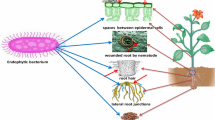
Abstract
Similar to human microbiome, plants are also colonized by a large number of tiny microorganisms. Plant endophytic bacteria colonize the internal tissues of plants without causing apparent diseases or showing any symptoms to their host. Endophytes that constitute the plant endobiome serve various functions from plant growth promotion and stress tolerance to the modulation of the plant’s chemistry. Medicinal plants are the inherent source for the production of a large number of bioactive phytochemicals, but with recent understanding on plant–microbe interactions, it has been observed that a significant number of these metabolites result from the contribution of associated microbial partners. Therefore, endophytes, particularly from medicinal plants whose microbiome remains largely unknown, have extended immense interest of scientific community in terms of bio-prospection for bioactive metabolites such as novel antimicrobial compounds, pharmacologically relevant drugs, and enzymes with novel properties. Majority of these bacterial endophytes producing bioactive metabolites belong to the Gram positive, high G + C, actinobacteria. For instance, endophytic Streptomyces spp. produce numerous novel antibiotics active against multi-drug-resistant bacteria. This chapter gives a comprehensive summary of the diversity of endophytic bacteria that colonize the internal tissues of various medicinal plants and their prospection for bioactive compounds. The chapter also describes the role of such compounds in agriculture, food, environment, and medicines. Possible future aspects of plant microbiome study in context to medicinal plants for better understanding the association of bacterial endophytes are also discussed.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Ahemad M, Kibret M (2014) Mechanisms and applications of plant growth promoting rhizobacteria: current perspective. J King Saud Univ Sci 26:1–20
Akinsanya MA, Goh JK, Lim SP et al (2015a) Diversity, antimicrobial and antioxidant activities of culturable bacterial endophyte communities in Aloe vera. FEMS Microbiol Lett 362(23):fnv184. https://doi.org/10.1093/femsle/fnv184
Akinsanya MA, Goh JK, Lim SP et al (2015b) Metagenomics study of endophytic bacteria in Aloe vera using next-generation technology. Genom Data 6:159–163. https://doi.org/10.1016/j.gdata.2015.09.004
Aranda FJ, Teruel JA, Ortiz A (2005) Further aspects on the haemolytic activity of the antibiotic lipopeptide iturin A. Biochim Biophys Acta 1713:51–56. https://doi.org/10.1016/j.bbamem.2005.05.003
Bauer A, Brönstrup M (2014) Industrial natural product chemistry for drug discovery and development. Nat Prod Rep 31:35–60
Bieber B, Nuske J, Ritzau M et al (1998) Alnumycin, a new naphthoquinone antibiotic produced by an endophytic Streptomyces sp. J Antibiot 51:381–382
Boonsnongcheep P, Nakashima T, Takahashi Y et al (2017) Diversity of endophytic actinomycetes isolated from roots and root nodules of Pueraria candollei Grah. ex Benth. and the analyses of their secondary metabolites. Chiang Mai J Sci 44:1–14
Brayfield A (ed) (2013) Doxorubicin. Martindale: the complete drug reference. Pharmaceut Press. Accessed 15 Apr 2014
Breidenbach B, Pump J, Dumont MG (2015) Microbial community structure in the rhizosphere of rice plants. Front Microbiol 6:1537. https://doi.org/10.3389/fmicb.2015.01537
Bukin Y, Galachyants Y, Morozov I et al (2019) The effect of 16S rRNA region choice on bacterial community metabarcoding results. Sci Data 6:190007. https://doi.org/10.1038/sdata.2019.7
