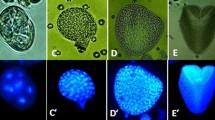
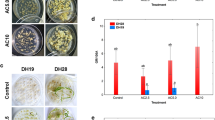
Abstract
Stress-induced microspore embryogenesis is an in vitro system in which the haploid microspore is reprogrammed by the application of stress treatments and, as a totipotent cell, enters into an embryogenesis pathway, producing double-haploid (DH) embryos and plants. DHs are important biotechnological tools in plant breeding programs, widely used by companies, as a source of new genetic variability, fixed in complete homozygous plants in only one generation step. However, their applications are limited in many crop and forest species due to low or null efficiency. Microspore embryogenesis is also a convenient system to analyze the cellular processes underlying cell reprogramming, totipotency acquisition, and embryogenesis. In the last years, investigation on stress-induced microspore embryogenesis has unveiled the involvement of several cellular processes like autophagy, cell death, epigenetic modifications, cell wall remodeling and auxin, whose role is not yet completely elucidated. In the present chapter, we review recent findings on the determinant factors that underlie stress-induced microspore embryogenesis induction and progression. These findings have provided new insights into the regulating mechanisms of microspore embryogenesis initiation and progression, opening up new intervention pathways through pharmacological treatments with small bioactive compounds (autophagy, epigenetic, and auxin modulators), very promising to enhance in vitro embryo production yield for breeding programs of crop and forest species, especially for those recalcitrant.
Communicated by Maria-Carmen Risueño
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Avin-Wittenberg T, Baluska F, Bozhkov PV, Elander PH, Fernie AR, Galili G, Hassan A, Hofius D, Isono E, Le Bars R, Masclaux-Daubresse C, Minina EA, Peled-Zehavi H, Coll NS, Sandalio LM, Satiat-Jeunemaitre B, Sirko A, Testillano PS, Batoko H (2018) Autophagy-related approaches for improving nutrient use efficiency and crop yield protection. J Exp Bot 69(6):1335–1353. https://doi.org/10.1093/jxb/ery069
Bárány I, González-Melendi P, Fadón B, Mitykó J, Risueño MC, Testillano PS (2005) Microspore-derived embryogenesis in pepper (Capsicum annuum L.): subcellular rearrangements through development. Biol Cell 97(9):709–722. https://doi.org/10.1042/BC20040142
Bárány I, Fadón B, Risueño MC, Testillano PS (2010) Cell wall components and pectin esterification levels as markers of proliferation and differentiation events during pollen development and pollen embryogenesis in Capsicum annuum L. J Exp Bot 61(4):1159–1175. https://doi.org/10.1093/jxb/erp392
Bárány I, Berenguer E, Solís MT, Pérez-Pérez Y, Santamaria ME, Crespo JL, Risueño MC, Diaz I, Testillano PS (2018) Autophagy is activated and involved in cell death with participation of cathepsins during stress-induced microspore embryogenesis in barley. J Exp Bot 69(6):1387–1402. https://doi.org/10.1093/jxb/erx455
Berenguer E, Bárány I, Solís MT, Pérez-Pérez Y, Risueño MC, Testillano PS (2017a) Inhibition of histone H3K9 methylation by BIX-01294 promotes stress-induced microspore totipotency and enhances embryogenesis initiation. Front Plant Sci 8:1161. https://doi.org/10.3389/fpls.2017.01161
Berenguer E, Solís MT; Pérez-Pérez Y, Minina Y, Risueño MC, Bozhkov P, Testillano PS (2017b) Metacaspases and autophagy are induced in microspore embryogenesis of Brassica napus. In: Proceedings 2nd meeting WG3 transautophagy cost action CA15138, Madrid, 23–24 Mar 2017
Borderies G, le Bechec M, Rossignol M, Lafitte C, Le Deunff E, Beckert M, Dumas C, Elisabeth MR (2004) Characterization of proteins secreted during maize microspore culture: arabinogalactan proteins (AGPs) stimulate embryo development. Eur J Cell Biol 83(5):205–212
Braybrook SA, Peaucelle A (2013) Mechano-chemical aspects of organ formation in Arabidopsis thaliana: the relationship between auxin and pectin. PLoS One 8(3):e57813. https://doi.org/10.1371/journal.pone.0057813
