchi W, Ichihara K, Mori K, Shimizu Y (2021) Biological sources of variations of tartrate-resistant acid phosphatase 5b in a healthy Japanese population. Ann Clin Biochem 58(4):358–367. https://doi.org/10.1177/00045632211003941
Nishizawa Y, Miura M, Ichimura S, from the Japan Osteoporosis Society Bone Turnover Marker Investigation Committee, et al (2019) Executive summary of the Japan Osteoporosis Society Guide for the use of bone turnover markers in the diagnosis and treatment of osteoporosis (2018 edition). Clin Chim Acta 498:101–107. https://doi.org/10.1016/j.cca.2019.08.012
Eastell R, Pigott T, Gossiel F, Naylor KE, Walsh JS, Peel NFA (2018) Diagnosis of endocrine disease: bone turnover markers: are they clinically useful? Eur J Endocrinol 178:R19–R31. https://doi.org/10.1530/EJE-17-0585
Lorentzon M, Branco J, Brandi ML et al (2019) Algorithm for the use of biochemical markers of bone turnover in the diagnosis, assessment and follow-up of treatment for osteoporosis. Adv Ther 36:2811–2824. https://doi.org/10.1007/s12325-019-01063-9
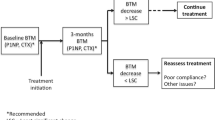
Tan RZ, Loh TP, Vasikaran S (2020) Bone turnover marker monitoring in osteoporosis treatment response. Eur J Endocrinol 183:C5–C7. https://doi.org/10.1530/EJE-19-0970
Naylor KE, Jacques RM, Paggiosi M et al (2016) Response of bone turnover markers to three oral bisphosphonate therapies in postmenopausal osteoporosis: the TRIO study. Osteoporos Int 27:21–31. https://doi.org/10.1007/s00198-015-3145-7
Krege JH, Lane NE, Harris JM, Miller PD (2014) PINP as a biological response marker during teriparatide treatment for osteoporosis. Osteoporos Int 25:2159–2171. https://doi.org/10.1007/s00198-014-2646-0
Adler RA, El-Hajj Fuleihan G, Bauer DC et al (2016) Managing osteoporosis in patients on long-term bisphosphonate treatment: report of a Task Force of the American Society for Bone and Mineral Research. J Bone Miner Res 31(1):16–35. https://doi.org/10.1002/jbmr.2708
Tsourdi E, Zillikens MC, Meier C et al (2020) Fracture risk and management of discontinuation of denosumab therapy: a systematic review and position statement by ECTS. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgaa756
Kobayakawa T, Suzuki T, Nakano M et al (2021) Real-world effects and adverse events of romosozumab in Japanese osteoporotic patients: a prospective cohort study. Bone Rep 14:101068. https://doi.org/10.1016/j.bonr.2021.101068
Tsuchiya K, Ishikawa K, Kudo Y et al (2021) Analysis of the subsequent treatment of osteoporosis by transitioning from bisphosphonates to denosumab, using quantitative computed tomography: a prospective cohort study. Bone Rep 14:101090. https://doi.org/10.1016/j.bonr.2021.101090
Mori Y, Kasai H, Ose A et al (2018) Modeling and simulation of bone mineral density in Japanese osteoporosis patients treated with zoledronic acid using tartrate-resistant acid phosphatase 5b, a bone resorption marker. Osteoporos Int 29:1155–1163. https://doi.org/10.1007/s00198-018-4376-1
Kasai H, Mori Y, Ose A, Shiraki M, Tanigawara Y (2021) Prediction of fracture risk from early-stage bone markers in patients with osteoporosis treated with once-yearly administered zoledronic acid. J Clin Pharmacol 61:606–613
Kunizawa K, Hiramatsu R, Hoshino J et al (2020) Denosumab for dialysis patients with osteoporosis: a cohort study. Sci Rep 10:2496. https://doi.org/10.1038/s41598-020-59143-8
Moe S, Drüeke T, Cunningham J et al (2006) Definition, evaluation, and classification of renal osteodystrophy: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int 69:1945–1953. https://doi.org/10.1038/sj.ki.5000414
Sprague SM, Bellorin-Font E, Jorgetti V et al (2016) Diagnostic accuracy of bone turnover markers and bone histology in patients with CKD treated by dialysis. Am J Kidney Dis 67:559–566. https://doi.org/10.1053/j.ajkd.2015.06.023
Salam S, Gallagher O, Gossiel F et al (2018) Diagnostic accuracy of biomarkers and imaging for bone turnover in renal osteodystrophy. J Am Soc Nephrol 29:1557–1565. https://doi.org/10.1681/ASN.2017050584