Carvalho PLN, Silva EO, Paula DAC et al (2016) Importance and implications of the production of phenolic secondary metabolites by endophytic fungi: a mini-review. Mini Rev Med Chem 16:259–271
Castillo UF, Strobel GA, Ford EJ et al (2002) Munumbicins, wide-spectrum antibiotics produced by Streptomyces NRRL30562, endophyticon Kennedia nigriscans. Microbiology 148:2675–2785. https://doi.org/10.1099/00221287-148-9-2675
Castillo UF, Strobel GA, Mullenberg K et al (2006) Munumbicins E-4 and E-5: novel broad-spectrum antibiotics from Streptomyces sp. NRRL 3052. FEMS Microbiol Lett 25:296–300. https://doi.org/10.1111/j.1574-6968.2005.00080.x
Checcucci A, Maida I, Bacci G et al (2017) Is the plant-associated microbiota of Thymus spp. adapted to plant essential oil? Res Microbiol 168(3):276–282. https://doi.org/10.1016/j.resmic.2016.11.004
Coleman CI, Hebert JH, Reddy P (2003) The effects of Panax ginseng on quality of life. J Clin Pharmacol 28:5–15
Corcoll N, Österlund T, Sinclair L et al (2017) Comparison of four DNA extraction methods for comprehensive assessment of 16S rRNA bacterial diversity in marine biofilms using high-throughput sequencing. FEMS Microbiol Lett 364(14):fnx139. https://doi.org/10.1093/femsle/fnx139
Cragg GM, Newman DJ (2013) Natural products: a continuing source of novel drug leads. Biochim Biophys Acta 1830:3670–3695. https://doi.org/10.1016/j.bbagen.2013.02.008
Danshiitsoodol N, Pinho CA, Matoba Y et al (2006) The mitomycin C (MMC)-binding protein from MMC-producing microorganisms protects from the lethal effect of bleomycin: crystallographic analysis to elucidate the binding mode of the antibiotic to the protein. J Mol Biol 360:398–408. https://doi.org/10.1016/j.jmb.2006.05.017
Delmotte N, Ahrens CH, Knief C et al (2010) An integrated proteomics and transcriptomics reference data set provides new insights into the Bradyrhizobium japonicum bacteroid metabolism in soybean root nodules. Proteomics 10:1391–1400
Ding L, Münch J, Goerls H et al (2010) Xiamycin, a pentacyclic indolosesquiterpene with selective anti-HIV activity from a bacterial mangrove verendophyte. Bioorg Med Chem Lett 20:6685–6687. https://doi.org/10.1016/j.bmcl.2010.09.010
Dong L, Cheng R, Xiao L et al (2018) Diversity and composition of bacterial endophytes among plant parts of Panax notoginseng. Chin Med 13:41. https://doi.org/10.1186/s13020-018-0198-5
El-Deeb B, Fayez K, Gherbawy Y (2013) Isolation and characterization of endophytic bacteria from Plectranthus tenuiflorus medicinal plant in Saudi Arabia desert and their antimicrobial activities. J Plant Interact 8(1):56–64. https://doi.org/10.1080/17429145.2012.680077
Espinasse S, Gohar M, Lereclus D et al (2002) An ABC transporter from Bacillus thuringiensis is essential for beta-exotoxin I production. J Bacteriol 184:5848–5854. https://doi.org/10.1128/JB.184.21.5848-5854
Etalo DW, Jeon J-S, Raaijmakers JM (2018) Modulation of plant chemistry by beneficial root microbiota. Nat Prod Rep 35:398–409. https://doi.org/10.1039/c7np00057j
Ezra D, Castillo UF, Strobel GA et al (2004) Coronamycins, peptide antibiotics produced by a verticillate Streptomyces sp. (MSU-2110) endophytic on Monstera sp. Microbiololgy 150:785–793. https://doi.org/10.1099/mic.0.26645-0
Francis IM, Jourdan S, Fanara S et al (2015) The cellobiose sensor CebR is the gatekeeper of Streptomyces scabies pathogenicity. mBio 6:02018-14. https://doi.org/10.1128/mBio.02018-14
Frank A, Saldierna Guzmán J, Shay J (2017) Transmission of bacterial endophytes. Microorganisms 5:70. https://doi.org/10.3390/microorganisms5040070