Bueno MA, Gómez A, Boscaiu M, Manzanera JA, Vicente O (1997) Stress-induced formation of haploid plants through anther culture in cork oak (Quercus suber). Physiol Plant 99:335–341
Chapman A, Blervacq AS, Vasseur J, Hilbert JL (2000) Arabinogalactan-proteins in Cichorium somatic embryogenesis: effect of β-glucosyl Yariv reagent and epitope localisation during embryo development. Planta 211(3):305–314
Chiancone B, Karasawa MM, Gianguzzi V, Abdelgalel AM, Bárány I, Testillano PS, Marinoni DT, Botta R, Germaná MA (2015) Early embryo achievement through isolated microspore culture in Citrus clementina Hort. ex Tan., cvs. ‘Monreal Rosso’ and ‘Nules’. Front Plant Sci 6:413. https://doi.org/10.3389/fpls.2015.00413
Corredoira E, Cano V, Bárány I, Solís MT, Rodríguez H, Vieitez AM, Risueño MC, Testillano PS (2017) Initiation of leaf somatic embryogenesis involves high pectin esterification, auxin accumulation and DNA demethylation in Quercus alba. J Plant Physiol 213:42–54. https://doi.org/10.1016/j.jplph.2017.02.012
Diaz-Mendoza M, Dominguez-Figueroa JD, Velasco-Arroyo B, Cambra I, Gonzalez-Melendi P, Lopez-Gonzalvez A, Garcia A, Hensel G, Kumlehn J, Diaz I, Martinez M (2016) HvPap-1 C1A protease and HvCPI-2 cystatin contribute to barley grain filling and germination. Plant Physiol 170(4):2511–2524. https://doi.org/10.1104/pp.15.01944
Earley KW, Shook MS, Brower-Toland B, Hicks L, Pikaard CS (2007) In vitro specificities of Arabidopsis co-activator histone acetyltransferases: implications for histone hyperacetylation in gene activation. Plant J 52(4):615–626. https://doi.org/10.1111/j.1365-313X.2007.03264.x
Elhiti M, Stasolla C (2011) Ectopic expression of the Brassica SHOOTMERISTEMLESS attenuates the deleterious effects of the auxin transport inhibitor TIBA on somatic embryo number and morphology. Plant Sci 180(2):383–390. https://doi.org/10.1016/j.plantsci.2010.10.014
El-Tantawy AA, Solís MT, Da Costa ML, Coimbra S, Risueño MC, Testillano PS (2013) Arabinogalactan protein profiles and distribution patterns during microspore embryogenesis and pollen development in Brassica napus. Plant Reprod 26(3):231–243. https://doi.org/10.1007/s00497-013-0217-8
El-Tantawy AA, Solís MT, Risueño MC, Testillano PS (2014) Changes in DNA methylation levels and nuclear distribution patterns after microspore reprogramming to embryogenesis in barley. Cytogenet Genome Res 143(1–3):200–208. https://doi.org/10.1159/000365232
Germaná MA (2009) Haploids and doubled haploids in fruit trees. In: Advances in haploid production in higher plants. Springer, Dordrecht, pp 241–263. https://doi.org/10.1007/978-1-4020-8854-4_21
Hofius D, Li L, Hafren A, Coll NS (2017) Autophagy as an emerging arena for plant-pathogen interactions. Curr Opin Plant Biol 38:117–123. https://doi.org/10.1016/j.pbi.2017.04.017
Kouzarides T (2007) Chromatin modifications and their function. Cell 128(4):693–705. https://doi.org/10.1016/j.cell.2007.02.005
Li H, Soriano M, Cordewener J, Muino JM, Riksen T, Fukuoka H, Angenent GC, Boutilier K (2014) The histone deacetylase inhibitor trichostatin a promotes totipotency in the male gametophyte. Plant Cell 26(1):195–209. https://doi.org/10.1105/tpc.113.116491
Liu C, Lu F, Cui X, Cao X (2010) Histone methylation in higher plants. Annu Rev Plant Biol 61:395–420. https://doi.org/10.1146/annurev.arplant.043008.091939
Liu X, Luo M, Wu K (2012) Epigenetic interplay of histone modifications and DNA methylation mediated by HDA6. Plant Signal Behav 7(6):633–635. https://doi.org/10.4161/psb.19994
Maluszynski M, Kasha K, Forster B, Szarejko I (eds) (2003) Doubled haploid production in crop plants: a manual. Kluwer, Dordrecht
Masclaux-Daubresse C, Chen Q, Have M (2017) Regulation of nutrient recycling via autophagy. Curr Opin Plant Biol 39:8–17. https://doi.org/10.1016/j.pbi.2017.05.001
