Abstract
Runella slithyformis Larkin and Williams 1978 is the type species of the genus Runella, which belongs to the Cytophagaceae, a family that was only recently classified to the order Cytophagales in the class Cytophagia. The species is of interest because it is able to grow at temperatures as low as 4°C. This is the first completed genome sequence of a member of the genus Runella and the sixth sequence from the family Cytophagaceae. The 6,919,729 bp long genome consists of a 6.6 Mbp circular genome and five circular plasmids of 38.8 to 107.0 kbp length, harboring a total of 5,974 protein-coding and 51 RNA genes and is a part of the Genomic Encyclopedia of Bacteria and Archaea project.
Similar content being viewed by others
Introduction
Strain LSU 4T (= DSM 19594 = ATCC 29530 = NCIMB 11436) is the type strain of the species Runella slithyformis, which is the type species of its genus Runella [1,2]. The genus currently consists of four validly named species [3]. The genus name is derived from ‘rune’, a runic letter and the Latin diminutive ending ‘ella’, yielding the Neo-Latin word ‘Runella’, meaning ‘that which resembles figures of the runic alphabet’ [3]. The species epithet is derived from slithy, a nonsense word from Lewis Carroll’s Jabberwocky for a fictional organism that is ‘slithy’ and the Latin word ‘suffix’ meaning ‘-like, in the shape of’, yielding the Neo-Latin word ‘slithyformis’ meaning ‘slithy in form’ [3]. R. slithyformis strain LSU 4T was isolated from University Lake near Baton Rouge, Louisiana, USA, and described by Larkin and Williams in 1978 [1]. Another strain of R. slithyformis, termed strain 6, was isolated from Elbow Bayou near Baton Rouge [1]. Members of the genus Runella colonize diverse environmental habitats, preferentially aquatic ecosystems, including water bodies in Baton Rouge [1], a wastewater treatment plant in South-Korea [4], environmental water samples and their biofilms in Japan [5], and an activated sludge process involved in enhanced biological removal of phosphor in Korea [6]. Another species of this genus was also isolated from the stems of surface-sterilized maize [7]. Here we present a summary classification and a set of features for R. slithyformis strain LSU 4T, together with the description of the complete finished genome sequencing and annotation.
Classification and features
A representative genomic 16S rRNA sequence of R. slithyformis LSU 4T was compared using NCBI BLAST [8,9] under default settings (e.g., considering only the high-scoring segment pairs (HSPs) from the best 250 hits) with the most recent release of the Greengenes database [10] and the relative frequencies of taxa and keywords (reduced to their stem [11]) were determined, weighted by BLAST scores. The most frequently occurring genera were Runella (31.0%), Dyadobacter (30.3%), Cytophaga (13.7%), Cyclobacterium (7.5%) and Algoriphagus (4.0%) (51 hits in total). Regarding the single hit to sequences from members of the species, the average identity within HSPs was 99.2%, whereas the average coverage by HSPs was 96.9%. Regarding the two hits to sequences from other members of the genus, the average identity within HSPs was 95.0%, whereas the average coverage by HSPs was 91.1%. Among all other species, the one yielding the highest score was R. zeae (NR_025004), which corresponded to an identity of 95.0% and an HSP coverage of 91.1%. (Note that the Greengenes database uses the INSDC (= EMBL/NCBI/DDBJ) annotation, which is not an authoritative source for nomenclature or classification.) The highest-scoring environmental sequence was GQ480089 (‘changes during sewage treated process activated sludge wastewater treatment plant clone BXHB50’), which showed an identity of 96.6% and an HSP coverage of 98.0%. The most frequently occurring keywords within the labels of all environmental samples which yielded hits were ‘skin’ (5.5%), ‘soil’ (2.1%), ‘sludg’ (2.0%), ‘biofilm’ (1.7%) and ‘forearm, volar’ (1.7%) (199 hits in total). While few of these keywords fit the aquatic and sludge environments from which strain LSU 4T originated, the majority of the hits point to human and even soil, which were, until now, not considered as habitats for R. slithyformis. However, environmental samples which yielded hits of a higher score than the highest scoring species were not found.
Figure 1 shows the phylogenetic neighborhood of R. slithyformis LSU 4T in a 16S rRNA based tree. The sequences of the two identical 16S rRNA gene copies in the genome do not differ from the previously published 16S rRNA sequence (M62786), which contains 13 ambiguous base calls.