Gendy MM, Bondkly AM (2010) Production and genetic improvement of a novel antimycotic agent, saadamycin, against dermatophytes and other clinical fungi from endophytic Streptomyces sp. Hedaya 48. J Ind Microbiol Biotechnol 37:831–841
Gilbert JA, Jansson JK, Knight R (2014) The earth microbiome project: successes and aspirations. BMC Biol 12:69. https://doi.org/10.1186/s12915-014-0069-1
Grady EN, MacDonald J, Liu L et al (2016) Current knowledge and perspectives of Paenibacillus: a review. Microb Cell Fact 15:203. https://doi.org/10.1186/s12934-016-0603-7
Greub G (2012) Culturomics: a new approach to study the human microbiome. Clin Microbiol Infect 18(12):1157–1159. https://doi.org/10.1111/1469-0691.12032
Großkinsky DK, Syaifullah SJ, Roitsch T (2018) Integration of multi-omics techniques and physiological phenotyping within a holistic phenomics approach to study senescence in model and crop plants. J Exp Bot 69:825–844. https://doi.org/10.1093/jxb/erx333
Hassan SED (2017) Plant growth-promoting activities for bacterial and fungal endophytes isolated from medicinal plant of Teucrium polium L. J Adv Res 8:687–695. https://doi.org/10.1016/j.jare.2017.09.001
Hassan MK, McInroy JA, Kloepper JW (2019) The interactions of rhizodeposits with plant growth-promoting rhizobacteria in the rhizosphere. Agric Rev 9:142. https://doi.org/10.3390/agriculture9070142
Huang Y (2019) Illumina-based analysis of endophytic bacterial diversity of four Allium species. Sci Rep 9:15271. https://doi.org/10.1038/s41598-019-51707-7
Igarashi Y (2004) Screening of novel bioactive compounds from plant associated actinomycetes. Actinomycetologica 18:63–66. https://doi.org/10.3209/saj.18_63
Igarashi Y, Yanase S, Sugimoto K et al (2011) Lupinacidin, C., an inhibitor of tumor cell invasion from Micromonospora lupini. J Nat Prod 74:862–865. https://doi.org/10.1021/np100779t
Islam SMA, Math RK, Kim JM et al (2010) Effect of plant age on endophytic bacterial diversity of balloon flower (Platycodon grandiflorum) root and their antimicrobial activities. Curr Microbiol 61:346. https://doi.org/10.1007/s00284-010-9618-1
Janardhan BS, Vijayan K (2012) Types of endophytic bacteria associated with traditional medicinal plant Lantana camara Linn. Phcog J 4(32):20–23. https://doi.org/10.5530/pj.2012.32.4
Jiang Z-k, Tuo L, Huang D-l et al (2018) Diversity, novelty, and antimicrobial activity of endophytic actinobacteria from mangrove plants in Beilun Estuary National Nature Reserve of Guangxi, China. Front Microbiol 9:868. https://doi.org/10.3389/fmicb.2018.00868
Jin H, Yang X-Y, Yan Z-Q et al (2014) Characterization of rhizosphere and endophytic bacterial communities from leaves, stems and roots of medicinal Stellera chamaejasme L. Syst Appl Microbiol 37(5):376–385. https://doi.org/10.1016/j.syapm.2014.05.001
Kandel SL, Joubert PM, Doty SL (2017) Bacterial endophyte colonization and distribution within plants. Microorganisms 5:77. https://doi.org/10.3390/microorganisms5040077
Khamna S, Yokota A, Peberdy JF et al (2010) Indole-3-acetic acid production by Streptomyces sp. isolated from some Thai medicinal plant rhizosphere soils. Eurasia J Biosci 4:23–32. https://doi.org/10.5053/ejobios.2010.4.0.4
Khan Chowdhury ME, Jeon J, Ok Rim S et al (2017) Composition, diversity and bioactivity of culturable bacterial endophytes in mountain-cultivated ginseng in Korea. Sci Rep 7:10098. https://doi.org/10.1038/s41598-017-10280-7