Miguel C, Marum L (2011) An epigenetic view of plant cells cultured in vitro: somaclonal variation and beyond. J Exp Bot 62(11):3713–3725. https://doi.org/10.1093/jxb/err155
Minina EA, Filonova LH, Fukada K, Savenkov EI, Gogvadze V, Clapham D, Sanchez-Vera V, Suarez MF, Zhivotovsky B, Daniel G, Smertenko A, Bozhkov PV (2013) Autophagy and metacaspase determine the mode of cell death in plants. J Cell Biol 203(6):917–927. https://doi.org/10.1083/jcb.201307082
Minina EA, Smertenko AP, Bozhkov PV (2014) Vacuolar cell death in plants: metacaspase releases the brakes on autophagy. Autophagy 10(5):928–929. https://doi.org/10.4161/auto.28236
Moreno-Risueño MA, Van Norman JM, Moreno A, Zhang J, Ahnert SE, Benfey PN (2010) Oscillating gene expression determines competence for periodic Arabidopsis root branching. Science 329(5997):1306–1311. https://doi.org/10.1126/science.1191937
Murovec J, Bohanec B (2012) Haploids and doubled haploids in plant breeding. In: Abdurakhmonov I (ed) Plant breeding. InTech http://www.intechopen.com/books/plant-breeding/haploids-and-doubled-haploids-in-plant-breeding
Pechan PM, Keller WA (1988) Identification of potentially embryogenic microspores in Brassica napus. Physiol Plant 74(2):377–384. https://doi.org/10.1111/j.1399-3054.1988.tb00646.x
Pelloux J, Rusterucci C, Mellerowicz EJ (2007) New insights into pectin methylesterase structure and function. Trends Plant Sci 12(6):267–277. https://doi.org/10.1016/j.tplants.2007.04.001
Pérez-Pérez Y, Solís MT, El-Tantawy A, Pitarch M, Gómez-Cadenas A, Risueño MC, Testillano PS (2017) Auxin biosynthesis and transprot are required for stress-induced microspore embryogenesis whereas the gametophytic pathway progresses with decreasing auxin in rapeseed and barley. In: Testillano PS, Martín MC, Benito E, Risueño MC (eds) Plants in vitro for the future, Proceedings of the XII congress of the Spanish Society of Plant in vitro culture. FGUA Publishing, Madrid
Pérez-Pérez Y, Bárány I, Berenguer E, Carneros E, Risueño MC, Testillano PS (2018a) Small compounds modulating autophagy and protease activities reduce cell death and improve stress-induced microspore embryogenesis initiation in rapeseed and barley. Plant Signal Behav. In revision
Pérez-Pérez Y, Carneros E, Bárány I, Pintos B, Gómez-Garay A, Risueño MC, Testillano PS (2018b) Changes in pectin esterification and AGPs indicate the cell wall remodeling during somatic embryogenesis of Quercus suber. Front Plant Sci. In revision
Petrasek J, Friml J (2009) Auxin transport routes in plant development. Development 136(16):2675–2688. https://doi.org/10.1242/dev.030353
Portillo L, Olmedilla A, Santacruz-Ruvalcaba F (2012) Cellular and molecular changes associated with somatic embryogenesis induction in Agave tequilana. Protoplasma 249(4):1101–1107. https://doi.org/10.1007/s00709-011-0354-6
Prem D, Solís MT, Bárány I, Rodríguez-Sánz H, Risueño MC, Testillano PS (2012) A new microspore embryogenesis system under low temperature which mimics zygotic embryogenesis initials, expresses auxin and efficiently regenerates doubled-haploid plants in Brassica napus. BMC Plant Biol 12:127. https://doi.org/10.1186/1471-2229-12-127
Rodríguez-Sanz H, Manzanera JA, Solís MT, Gómez-Garay A, Pintos B, Risueño MC, Testillano PS (2014a) Early markers are present in both embryogenesis pathways from microspores and immature zygotic embryos in cork oak, Quercus suber L. BMC Plant Biol 14(1):224. https://doi.org/10.1186/s12870-014-0224-4
Rodríguez-Sanz H, Moreno-Romero J, Solís MT, Kohler C, Risueño MC, Testillano PS (2014b) Changes in histone methylation and acetylation during microspore reprogramming to embryogenesis occur concomitantly with Bn HKMT and Bn HAT expression and are associated with cell totipotency, proliferation, and differentiation in Brassica napus. Cytogenet Genome Res 143:209–218. https://doi.org/10.1159/000365261