Phylogenetic tree highlighting the position of R. slithyformis relative to the type strains of the type species of the other genera within the family Cytophagaceae. The tree was inferred from 1,330 aligned characters [12,13] of the 16S rRNA gene sequence under the maximum likelihood (ML) criterion [14]. Rooting was done initially using the midpoint method [15] and then checked for its agreement with the current classification (Table 1). The branches are scaled in terms of the expected number of substitutions per site. Numbers adjacent to the branches are support values from 400 ML bootstrap replicates [16] (left) and from 1,000 maximum parsimony bootstrap replicates [17] (right) if larger than 60%. Lineages with type strain genome sequencing projects registered in GOLD [18] are labeled with one asterisk, those also listed as ‘Complete and Published’ with two asterisks [19–22].
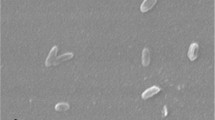
The cells of strain LSU 4T are generally curved rods, with the degree of curvature of individual cells within a culture varying from nearly straight to crescent shape. Cell diameter varies from 0.5 to 0.9 µm, and the length from 2.0 to 3.0 µm [1]. With its curved rod shape, strain LSU 4T differs from other members of the genus, such as R. limosa which has long rods while R. zeae is bent rod-shaped [6]. On the MS agar medium used at the time of isolation, R. slithyformis rarely formed long spirals. However, Chelius and Triplett [23] reported the formation of long spirals by the strain LSU 4T when cells were allowed to grow in R2A broth medium (see Figure 2). Larkin and Williams [1] reported a possible production of filaments up to 14 µm in length, which are not coiled. This contrasts the findings of Chelius et al. [7] who described the cells of the strain LSU 4T as circular with swollen ends that would not form filaments. Rings with an outer diameter of 2.0 to 3.0 µm may also occur [1]. Colonies produced a pale pink, nondiffusible, nonfluorescent pigment on MS agar [1]. The strain LSU 4T is a Gram-negative bacterium (Table 1). Strain LSU 4T is non-motile, aerobic and chemoorganotrophic [1]. It does not grow on media with NaCl concentrations of 1.5% or higher [23]. This feature was similar to that of another member of this genus, R. zeae [7]. The temperature range for growth is between 4°C-37°C, with an optimum between 20°C-30°C [6]; the strain being unable to grow at temperatures above 37°C [23]. The sole carbon sources used by the strain LSU 4T for growth on MS agar are glycogen, D-arabitol, dulcitol, inositol, mannitol, sorbitol and sorbose, but the growth was weak except in the presence of glycogen [23]. Some of these features are however contradictory to the findings of Chelius et al. [7] whose attempt to grow the strain LSU 4T in the presence of glycogen in R2A medium was unsuccessful. Further detailed physiological insight, e.g., carbon source utilization in R2A medium, MS agar medium, or by the API 50 CH test, have been reported previously [7,23]. Also, resistance to a variety of antibiotics has been reported [7,23].
Chemotaxonomy
The principal cellular fatty acids of strain LSU 4T are iso-C15:0 2-OH/C16:1ω7c (32.1%), iso-C15:0 (19.8%) and C16:1ω5c (16.5%) [23]. Minor fatty acids include C16:0 (7.1%), iso-C17:0 3-OH (7.0%), anteiso-C15:0 (4.3%), iso-C15:0 3-OH (4.1%), iso-C15:1 G (2.4%), C16:0 3-OH (2.0%), an unknown one (ECL 13.6) (1.83%) and C15:0 (1.5%) [23]. Major polar lipids were not reported for strain LSU 4T, but those of the genus Runella could be retrieved from R. defluvii strain EMB13T and R. limosa strain EMB111T [4,6].
Genome sequencing and annotation
Genome project history
This organism was selected for sequencing on the basis of its phylogenetic position [34], and is part of the Genomic Encyclopedia of Bacteria and Archaea project [35]. The genome project is deposited in the Genomes On Line Database [18] and the complete genome sequence is deposited in GenBank. Sequencing, finishing and annotation were performed by the DOE Joint Genome Institute (JGI). A summary of the project information is shown in Table 2.