Kim TU, Cho SH, Han JH et al (2012) Diversity and physiological properties of root endophytic actinobacteria in native herbaceous plants of Korea. J Microbiol 50:50–57
Korkina LG (2007) Phenylpropanoids as naturally occurring antioxidants: from plant defense to human health. Cell Mol Biol 53:15–25. https://doi.org/10.1170/T772
Lagier J-C, Armougom F, Million M et al (2012) Microbial culturomics: paradigm shift in the human gut microbiome study. Clin Microbiol Infect 18:1185–1193
Lagier J-C, Hugon P, Khelaifia S et al (2015) The rebirth of culture in microbiology through the example of culturomics to study human gut microbiota. Clin Microbiol Rev 28:237–264. https://doi.org/10.1128/CMR.00014-14
Levy A, Conway JM, Dangi JL et al (2018) Elucidating bacterial gene functions in the plant microbiome. Cell Host Microbe 24:475–485. https://doi.org/10.1016/j.chom.2018.09.005
Li J, Zhao GZ, Varma A, Qin S et al (2012) An endophytic Pseudonocardia species induces the production of artemisinin in Artemisia annua. PLoS One 7:e51410. https://doi.org/10.1371/journal.pone.0051410
Li FN, Pan Z, Tuo L et al (2017) Studies on the diversity and novelty of endophytic actinobacteria isolated from mangrove plants collected in Macao. Chin J Antibiot 42:26–34. https://doi.org/10.13461/j.cnki.cja.005906
Liu YH, Liang ZS, Chen B et al (2010) Elicitation of alkaloids in in vitro PLB (protocorm-like body) cultures of Pinellia ternate. Enzym Microb Technol 46:28–31
Liu Y, Liu W, Liang Z (2015) Endophytic bacteria from Pinellia ternata, a new source of purine alkaloids and bacterial manure. Pharm Biol 5:1545–1548. https://doi.org/10.3109/13880209.1016580
Liu YH, Guo JW, Salam N et al (2016) Culturable endophytic bacteria associated with medicinal plant Ferula songorica: molecular phylogeny, distribution and screening for industrially important traits. 3 Biotech 6:209. https://doi.org/10.1007/s13205-016-0522-7
Liu H, Carvalhais LC, Crawford M et al (2017a) Inner plant values: diversity, colonization and benefits from endophytic bacteria. Front Microbiol 8:2552. https://doi.org/10.3389/fmicb.2017.02552
Liu Y, Guo J, Li L et al (2017b) Endophytic bacteria associated with endangered plant Ferula sinkiangensis K.M. Shen in an arid land: diversity and plant growth-promoting traits. J Arid Land 9:432–445
Liu T, Zhang X, Tian S et al (2020a) Bioinformatics analysis of endophytic bacteria related to berberine in the Chinese medicinal plant Coptis teeta wall. 3 Biotech 10:96. https://doi.org/10.1007/s13205-020-2084-y
Liu Z, Ma A, Mathé E et al (2020b) Network analyses in microbiome based on high-throughput multi-omics data. Brief Bioinform 22(2):1639–1655. https://doi.org/10.1093/bib/bbaa005
Loots DT, Van der Westhuizen FH, Botes L (2007) Aloe ferox leaf gel phytochemical content, antioxidant capacity, and possible health benefits. J Agr Food Chem 55:6891–6896
Lowicki D, Nski AH (2013) Structure and antimicrobial properties of Monensin A and its derivatives: summary of the achievements. Biomed Res Int 2013:742149. https://doi.org/10.1155/2013/742149
Lu C, Shen Y (2007) A novel ansamycin, naphthomycin K from Streptomyces sp. J Antibiot 60:649–653. https://doi.org/10.1038/ja.2007.84
Lucaciu R, Pelikan C, Gerner SM et al (2019) A bioinformatics guide to plant microbiome analysis. Front Plant Sci 10:1313. https://doi.org/10.3389/fpls.2019.01313
Ma L, Cao YH, Cheng MH et al (2013) Phylogenetic diversity of bacterial endophytes of Panax notoginseng with antagonistic characteristics towards pathogens of root-rot disease complex. Antonie Van Leeuwenhoek 103:299. https://doi.org/10.1007/s10482-012-9810-3
