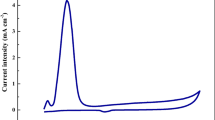
Abstract
To detect lead ions using electrochemical voltammetric analysis, Infrared Photo-Diode Electrode (IPDE) was applied via cyclic and square wave stripping voltammetry. Lead ions were deposited at 0.5 V (versus Ag/AgCl) accumulation potential. Instrumental measurements systems were made based on a simple and compact detection system. The stripping voltammetric and cyclic voltammetric optimal parameters were searched. The results yielded a cyclic range of 40∼240 mgl−1 Pb(II) and a square wave stripping working range of 0.5∼5.00 mgl−1 Pb(II). The relative standard deviation at 2 and 4 mgl−1 Pb(II) was 0.04% and 0.02% (n = 15), respectively, using the stripping voltammetric conditions. The detection limit was found to be 0.05 mgl−1 with a 40 sec preconcentration time. Analytical interference ions were also evaluated. The proposed method was applied to determine lead ions in various samples.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Chau, Y.K. and Yang, F. (1997). A novel technique for direct derivatization of ionic organotin and alkyl-lead compounds in sediment: Simultaneous determination of organotin and alkyl-lead compounds in sediment by gas chromatographyplasma atomic emission (GC-AED) dual-channel detection. Applied Organometallic Chemistry, 11, 851–858.
Christian, D.G., Adam, M.S., Greg, P.S., Jansuz, K., Laren, M.T. and Jin, Z.Z. (2003). Optical and dlectrochemical characterization of poly(3-undecyl-2,2-bithiophene)in thin film solid state TiO2 photovoltaic solar cells. Synthetic Metals, 132, 197–204.
Cidalia, M.S.B., Rui, A.R.B., Maria, L.S.G. and Ana, M.M. (2001). Dissolved Pb(II) speciation in a polluted river. Electroanalysis, 13, 1497–1540.
Cristina, M.B. and Margarida, M.C. (2001). Speciation of inorganic arsenic in natural waters by square-wave cathodic stripping voltammetry. Electroanalysis, 13, 1098–1104.
Dong, X., Qundong, S., Suyang, Z. and Xiqun, J. (2003). Synthesis and characterization of novel soluble alternating copoly(phenylene vinylene) derivative for light-emitting electrochemical cell. Journal of Applied Polymer Science, 88, 1350–1356.
El-Hefnawey, G.B., El-Hallag, I.S., Ghoneim, E.M. and Ghoneim, M.M. (2004). Voltammetric behavior and quantification of the sedative-hypnotic drug chlordiazepoxide in bulk form, pharmaceutical formulation and human serum at a mercury electrode. Journal of Pharmaceutical and Biomedical Analysis, 34, 75–86.
Emanuele, S., Alessandra, L., Lucia, P., Maria, R.S. and Giovanni, F. (2003). Blood and hair lead levels in boys and girls living in two Sardinian towns at different risks of lead pollution. Ecotoxicology and Environmental Safety, 55, 293–299.
Eninari, V.Z., Parvitte, B., Courtois, D., Kapitanov, V.A. and Yu, N.P. (2003). Methane detection on the sub-ppm level with a near-infrared diode laser photoacoustic sensor. Technology, 44, 253–261.
Eric, H.C. and Khalil, J.S. (1997). Retrospective analysis of anthropogenic inputs of lead and other heavy metals to the hawaiian sedimentary environment. Applied Organometallic Chemistry, 11, 415–437.
Gagliardi, G. and Gianfrani, L. (2002). Trace-gas analysis using diode lasers in the near-IRand long-path techniques. Optics and Lasers in Engineering, 37, 509–520.
Georges, D. (2002). Balloon-borne near-infrared diode laser spectroscopy for in situ measurements of atmospheric CH4 and H2O. Spectrochimica Acta Part A, 57, 1855–1863.
Greg, A.G. and Alexander, F.H. (2001). Effects of recording media composition on the responses of Nafion-coated carbon fiber microelectrodes measured using high-speed chronoamperometry. Journal of Neuroscience Methods, 109, 13–21. Hai- bo, Q., David, T., Paul, W.S. and Michael, J.P. (2000). Dynamic frequency stabilization of infrared diode laser for kinetic studies. Chemical Physics Letters, 322, 57–64.
