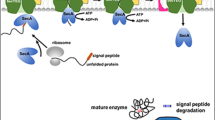
Abstract
Bacillus subtilis has been widely used for the industrial production of useful proteins because of its high protein secretion ability and safety. We focused on genome reduction as a new concept for enhancing production of recombinant enzymes in B. subtilis cells based on detailed analysis of the genome mechanism. First, we reported that a novel B. subtilis strain, MGB874, depleted 20.7 % of the genomic sequence of the wild type by rationally designed deletions to create simplified cells for protein production. When compared with wild-type cells, the productivity of cellulase and protease from transformed plasmids harboring the corresponding genes was markedly enhanced. These results indicate that a bacterial factory specializing in the production of substances can be constructed by deleting the genomic regions unimportant for growth and substance production from B. subtilis. Second, deletion of the rocDEF-rocR region, which is involved in arginine degradation, was found to contribute to the improvement of enzyme production in strain MGB874. The present study indicated that our results demonstrated the effectiveness of a synthetic genomic approach with reduction of genome size to generate novel and useful bacteria for industrial uses. Furthermore, the design of the changes in the transcriptional regulatory network of the nitrogen metabolic pathway in B. subtilis cells could facilitate the generation of improved industrial protein production.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Ara K et al (2007) Bacillus minimum genome factory: effective utilization of microbial genome information. Biotechnol Appl Biochem 46:169–178
Bohannon DE et al (1989) Positive regulation of glutamate biosynthesis in Bacillus subtilis. J Bacteriol 171:4718–4727
Commichau F et al (2007) A regulatory protein–protein interaction governs glutamate biosynthesis in Bacillus subtilis: the glutamate dehydrogenase RocG moonlights in controlling the transcription factor GltC. Mol Microbiol 65:642–654
Fabret C et al (2002) A new mutation delivery system for genome-scale approaches in Bacillus subtilis. Mol Microbiol 46:25–36
Hakamada Y et al (2000) Deduced amino acid sequence and possible catalytic residues of a thermostable, alkaline cellulase from an alkaliphilic Bacillus strain. Biosci Biotechnol Biochem 64:2281–2289
Kageyama Y et al (2009) Genome reduction in Bacillus subtilis and enhanced productivities of recombinant proteins. In: Knudsen WD, Bruns SS (eds) Bacterial DNA, DNA polymerase and DNA helicases. Nova Science, New York
Kakeshita H et al (2010) Enhanced extracellular production of heterologous proteins in Bacillus subtilis by deleting the C-terminal region of the SecA secretory machinery. Mol Biotechnol 46:250–257
Kakeshita H et al (2011a) Improvement of heterologous protein secretion by Bacillus subtilis. In: Petre M (ed) Advances in applied biotechnology. In Tech (Open access publisher)
Kakeshita H et al (2011b) Propeptide of Bacillus subtilis amylase enhances extracellular production of human interferon-α in Bacillus subtilis. Appl Microbiol Biotechnol 89:1509–1517
Kakeshita H et al (2011c) Secretion of biologically active human interferon-β by Bacillus subtilis. Biotechnol Lett 33:1847–1852
Kobayashi T et al (1995) Purification and properties of an alkaline protease from alkalophilic Bacillus sp. KSM-K16. Appl Microbiol Biotechnol 43:473–481
Kobayashi K et al (2003) Essential Bacillus subtilis genes. Proc Natl Acad Sci USA 100(8):4678–4683
Kobayashi T et al (2012) Identification and characterization of a novel polysaccharide deacetylase C(PdaC) from B. subtilis. J Biol Chem 287:9765–9776
Kodama T et al (2007a) Effect of the Bacillus subtilis spo0A mutation on cell wall lytic enzymes and extracellular proteases, and prevention of cell lysis. J Biosci Bioeng 103:13–21
Kodama T et al (2007b) Bacillus subtilis AprX involved in degradation a heterologous protein during the late stationary growth phase. J Biosci Bioeng 104:135–143
Kodama T et al (2011) Approaches for improving protein production in multiple protease-deficient Bacillus subtilis host strains. In: Petre M (ed) Advances in applied biotechnology. In Tech (Open access publisher)
Kunst F et al (1997) The complete genome sequence of the gram-positive bacterium Bacillus subtilis. Nature (Lond) 90:249–256