Growth conditions and DNA isolation
R. slithyformis strain LSU 4T, DSM 19594, was grown in DSMZ medium 7 (Ancyclobacter-Spirosoma medium) [36] at 28°C. DNA was isolated from 0.5–1 g of cell paste using MasterPure Gram-positive DNA purification kit (Epicentre MGP04100) following the standard protocol as recommended by the manufacturer with modification st/DL for cell lysis as described in Wu et al. 2009 [35]. DNA is available through the DNA Bank Network [31].
Genome sequencing and assembly
The genome was sequenced using a combination of Illumina and 454 sequencing platforms. All general aspects of library construction and sequencing can be found at the JGI website [37]. Pyrosequencing reads were assembled using the Newbler assembler (Roche). The initial Newbler assembly consisting of 121 contigs in two scaffolds was converted into a phrap [38] assembly by making fake reads from the consensus, to collect the read pairs in the 454 paired end library. Illumina GAii sequencing data (638.9 Mb) was assembled with Velvet [39] and the consensus sequences were shredded into 2.0 kb overlapped fake reads and assembled together with the 454 data. The 454 draft assembly was based on 206.2 Mb 454 draft data and all of the 454 paired end data. Newbler parameters are -consed -a 50 -l 350 -g -m -ml 20. The Phred/Phrap/Consed software package [38] was used for sequence assembly and quality assessment in the subsequent finishing process. After the shotgun stage, reads were assembled with parallel phrap (High Performance Software, LLC). Possible mis-assemblies were corrected with gapResolution [37], Dupfinisher [40], or sequencing cloned bridging PCR fragments with subcloning. Gaps between contigs were closed by editing in Consed, by PCR and by Bubble PCR primer walks (J.-F. Chang, unpublished). A total of 289 additional reactions and 3 shatter libraries were necessary to close gaps and to raise the quality of the finished sequence. Illumina reads were also used to correct potential base errors and increase consensus quality using a software Polisher developed at JGI [41]. The error rate of the completed genome sequence is less than 1 in 100,000. Together, the combination of the Illumina and 454 sequencing platforms provided 128.6 × coverage of the genome. The final assembly contained 540,807 pyrosequence and 19,068,176 Illumina reads.
Genome annotation
Genes were identified using Prodigal [42] as part of the Oak Ridge National Laboratory genome annotation pipeline, followed by a round of manual curation using the JGI GenePRIMP pipeline [43]. The predicted CDSs were translated and used to search the National Center for Biotechnology Information (NCBI) nonredundant database, UniProt, TIGR-Fam, Pfam, PRIAM, KEGG, COG, and InterPro databases. Additional gene prediction analysis and functional annotation was performed within the Integrated Microbial Genomes - Expert Review (IMG-ER) platform [44].
Genome properties
The genome consists of one circular chromosome with a length of 6,568,739 bp and a G+C content of 47%, and five circular plasmids with 38,784 bp, 44,754 bp, 66,926 bp, 93,527 bp and 106,999 bp length, respectively (Table 3 and Figure 3). Of the 6,025 genes predicted, 5,974 were protein-coding genes, and 51 RNAs; 182 pseudogenes were also identified. The majority of the protein-coding genes (59.7%) were assigned a putative function while the remaining ones were annotated as hypothetical proteins. The distribution of genes into COGs functional categories is presented in Table 4.
Graphical map of the circular chromosome (plasmids not shown, but accessible through the img/er pages on the JGI web pages [37]). From outside to center: Genes on forward strand (color by COG categories), Genes on reverse strand (color by COG categories), RNA genes (tRNAs green, rRNAs red, other RNAs black), GC content, GC skew.