Huaisheng, W., Aimei, Z., Hui, C., Daojie, L. and Renmin, L. (1998). Adsorptive stripping voltammetric determination of phenol at an electrochemically pretreated carbon-paste electrode with solid paraffin as a binder. Microchimical Journal, 59, 448–456.
Jahan, B.R., Reza, O. and Abolfazl, K. (2001). Carbon paste electrode spiked with ferrocene carboxylic acid and its application to the electrocatalytic determination of ascorbic acid. Journal of Electroanalytical Chemistry, 515, 45–51.
Jeong, W.C., Yun, S.N., Byung, K.O., Won, H.L. and Masamichi, F. (2001). Artificial molecular photodiode consisting of GFP/viologen hetero-thin film. Synthetic Metals, 117, 241–243.
Jing, L., Jun, Z., Lili, Z., Tao, C., Guangyun, X. and Xiwen, H. (2001). The effects of multi minerals on susceptibility to lead tpxicity in rats. J. Toxicol. Pub. Health, 17, 135–138.
Jose, G.H., Pedro, H., Yolanda, B. and Lucas, H. (2001). Determination of linuron in soil by stripping voltammetry with a carbon fiber microelectrode. Talanta, 54, 1077–1085.
Joseph, W., Beatriz, S., Suw, Y.L., Jianmin, L. and Jose, M.P. (2001). Determination of micromolar bromate concentrations by adsorptive-catalytic stripping votammetry of the molybdenum-3-methoxy-4-hydroxymandelic acid complex 2001. Talanta, 54, 147–151.
Jyh, M.Z., Hsieh, H.C. and Govindasamy, I. (1999). Selective voltammetric determination of lead(II) on partially quaternized poly(4-vinylpyridine)ymercury film electrodes. Electroanalysis, 11, 108–113.
Kai, M.A., Horst, S., Robert, S., Franz, S., Farideh, M., Alexander, P., Gunther, L., Kai, C.M and Jurgen, O.B. (2004). Blue-green light emitting poly(phenylenevinylene) derivatives as candidates for polymer LEDs: Synthesis and characterization. Chem. Phys., 205, 1840–1850.
Kevin, A., Meredith, H., Lynn, H.T., Judy, D., Lee S. and Peter, F.B. (1998). Field Investigation of On-Site Techniques for the Measurement of Lead in Paint Films. Field Analytical Chemistry And Technology, 2, 39–50.
Khiena, Z.B., Inna, V.K., Elena, G.M., and Stanislav, I.P., Yuri, G.M. and Andrei, E.V. (2001). Small-size sensors for the in-field stripping voltammetric analysis of water. Field Analytical Chemistry and Technology, 5, 260–271.
Kozelka, P.B., Sanudo, W., Flegalc, A.R. and Bruland, E. (1997). Physico-chemical speciation of lead in south san francisco bay. Coastal and Shelf Science, 44, 649–658.
Kwang, S.P., Pil, S.C., Sang, H.L., Chul, W.L., Ji, S.R., Sung, S.C., Hong, I.R. and Dong, I.C (1998). Endocrine disrupting effects of lead on the ontogeny of oryzias latips. J. Toxicol. Pub. Health, 14, 379–384.
Lauralynn, T., Robert, L.J., Lorna, K., James, A.D., Kevin, A. and Wayne, T.S. (2001). Evaluation of a portable blood lead analyzer with occupationally exposed populations. American Journal of ndustrial Medicine, 40, 354–362.
Lung, C.C. and Yin, L.H. (2004). High reliability GaN-based light-emitting diodes with photo-enhanced wet etching. Solid-State Electronics, 48, 1239–1242.
Maria, S.D., Miriam, E.P. and Beatriz, S.F.B. (2004). A sensitive spectrophotometric method for lead determination by flow injection analysis with on-line preconcentration. Talanta, 63, 405–409.