Rodríguez-Sanz H, Solís MT, López MF, Gómez-Cadenas A, Risueño MC, Testillano PS (2015) Auxin biosynthesis, accumulation, action and transport are involved in stress-induced microspore embryogenesis initiation and progression in Brassica napus. Plant Cell Physiol 56(7):1401–1417. https://doi.org/10.1093/pcp/pcv058
Rodríguez-Serrano M, Bárány I, Prem D, Coronado M-J, Risueño MC, Testillano PS (2012) NO, ROS, and cell death associated with caspase-like activity increase in stress-induced microspore embryogenesis of barley. J Exp Bot 63(5):2007–2024. https://doi.org/10.1093/jxb/err400
Seguí-Simarro JM, Bárány I, Suárez R, Fadón B, Testillano P, Risueño MC (2006) Nuclear bodies domain changes with microspore reprogramming to embryogenesis. Eur J Histochem 50(1):35–44
Seguí-Simarro JM, Corral-Martínez P, Corredor E, Raska I, Testillano PS, Risueño MC (2011) A change of developmental program induces the remodeling of the interchromatin domain during microspore embryogenesis in Brassica napus L. J Plant Physiol 168(8):746–757. https://doi.org/10.1016/j.jplph.2010.10.014
Seifert GJ, Roberts K (2007) The biology of arabinogalactan proteins. Annu Rev Plant Biol 58:137–161. https://doi.org/10.1146/annurev.arplant.58.032806.103801
Solís MT, Pintos B, Prado MJ, Bueno MA, Raska I, Risueño MC, Testillano PS (2008) Early markers of in vitro microspore reprogramming to embryogenesis in olive (Olea europaea L.). Plant Sci 174(6):597–605. https://doi.org/10.1016/j.plantsci.2008.03.014
Solís MT, Rodríguez-Serrano M, Meijón M, Canal MJ, Cifuentes A, Risueño MC, Testillano PS (2012) DNA methylation dynamics and MET1a-like gene expression changes during stress-induced pollen reprogramming to embryogenesis. J Exp Bot 63(18):6431–6444. https://doi.org/10.1093/jxb/ers298
Solís MT, El-Tantawy AA, Cano V, Risueño MC, Testillano PS (2015) 5-Azacytidine promotes microspore embryogenesis initiation by decreasing global DNA methylation, but prevents subsequent embryo development in rapeseed and barley. Front Plant Sci 6:472. https://doi.org/10.3389/fpls.2015.00472
Solís MT, Berenguer E, Risueño MC, Testillano PS (2016) BnPME is progressively induced after microspore reprogramming to embryogenesis, correlating with pectin de-esterification and cell differentiation in Brassica napus. BMC Plant Biol 16(1):176. https://doi.org/10.1186/s12870-016-0863-8
Tang XC, He YQ, Wang Y, Sun MX (2006) The role of arabinogalactan proteins binding to Yariv reagents in the initiation, cell developmental fate, and maintenance of microspore embryogenesis in Brassica napus L. cv. Topas. J Exp Bot 57(11):2639–2650. https://doi.org/10.1093/jxb/erl027
Testillano PS, Risueño MC (2009) Tracking gene and protein expression during microspore embryogenesis by confocal laser scanning microscopy. In: Touraev A, Forster BP, Mohan Jain S (eds) Advances in haploid production in higher plants. Springer, Dordrecht, pp 339–347
Testillano PS, Rodríguez MD (2012) Cell biology of plant development and adaptation. In: Becerra J, Santos-Ruíz L (eds) Hot topics in cell biology. Biohealthcare, Oxford, pp 61–62
Testillano PS, Coronado MJ, Seguí JM, Domenech J, González-Melendi P, Raska I, Risueño MC (2000) Defined nuclear changes accompany the reprogramming of the microspore to embryogenesis. J Struct Biol 129(2–3):223–232
Testillano PS, Ramírez C, Domenech J, Coronado MJ, Vergne P, Matthys-Rochon E, Risueño MC (2002) Young microspore-derived maize embryos show two domains with defined features also present in zygotic embryogenesis. Int J Dev Biol 46(8):1035–1047
Testillano PS, González-Melendi P, Coronado MJ, Seguí-Simarro JM, Moreno-Risueño MA, Risueño MC (2005) Differentiating plant cells switched to proliferation remodel the functional organization of nuclear domains. Cytogenet Genome Res 109(1–3):166–174. https://doi.org/10.1159/000082396
Testillano PS, Solís MT, Riseuño MC (2013) The 5-methyl-deoxy-cytidine (5mdC) localization to reveal in situ the dynamics of DNA methylation chromatin pattern in a variety of plant organ and tissue cells during development. Physiol Plant 149(1):104–113. https://doi.org/10.1111/ppl.12015