References
Larkin JM, Williams PM. Runella slithyformis gen. nov., sp. nov., a curved, nonflexible, pink bacterium. Int J Syst Bacteriol 1978; 28:32–36. http://dx.doi.org/10.1099/00207713-28-1-32
Skerman VBD, McGowan V, Sneath PHA. Approved lists of bacterial names. Int J Syst Bacteriol 1980; 30:225–420. http://dx.doi.org/10.1099/00207713-30-1-225
Euzéby JP. List of bacterial names with standing in nomenclature: a folder available on the Internet. Int J Syst Bacteriol 1997; 47:590–592. PubMed http://dx.doi.org/10.1099/00207713-47-2-590
Lu S, Lee JR, Ryu SH, Chung BS, Choe WS, Jeon CO. Runella defluvii sp. nov., isolated from a domestic wastewater treatment plant. Int J Syst Evol Microbiol 2007; 57:2600–2603. PubMed http://dx.doi.org/10.1099/ijs.0.65252-0
Furuhata K, Kato Y, Goto K, Saitou K, Sugiyama J, Hara M, Fukuyama M. Identification of pink-pigmented bacteria isolated from environmental water samples and their biofilm fromation abilities. Biocontrol Sci 2008; 13:33–39. PubMed http://dx.doi.org/10.4265/bio.13.33
Ryu SH, Nguyen TTH, Park W, Kim CJ, Jeon CO. Runella limosa sp. nov., isolated from activated sludge. Int J Syst Evol Microbiol 2006; 56:2757–2760. PubMed http://dx.doi.org/10.1099/ijs.0.64460-0
Chelius MK, Henn JA, Triplett EW. Runella zeae sp. nov., a novel Gram-negative bacterium from the stems of surface-sterilized Zea mays. Int J Syst Evol Microbiol 2002; 52:2061–2063. PubMed http://dx.doi.org/10.1099/ijs.0.02203-0
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ. Basic local alignment search tool. J Mol Biol 1990; 215:403–410. PubMed
Korf I, Yandell M, Bedell J. BLAST, O’Reilly, Sebastopol, 2003.
DeSantis TZ, Hugenholtz P, Larsen N, Rojas M, Brodie EL, Keller K, Huber T, Dalevi D, Hu P, Andersen GL. Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl Environ Microbiol 2006; 72:5069–5072. PubMed http://dx.doi.org/10.1128/AEM.03006-05
Porter MF. An algorithm for suffix stripping. Program: electronic library and information systems. 1980; 14:130–137.
Castresana J. Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol Biol Evol 2000; 17:540–552. PubMed http://dx.doi.org/10.1093/oxfordjournals.molbev.a 026334
Lee C, Grasso C, Sharlow MF. Multiple sequence alignment using partial order graphs. Bioinformatics 2002; 18:452–464. PubMed http://dx.doi.org/10.1093/bioinformatics/18.3.452
Stamatakis A, Hoover P, Rougemont J. A rapid bootstrap algorithm for the RAxML web-servers. Syst Biol 2008; 57:758–771. PubMed http://dx.doi.org/10.1080/10635150802429642
Hess PN, De Moraes Russo CA. An empirical test of the midpoint rooting method. Biol J Linn Soc Lond 2007; 92:669–674. http://dx.doi.org/10.1111/j.10958312.2007.00864.x
Pattengale ND, Alipour M, Bininda-Emonds ORP, Moret BME, Stamatakis A. How many bootstrap replicates are necessary? Lect Notes Comput Sci 2009; 5541:184–200. http://dx.doi.org/10.1007/978-3-642-02008-7_13
Swofford DL. PAUP*: Phylogenetic Analysis Using Parsimony (*and Other Methods), Version 4.0 b10. Sinauer Associates, Sunderland, 2002.
Liolios K, Chen IM, Mavromatis K, Tavernarakis N, Hugenholtz P, Markowitz VM, Kyrpides NC. The Genomes On Line Database (GOLD) in 2009: status of genomic and metagenomic projects and their associated metadata. Nucleic Acids Res 2010; 38:D346–D354. PubMed http://dx.doi.org/10.1093/nar/gkp848
Abt B, Teshima H, Lucas S, Lapidus A, Del Rio TG, Nolan M, Tice H, Cheng JF, Pitluck S, Liolios K, et al. Complete genome sequence of Leadbetterella byssophila type strain (4M15T). Stand Genomic Sci 2011; 4:2–12. PubMed http://dx.doi.org/10.4056/sigs.1413518
Lail K, Sikorski J, Saunders E, Lapidus A, Del Rio TG, Copeland A, Tice H, Cheng JF, Lucas S, Nolan M, et al. Complete genome sequence of Spirosoma linguale type strain (1T). Stand Genomic Sci 2010; 2:176–185. PubMed http://dx.doi.org/10.4056/sigs.741334