Seifert ME, Hruska KA (2016) The kidney-vascular-bone axis in the chronic kidney disease-mineral bone disorder. Transplantation 100:497–505. https://doi.org/10.1097/TP.0000000000000903
Delanaye P, Souberbielle J-C, Lafage-Proust M-H et al (2014) Can we use circulating biomarkers to monitor bone turnover in CKD haemodialysis patients? Hypotheses and facts. Nephrol Dial Transplant 29:997–1004. https://doi.org/10.1093/ndt/gft275
Magnusson P, Sharp CA, Magnusson M et al (2001) Effect of chronic renal failure on bone turnover and bone alkaline phosphatase isoforms. Kidney Int 60:257–265. https://doi.org/10.1046/j.1523-1755.2001.00794.x
Tridimas A, Milan A, Marks E (2021) Assessing bone formation in patients with chronic kidney disease using procollagen type I N-terminal propeptide (PINP): the choice of assay makes a difference. Ann Clin Biochem 58:528–536. https://doi.org/10.1177/00045632211025567
Cavalier E, Lukas P, Carlisi A et al (2013) Aminoterminal propeptide of type I procollagen (PINP) in chronic kidney disease patients: the assay matters. Clin Chim Acta 425:117–118. https://doi.org/10.1016/j.cca.2013.07.016
Yamada S, Inaba M, Kurajoh M et al (2008) Utility of serum tartrate-resistant acid phosphatase (TRACP5b) as a bone resorption marker in patients with chronic kidney disease: independence from renal dysfunction. Clin Endocrinol 69:189–196. https://doi.org/10.1111/j.1365-2265.2008.03187.x
Malluche HH, Davenport DL, Cantor T, Monier-Faugere M-C (2014) Bone mineral density and serum biochemical predictors of bone loss in patients with CKD on dialysis. Clin J Am Soc Nephrol 9:1254–1262. https://doi.org/10.2215/CJN.09470913
Yamada S, Tsuruya K, Yoshida H et al (2013) The clinical utility of serum tartrate-resistant acid phosphatase 5b in the assessment of bone resorption in patients on peritoneal dialysis. Clin Endocrinol (Oxf) 78:844–851. https://doi.org/10.1111/cen.1207
Shidara K, Inaba M, Okuno S et al (2008) Serum levels of TRAP5b, a new bone resorption marker unaffected by renal dysfunction, as a useful marker of cortical bone loss in hemodialysis patients. Calcif Tissue Int 82:278–287. https://doi.org/10.1007/s00223-008-9127-4
Evenepoel P, Claes K, Meijers B et al (2020) Natural history of mineral metabolism, bone turnover and bone mineral density in de novo renal transplant recipients treated with a steroid minimization immunosuppressive protocol. Nephrol Dial Transplant 35:697–705. https://doi.org/10.1093/ndt/gfy306
Suzuki H, Kihara M, Mano S et al (2017) Efficacy and safety of denosumab for the treatment of osteoporosis in patients with chronic kidney disease. J Clin Exp Nephrol 2:1–5. https://doi.org/10.21767/2472-5056.100030
Ruggiero SL, Dodson TB, Fantasia J, American Association of Oral and Maxillofacial Surgeons et al (2014) American Association of Oral and Maxillofacial Surgeons position paper on medication-related osteonecrosis of the jaw—2014 update. J Oral Maxillofac Surg 72:1938–1956. https://doi.org/10.1016/j.joms.2014.04.031
Shane E, Burr D, Ebeling PR et al (2010) Atypical subtrochanteric and diaphyseal femoral fractures: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res 25:2267–2294
Marx RE, Cillo JE Jr, Ulloa JJ (2007) Oral bisphosphonate-induced osteonecrosis: risk factors, prediction of risk using serum CTX testing, prevention, and treatment. J Oral Maxillofac Surg 65:2397–2410. https://doi.org/10.1016/j.joms.2007.08.003
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Disclosures
EC is a consultant for Diasorin, IDS, Fujirebio, Menarini and Nittobo. MM is a consulatant for Beckmann-Coulter Japan and Nittobo Medical. RP is a member of advisory board for Amgen Czech Republic and has received a speaker honorarium from Amgen, Takeda, Roche, DiaSorin, Abbott and Beckmann-Coulter. HB and SV have no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Vasikaran, S.D., Miura, M., Pikner, R. et al. Practical Considerations for the Clinical Application of Bone Turnover Markers in Osteoporosis. Calcif Tissue Int 112, 148–157 (2023). https://doi.org/10.1007/s00223-021-00930-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-021-00930-4