Testillano PS, Gómez-Garay A, Pintos B, Risueño MC (2018a) Somatic embryogenesis from immature zygotic embryos in Quercus suber L. In: Loyola-Vargas V, Ochoa-Alejo N (eds) Plant cell culture protocols, Methods in molecular biology, vol 1815. Springer, Berlin. https://doi.org/10.1007/978-1-4939-8594-4_16
Testillano PS, Pintos B, Gómez-Garay A, Risueño MC (2018b) Stress-induced microspore embryogenesis by anther culture of Quercus suber L. In: Jain SM, Gupta P (eds) Step wise protocols for somatic embryogenesis in important Woody plants, Forestry sciences, vol 84. Springer, Berlin. https://doi.org/10.1007/978-3-319-89483-6_7
Thompson HJM, Knox JP (1998) Stage-specific responses of embryogenic carrot cell suspension cultures to arabinogalactan protein-binding β-glucosyl Yariv reagent. Planta 205:32–38
van der Hoorn RAL, Jones JD (2004) The plant proteolytic machinery and its role in defence. Curr Opin Plant Biol 7(4):400–407. https://doi.org/10.1016/j.pbi.2004.04.003
van der Hoorn RAL, Rivas S (2018) Unravelling the mode of action of plant proteases. New Phytol 218(3):879–881. https://doi.org/10.1111/nph.15156
van Hengel AJ, Tadesse Z, Immerzeel P, Schols H, van Kammen A, de Vries SC (2001) N-acetylglucosamine and glucosamine-containing arabinogalactan proteins control somatic embryogenesis. Plant Physiol 125(4):1880–1890
Velasco-Arroyo B, Diaz-Mendoza M, Gandullo J, Gonzalez-Melendi P, Santamaria ME, Dominguez-Figueroa JD, Hensel G, Martinez M, Kumlehn J, Diaz I (2016) HvPap-1 C1A protease actively participates in barley proteolysis mediated by abiotic stresses. J Exp Bot 67(14):4297–4310. https://doi.org/10.1093/jxb/erw212
Verdeil JL, Alemanno L, Niemenak N, Tranbarger TJ (2007) Pluripotent versus totipotent plant stem cells: dependence versus autonomy? Trends Plant Sci 12(6):245–252. https://doi.org/10.1016/j.tplants.2007.04.002
Willats WGT, McCartney L, Mackie W, Knox JP (2001) Pectin: cell biology and prospects for functional analysis. Plant Mol Biol 47(1–2):9–27. https://doi.org/10.1023/a:1010662911148
Willats WGT, Knox P, Mikkelsen JD (2006) Pectin: new insights into an old polymer are starting to gel. Trends Food Sci Technol 17(3):97–104. https://doi.org/10.1016/j.tifs.2005.10.008
Xu CR, Liu C, Wang YL, Li LC, Chen WQ, Xu ZH, Bai SN (2005) Histone acetylation affects expression of cellular patterning genes in the Arabidopsis root epidermis. Proc Natl Acad Sci U S A 102(40):14469–14474. https://doi.org/10.1073/pnas.0503143102
Yuan S, Su Y, Liu Y, Fang Z, Yang L, Zhuang M, Zhang Y, Sun P (2012) Effects of pH, MES, arabinogalactan-proteins on microspore cultures in white cabbage. Plant Cell Tiss Org Cult 110:69–76. https://doi.org/10.1007/s11240-012-0131-z
Zur I, Dubas E, Golemiec E, Szechynska-Hebda M, Golebiowska G, Wedzony M (2009) Stress-related variation in antioxidative enzymes activity and cell metabolism efficiency associated with embryogenesis induction in isolated microspore culture of triticale (x Triticosecale Wittm.). Plant Cell Rep 28(8):1279–1287. https://doi.org/10.1007/s00299-009-0730-2
Acknowledgments
Work supported by projects (AGL2014-52028-R and AGL2017-82447-R) funded by Spanish Ministry of Economy and Competitiveness (MINECO) and European Regional Development Fund (ERDF/FEDER), and by the COST Action TRANSAUTOPHAGY (CA15138), European Network of Multidisciplinary Research and Translation of Autophagy Knowledge.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Testillano, P.S. (2018). Stress-Induced Microspore Embryogenesis in Crop Plants: Cell Totipotency Acquisition and Embryo Development. In: Cánovas, F., Lüttge, U., Leuschner, C., Risueño, MC. (eds) Progress in Botany Vol. 81. Progress in Botany, vol 81. Springer, Cham. https://doi.org/10.1007/124_2018_24
Download citation
DOI: https://doi.org/10.1007/124_2018_24
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-36326-0
Online ISBN: 978-3-030-36327-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)