Machavariani NG, Ivankova TD, Sineva ON et al (2014) Isolation of endophytic actinomycetes from medicinal plants of the Moscow region, Russia. World Appl Sci J 30:1599–1604
Meneses CHSG, Rouws LFM, Simoes-Araujo JL et al (2011) Exopolysaccharide production is required for biofilm formation and plant colonization by the nitrogen-fixing endophyte Gluconacetobacter diazotrophicus. Mol Plant Microbe Interact 24:1448–1458
Mitter B, Pfaffenbichler N, Flavell R et al (2017) A new approach to modify plant microbiomes and traits by introducing beneficial bacteria at flowering into progeny seeds. Front Microbiol 8:1–10. https://doi.org/10.3389/fmicb.2017.00011
Nguyen TM, Seo C, Ji M et al (2018) Effective soil extraction method for cultivating previously uncultured soil bacteria. Appl Environ Microbiol 84:e01145–e01118. https://doi.org/10.1128/AEM.01145-18
Nongkhlaw FMW, Joshi SR (2015) Investigation on the bioactivity of culturable endophytic and epiphytic bacteria associated with ethnomedicinal plants. J Infect Dev Ctries 9:954–961. https://doi.org/10.3855/jidc.4967
Oberhofer M, Hess J, Leutgeb M et al (2019) Exploring actinobacteria associated with rhizosphere and endosphere of the native alpine medicinal plant Leontopodium nivale subspecies alpinum. Front Microbiol 10:2531. https://doi.org/10.3389/fmicb.2019.02531
Okazaki T (2003) Studies on actinomycetes isolated from plant leaves. In: Kurtböke I (ed) Selective isolation of rare Actinomycetes. National Library of Australia, Canberra, pp 102–122
Panche AN, Diwan AD, Chandra SR (2016) Flavonoids: an overview. J Nutr Sci 5:115. https://doi.org/10.1017/jns.2016.41
Passari AK, Mishra VK, Saikia R et al (2015) Isolation, abundance and phylogenetic affiliation of endophytic actinomycetes associated with medicinal plants and screening for their in vitro antimicrobial biosynthetic potential. Front Microbiol 6:273
Passari AK, Mishra VK, Singh G et al (2017) Insights into the functionality of endophytic actinobacteria with a focus on their biosynthetic potential and secondary metabolites production. Sci Rep 7:11809. https://doi.org/10.1038/s41598-017-12235-4
Pesic A, Steinhaus B, Kemper S et al (2014) Isolation and structure elucidation of the nucleoside antibiotic strepturidin from Streptomyces albus DSM 40763. J Antibiot 67:471–477. https://doi.org/10.1038/ja.2014.16
Pinchuk IV, Bressollier P, Sorokulova IB et al (2002) Amicoumacin antibiotic production and genetic diversity of Bacillus subtilis strains isolated from different habitats. Res Microbiol 153:269–276. https://doi.org/10.1016/S0923-2508(02)01320-7
Plett JM, Martin FM (2018) Know your enemy, embrace your friend: using omics to understand how plants respond differently to pathogenic and mutualistic microorganisms. Plant J 93:729–746
Pullen C, Schmitz P, Meurer K et al (2002) New and bioactive compounds from Streptomyces strains residing in the wood of Celastraceae. Planta 216:162–167. https://doi.org/10.1007/s00425-002-0874-6
Purushotham N, Jones E, Monk J et al (2020) Community structure, diversity and potential of endophytic bacteria in the primitive New Zealand medicinal plant Pseudowintera colorata. Plants-Basel 9(2):E156. https://doi.org/10.3390/plants9020156
Qin S, Li J, Chen HH et al (2009) Isolation, diversity, and antimicrobial activity of rare actinobacteria from medicinal plants of tropical rain forests in Xishuangbanna, China. Appl Environ Microbiol 75(19):6176–6186. https://doi.org/10.1128/AEM.01034-09