Mohammed, B., Jean, P.P., Joel, J.E.M., Francoise, S.S. and Raigna, A.S. (2002). A thienylene-phenylene copolymer with di(ethylene oxide) side chains and its use in light emitting diodes. Synthetic Metals, 126, 241–244.
Ouassim, G., Gwenael, C. and Daniel, B. (2004). Chemical reactivity of 4 bromophenyl modified glassy carbon electrode. Electrochemistry Communications, 6, 254–258.
Prinya, M., Saisunee, L., Yuthsak, V. and Boonsom, L. (2002). Design and fabrication of a low-cost flow-through cell for the determination of acetaminophen in pharmaceutical formulations by flow injection cyclic voltammetry. Talanta, 58, 1221–1234.
Priya, V.J., Vijaykumar, S.I. and Ashwini, K.S. (2001). Voltammetric behavior of a-tocopherol and its determination using surfactant +ethanol +water and surfactant +acetonitrile +water mixed solvent systems. Analytica Chimica Acta., 441, 201–206.
Robert, J.R., Janie, L.G., James, A.D., Martin, R.P. and William, E.H. (1999). Blood lead levels among children of lead-exposed workers: A meta-analysis. American Journal of Industrial Medicine, 36, 475–481.
Scarponi, G., Barbante, C., Turetta, C., Gambaro, A. and Cescon, P. (1997). Chemical contamination of antarctic snow: The case of lead. Microchemical Journal, 55, 24–32.
Schlosser, E., Fernholz, T., Teichert, H. and Ebert, V. (2002). In situ detection of potassium atoms in high-temperature coal-combustion systems using near-infrared-diode lasers. Spectrochimica Acta Part A, 58, 2347–2359.
Sung, O.J., Youn, G.K., Hyung, S.K., Yun, H.K., Chang, L.L., Jang, J.K., Sung, K.L. and Soon, K.K. (2004). Effect of substitution of methyl groups on the luminescence performance of Ir III complexes: Preparation, structures, electrochemistry, photophysical properties and their applications in organic light-emitting diodes (OLEDs). Eur. J. Inorg. Chem., 3415–3423.
Suw, Y.L. (2008). Voltammetric Assay of Mercury ion in fish Kidneys. Toxicol. Res., 24, 23–28.
Suw, Y.L., Duck, H.K. and Myung, H.K. (2002). Square-wave cathodic stripping voltammetric analysis of RDX using mercury-film plated glassy carbon electrode. Talanta, 58, 919–926.
Suw, Y.L., Young, S.J., Myung, H.K., In, K.H., Woon, W.J. and Hyun, S.K. (2004). Measuring caffeine using a simple graphite pencil electrode with square wave anodic stripping voltammetry. Microchim, Acta, 146, 207–213.
Xueji, Z., Bozidar, O., Marjan, R., Marko, K. and Robert, Z. (1999). Cathophoresis paint insulated carbon ®bre ultramicro disc electrode and its application to in vivo amperometric monitoring of quantal secretion from single rat melanotrophs. Analytica Chimica Acta, 378, 135–143.
Yamamoto, K. and Yoshida, N. (2002). High-precision isotopic ratio measurement system for methane (12CH3 D12CH4, 13CH4/12CH4) by using near-infrared diode laser absorption spectroscopy. Spectrochimica Acta Part A, 58, 2699–2707.
Yu, C.T., Barry, A.C., Katherine, H., John, S.F., Frank, M. and Richard, G.C. (2001). Selective voltammetric determination of lead(II) on partially quaternized poly(4-vinylpyridine)ymercury film electrodes. Electroanalysis, 13, 831–835.
Yuwu, C., Jianping, D., Zheng, F.Z., Hongqinq. and C., Guonan, C. (2003). A Study on the electrochemical and electrochemiluminescent behavior of homogentisic acid at carbon electrodes. Electroanalysis, 15, 208–218.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ly, S.Y., Lee, H.K., Kwak, K.J. et al. Analysis of Lead Ions in a Waste Solution Using Infrared Photo-Diode Electrode. Toxicol Res. 24, 227–233 (2008). https://doi.org/10.5487/TR.2008.24.3.227
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.5487/TR.2008.24.3.227