Liu S et al (2007) The accurate replacement of long genome region more than several hundred kilobases in Bacillus subtilis. Genes Genet Syst 82:9
Liu S et al (2008) Introduction of marker-free deletions in Bacillus subtilis using the AraR repressor and the ara promoter. Microbiology 154:2562–2570
Makarova KS et al (2001) Conservation of the binding site for the arginine repressor in all bacterial lineages. Genome Biol 2:RESEARCH0013
Manabe K et al (2011) Combined effect of improved cell yield and increased specific productivity enhances recombinant enzyme production in genome-reduced Bacillus subtilis strain MGB874. Appl Environ Microbiol 77:8370–8381
Manabe K et al (2012) High external pH enables more efficient secretion of alkaline α-amylase AmyK38 by Bacillus subtilis. Microb Cell Fact 11:74
Mizoguchi H et al (2007) Escherichia coli minimum genome factory. Biotechnol Appl Biochem 46:157–167
Mizoguchi H et al (2008) Superpositioning of deletions promotes growth of Escherichia coli with a reduced genome. DNA Res 15:277–284
Morimoto T et al (2008) Enhanced recombinant protein productivity by genome reduction in Bacillus subtilis. DNA Res 4:1–9
Morimoto T et al (2009) A new simple method to introduce marker-free deletions in the Bacillus subtilis genome. Genes Genet Syst 84:315–318
Morimoto T et al (2011a) A new simple method to introduce marker-free deletions in the Bacillus subtilis genome. Methods Mol Biol 765:345–358
Morimoto T et al (2011b) Simple method for introducing marker-free deletions in the Bacillus subtilis genome. In: Williams JA (ed) Strain engineering: methods and protocols. Humana Press, Totowa
Picossi S et al (2007) Molecular mechanism of the regulation of Bacillus subtilis gltAB expression by GltC. J Mol Biol 365:1298–1313
Posfai G et al (2006) Emergent properties of reduced-genome Escherichia coli. Science 312:1044–1046
Schumann W, Ehrlich SD, Ogasawara N (2000) Functional analysis of bacterial genes: a practice manual. Wiley, Chichester
Sekowska A et al (2001) Extracting biological information from DNA arrays: an unexpected link between arginine and methionine metabolism in Bacillus subtilis. Genome Biol 2:RESEARCH0019
Simonen M, Palva I (1993) Protein secretion in Bacillus species. Microbiol Rev 57:109–137
Tagami K et al (2012) Expression of a small (p)ppGpp synthetase, YwaC, in the (p)ppGpp(0) mutant of Bacillus subtilis triggers YvyD-dependent dimerization of ribosome. Microbiology Open 1:115–134
Westers H et al (2003) Genome engineering reveals large dispensable regions in Bacillus subtilis. Mol Biol Evol 20(12):2076–2090
Acknowledgments
The research and development of Bacillus subtilis RGF was conducted as part of a subproject, “Development of a Technology for the Creation of a Host Cell” included within the industrial technology project, “Development of Generic Technology for Production Process Starting Productive Function,” of the Ministry of Economy, Trade and Industry, entrusted by the New Energy and Industrial Technology Development Organization (NEDO), Japan. We are grateful to Dr. Shu Ishikawa and Dr. Taku Oshima (Nara Institute of Science and Technology) for help in transcriptome analysis, and Mr. Naoki Kondo and Ms. Eri Shimizu (Kao Corporation) for help in metaborome analysis. We also thank Dr. Tatsuro Fujio, Dr. Hideaki Nanamiya (Tokyo University), Dr. Hiroki Yamamoto (Shinshu University), Dr. Masayuki Hashimoto (Shinshu University), Dr. Tatsuya Fukushima (Shinshu University), Dr. Ken Kurokawa (Tokyo Institute of Technology), Dr. Mitsuhiro Itaya (Keio University), Dr. Hironori Niki (National Institute of Genetics), Dr. Hirofumi Yoshikawa (Tokyo University of Agriculture), Dr. Kei Asai (Saitama University), Dr. Hiroki Takahashi (Nara Institute of Science and Technology), Dr. Shigeo Inoue (Kao Corporation), and Dr. Toshiharu Numata (Kao Corporation) for valuable advice in conducting this study.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Japan
About this chapter
Cite this chapter
Ara, K. et al. (2014). Creation of Novel Technologies for Extracellular Protein Production Toward the Development of Bacillus subtilis Genome Factories. In: Anazawa, H., Shimizu, S. (eds) Microbial Production. Springer, Tokyo. https://doi.org/10.1007/978-4-431-54607-8_1
Download citation
DOI: https://doi.org/10.1007/978-4-431-54607-8_1
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-54606-1
Online ISBN: 978-4-431-54607-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)