Qin S, Chen H-H, Zhao G-Z et al (2012) Abundant and diverse endophytic actinobacteria associated Swith medicinal plant Maytenus austroyunnanensis in Xishuangbanna tropical rainforest revealed by culture-dependent and culture-independent methods. Environ Microbiol Rep 4(5):522–531. https://doi.org/10.1111/j.1758-2229.2012.00357.x
Raghavan D, Veeraraghavan S, Sheeja TE et al (2017) Endophytic actinobacteria: diversity, secondary metabolism and mechanisms to unsilence biosynthetic gene clusters. Crit Rev Microbiol 43(5):546–566. https://doi.org/10.1080/1040841X.2016.1270895
Rahman MA, Mossa JS, Al-Said MS et al (2004) Medicinal plant diversity in the flora of Saudi Arabia 1: a report on seven plant families. Fitoterapia 75:149–161
Regalado J, Lundberg DS, Deusch O et al (2019) Combining whole genome shotgun sequencing and rDNA amplicon analyses to improve detection of microbe-microbe interaction networks in plant leaves. bioRxiv 823492. https://doi.org/10.1101/823492
Ren JH, Ye JR, Liu H et al (2011) Isolation and characterization of a new Burkholderia pyrrocinia strain JK-SH007 as a potential biocontrol agent. World J Microbiol Biotechnol 27(9):2203–2215
Roh H, Uguru GC, Ko HJ et al (2011) Genome sequence of the abyssomicin- and proximicin-producing marine actinomycete Verrucosispora maris AB-18-032. J Bacteriol Res 193(13):3391–3392. https://doi.org/10.1128/JB.05041-11
Salehi B, Abu-Darwishet MS, Tarawneh AH et al (2019) Thymus spp. plants—food applications and phytopharmacy properties. Trends Food Sci Technol 85:287–306. https://doi.org/10.1016/j.tifs.2019.01.020
Sansinenea E, Ortiz A (2011) Secondary metabolites of soil Bacillus spp. Biotechnol Lett 33:1523–1538. https://doi.org/10.1007/s10529-011-0617-5
Sarhan MS, Hamza MA, Youssef HH et al (2019) Culturomics of the plant prokaryotic microbiome and the dawn of plant-based culture media—a review. J Adv Res 19:5–27. https://doi.org/10.1016/j.jare.2019.04.002
Sasaki T, Igarashi Y, Saito N et al (2001) Cedarmycins, A., and B, new antimicrobial antibiotics from Streptomyces sp. TP-A0456. J Antibiot 54:567–572. https://doi.org/10.7164/antibiotics.54.567
Sasaki T, Igarashi Y, Ogawa M et al (2002) Identification of 6-prenylindole as an antifungal metabolite of Streptomyces sp. TP-A0595 an synthesis and bioactivity of 6-substituted indoles. J Antibiot 55:1009–1012. https://doi.org/10.1002/chin.200319203
Sathiyaseelan K, Stella D (2011) Isolation, identification and antimicrobial activity of marine actinomycetes isolated from Parangipettai. Recent Res Sci Technol 3:74–77
Savi DC, Haminiuk CW, Sora GTS et al (2015) Antitumor, antioxidant and antibacterial activities of secondary metabolites extracted by endophytic actinomycetes isolated from Vochysia divergens. Int J Pharm Chem Biol Sci 5:347–356
Shan W, Zhou Y, Liu H et al (2018) Endophytic actinomycetes from tea plants (Camellia sinensis): isolation, abundance, antimicrobial, and plant-growth-promoting activities. BioMed Res Int 2018:1470305. https://doi.org/10.1155/2018/1470305
Shimizu M, Igarashi Y, Furumai T et al (2004) Identification of endophytic Streptomyces sp. R-5 and analysis of its antimicrobial metabolites. J Gen Plant Pathol 70:66–68. https://doi.org/10.1007/s10327-003-0082-7
Singh S, Bindu H, Raghu J et al (2013) Isolation of endophytic bacteria producing the anti-cancer alkaloid camptothecine from Miqueliadentata Bedd. (Icacinaceae). Phytomedicine 20:913–917. https://doi.org/10.1016/j.phymed.2013.04.004
Singh M, Kumar A, Singh R et al (2017) Endophytic bacteria: a new source of bioactive compounds. 3 Biotech 7(5):315. https://doi.org/10.1007/s13205-017-0942-z
Song X, Wu H, Yin Z et al (2017) Endophytic bacteria isolated from Panax ginseng improved ginsenoside accumulation in adventitious ginseng root culture. Molecules 22:837
de Souza RSC, Armanhi JSL, Damasceno NB et al (2019) Genome sequences of a plant beneficial synthetic bacterial community reveal genetic features for successful plant colonization. Front Microbiol 10:1779. https://doi.org/10.3389/fmicb.2019.01779
Spaepen S, Vanderleyden J (2011) Auxin and plant-microbe interactions. FEMS Microbiol Rev 11:125–148
Stein T (2005) Bacillus subtilis antibiotics: structures, syntheses and specific functions. Mol Microbiol 56:845–857. https://doi.org/10.1111/j.1365-2958.2005.04587.x
Sun L, Shi SB, Zhu J et al (2013) Traditional use and modern research review of Ferula in China. Mod Chinese Med 15:620–626
Taechowisan T, Wanbanjob A, Tuntiwachwuttikul P et al (2006) Identification of Streptomyces sp. Tc022, an endophyte in Alpinia galanga, and the isolation of actinomycin D. Ann Microbiol 56:113–117. https://doi.org/10.1007/BF03174991
Taechowisan T, Chuaychot N, Chanaphat S et al (2009) Antioxidative and inhibitory activity on nitric oxide production of flavonoids from Streptomyces sp. Tc052. J Pharm Res 2:313–316
Taechowisan T, Chanaphat S, Ruensamran W et al (2014) Antibacterial activity of new flavonoids from Streptomyces sp. BT01; an endophyte in (L.) Mansf. J Appl Pharm Sci 4:8–13. https://doi.org/10.7324/JAPS.2014.40402
Tamehiro N, Okamot-Hosova Y, Okamoto S et al (2002) Bacilysocin, a novel phospholipid antibiotic produced by Bacillus subtilis 168. Antimicrob Agents Chemother 46:315–320. https://doi.org/10.1128/AAC.46.2.315-320.2002
Tan Y, Cui Y, Kuang A et al (2016) Diversity and composition of rhizospheric soil and root endogenous bacteria in Panax notoginseng during continuous cropping practices. J Basic Microbiol 57(4):337–344. https://doi.org/10.1002/jobm.201600464
Tholl D (2015) Biosynthesis and biological functions of terpenoids in plants. Adv Biochem Eng Biotechnol 148:63–106. https://doi.org/10.1007/10_2014_295
Tiwari R, Awasthi A, Mall M et al (2013) Bacterial endophyte-mediated enhancement of in planta content of key terpenoid indole alkaloids and growth parameters of Catharanthus roseus. Ind Crop Prod 43:306–310. https://doi.org/10.1016/j.indcrop.2012.07.045
Tuntiwachwuttikul P, Taechowisan T, Wanbanjob A (2008) Secondary metabolites from Streptomyces sp. SUC1. Tetrahedron 64:7583–7586. https://doi.org/10.1016/j.tet.2008.05.104
Ullah A, Mushtaq H, Fahad S et al (2017) Plant growth promoting potential of bacterial endophytes in novel association with Olea ferruginea and Withania coagulans. Microbiology 86:119. https://doi.org/10.1134/S0026261717010155
Valdes L, Cuervo A, Salazar N et al (2015) The relationship between phenolic compounds from diet and microbiota: impact on human health. J Funct Food 6:2424–2439. https://doi.org/10.1039/c5fo00322a
Verma VC, Gond SK, Kumar A et al (2009) Endophytic actinomycetes from Azadirachta indica A. Juss: isolation, diversity, and antimicrobial activity. Microb Ecol 57:749–756
Verma VC, Singh SK, Prakash S (2011) Biocontrol and plant growth promotion potential of siderophore producing endophytic Streptomyces from Azadirachta indica A. Juss J Basic Microbiol 51:550–556. https://doi.org/10.1002/jobm.201000155
Villamil SF, Stoppani AOM, Dubin M (2004) Redox cycling of β-Lapachone and structural analogues in microsomal and cytosol liver preparations. Method Enzymol 378:67–87. https://doi.org/10.1016/S0076-6879(04)78004-0
Wang S, Hu T, Jiao Y et al (2009) Isolation and characterization of Bacillus subtilis EB-28, an endophytic bacterium strain displaying biocontrol activity against Botrytis cinerea Pers. Front Agric China 3(3):247–252
Wang S, Liu J, Sun J et al (2019a) Diversity of culture-independent bacteria and antimicrobial activity of culturable endophytic bacteria isolated from different Dendrobium stems. Sci Rep 9:10389. https://doi.org/10.1038/s41598-019-46863-9
Wang J, Wang L, Lou G-H et al (2019b) Coptidis rhizoma: a comprehensive review of its traditional uses, botany, phytochemistry, pharmacology and toxicology. Pharm Biol 57(1):193–225
Weselowski B, Nathoo N, Eastman AW et al (2016) Isolation, identification and characterization of Paenibacillus polymyxa CR1 with potentials for biopesticide, biofertilization, biomass degradation and biofuel production. BMC Microbiol 16:244
Xiong Z-J, Huang J, Yan Y et al (2018) Isolation and biosynthesis of labdanmycins: new labdane diterpenes from endophytic Streptomyces. Org Chem Front 5(8):1272–1279. https://doi.org/10.1039/C8QO00085A
Xu L, Naylor D, Dong Z et al (2018) Drought delays development of the sorghum root microbiome and enriches for monoderm bacteria. Proc Natl Acad Sci U S A 115:E4284–E4293. https://doi.org/10.1073/pnas.1717308115
Yamazaki Y, Someno T, Igarashi M et al (2015) Androprostamines A and B, the new anti-prostate cancer agents produced by Streptomyces sp. MK932-CF8. J Antibiot 68:279–285. https://doi.org/10.1038/ja.2014.135
Yang R, Liu P, Ye W (2017) Illumina-based analysis of endophytic bacterial diversity of tree peony (Paeonia sect. Moutan) roots and leaves. Braz J Microbiol 48(4):695–705
Zhang Y, Liu H, Zhang X et al (2014) Micromonospora violae sp. nov., isolated from a root of Viola philippica Car. Antonie Van Leeuwenhoek 106:219–225. https://doi.org/10.1007/s10482-014-0184-6
Zhang X, Gao Z, Zhang M et al (2016) Analysis of endophytic actinobacteria species diversity in the stem of Gynura cusimbua by 16S rRNA gene clone library. Microbiology 85:379–385. https://doi.org/10.1134/S0026261716030176
Zhao K, Li J, Shen M (2018) Actinobacteria associated with Chinaberry tree are diverse and show antimicrobial activity. Sci Rep 8:11103. https://doi.org/10.1038/s41598-018-29442-2
Zhou JY, Yuan J, Li X et al (2015) Endophytic bacterium triggered reactive oxygen species directly increase oxygenous sesquiterpenoid content and diversity in Atractylodes lancea. Appl Environ Microbiol 82:1577–1585. https://doi.org/10.1128/AEM.03434-15
Zin NM, Baba MS, Zainal-Abidin AH (2017) Gancidin, W, a potential low-toxicity antimalarial agent isolated from an endophytic Streptomyces SUK10. Drug Des Devel Ther 11:351–363. https://doi.org/10.2147/DDDT.S121283
Acknowledgements
The editors of “Bacterial Endophytes: Advances in Sustainable Agricultural and Environmental Management” are gratefully acknowledged for extending the invitation for sharing our views on one of the most promising research areas of plant–microbe interactions.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Singh, N.A., Jain, R. (2022). Diversity and Bioactive Potential of Endophytic Bacteria from High-Value Medicinal Plants. In: Singh, A.K., Tripathi, V., Shukla, A.K., Kumar, P. (eds) Bacterial Endophytes for Sustainable Agriculture and Environmental Management. Springer, Singapore. https://doi.org/10.1007/978-981-16-4497-9_3
Download citation
DOI: https://doi.org/10.1007/978-981-16-4497-9_3
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-4496-2
Online ISBN: 978-981-16-4497-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)