Lang E, Lapidus A, Chertkov O, Brettin T, Detter JC, Han C, Copeland A, Del Rio TG, Nolan M, Chen F, et al. Complete genome sequence of Dyadobacter fermentans type strain (NS114T). Stand Genomic Sci 2009; 1:133–140. PubMed http://dx.doi.org/10.4056/sigs.19262
Xie G, Bruce DC, Challacombe JF, Chertkov O, Detter JC, Gilna P, Han CS, Lucas S, Misra M, Myers GL, et al. Genome sequence of the cellulolytic gliding bacterium Cytophaga hutchinsonii. Appl Environ Microbiol 2007; 73:3536–3546. PubMed http://dx.doi.org/10.1128/AEM.00225-07
Chelius MK, Triplett EW. gen. nov., sp. nov., a novel Gram-negative bacterium isolated from surface-sterilized Zea mays stems. Int J Syst Evol Microbiol 2000; 50:751–758. PubMed http://dx.doi.org/10.1099/00207713-50-2-751
Field D, Garrity G, Gray T, Morrison N, Selengut J, Sterk P, Tatusova T, Thomson N, Allen MJ, Angiuoli SV, et al. The minimum information about a genome sequence (MIGS) specification. Nat Biotechnol 2008; 26:541–547. PubMed http://dx.doi.org/10.1038/nbt1360
Woese CR, Kandler O, Wheelis ML. Towards a natural system of organisms: proposal for the domains, and Eucarya. Proc Natl Acad Sci USA 1990; 87:4576–4579. PubMed http://dx.doi.org/10.1073/pnas.87.12.4576
Krieg NR, Ludwig W, Euzéby J, Whitman WB. Phylum XIV. Bacteroidetes phyl. nov. In: Krieg NR, Staley JT, Brown DR, Hedlund BP, Paster BJ, Ward NL, Ludwig W, Whitman WB (eds), Bergey’s Manual of Systematic Bacteriology, second edition, vol. 4 (The Bacteroidetes, Spirochaetes, Tenericutes (Mollicutes), Acidobacteria, Fibrobacteres, Fusobacteria, Dictyoglomi, Gemmatimonadetes, Lentisphaerae, Verrucomicrobia, Chlamydiae, and Planctomycetes), Springer, New York, 2010, p. 25.
List Editor. Validation List N∘ 143. Int J Syst Evol Microbiol 2012; 62:1–4. http://dx.doi.org/10.1099/ijs.0.039487-0
Nakagawa Y. Class IV. Cytophagia class. nov. In: Krieg NR, Staley JT, Brown DR, Hedlund BP, Paster BJ, Ward NL, Ludwig W, Whitman WB (eds), Bergey’s Manual of Systematic Bacteriology, second edition, vol. 4 (The Bacteroidetes, Spirochaetes, Tenericutes (Mollicutes), Acidobacteria, Fibrobacteres, Fusobacteria, Dictyoglomi, Gemmatimonadetes, Lentisphaerae, Verrucomicrobia, Chlamydiae, and Planctomycetes), Springer, New York, 2010, p. 370.
Leadbetter ER. Order II. Cytophagales Nomen novum. In: Buchanan RE, Gibbons NE (eds), Bergey’s Manual of Determinative Bacteriology, Eighth Edition, The Williams and Wilkins Co., Baltimore, 1974, p. 99.
Stanier RY. Studies on the Cytophagas. J Bacteriol 1940; 40:619–635. PubMed
Gemeinholzer B, Dröge G, Zetzsche H, Haszprunar G, Klenk HP, Güntsch A, Berendsohn WG, Wägele JW. The DNA Bank Network: the start from a German initiative. Biopreserv Biobank 2011; 9:51–55. http://dx.doi.org/10.1089/bio.2010.0029
BAuA. 2010. Classification of bacteria and archaea in risk groups. TRBA 466, p. 194. www.baua.de Bundesanstalt für Arbeitsschutz und Arbeitsmedizin, Germany.
Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT, et al. Gene Ontology: tool for the unification of biology. Nat Genet 2000; 25:25–29. PubMed http://dx.doi.org/10.1038/75556
Klenk HP, Göker M. En route to a genome-based classification of and? Syst Appl Microbiol 2010; 33:175–182. PubMed http://dx.doi.org/10.1016/j.syapm.2010.03.003
Wu D, Hugenholtz P, Mavromatis K, Pukall R, Dalin E, Ivanova NN, Kunin V, Goodwin L, Wu M, Tindall BJ, et al. A phylogeny-driven genomic encyclopaedia of and. Nature 2009; 462:1056–1060. PubMed http://dx.doi.org/10.1038/nature08656
List of growth media used at DSMZ: http://www.dsmz.de/catalogues/catalogue-microorganisms/culture-technology/list-of-media-for-microorganisms.html.
JGI website. http://www.jgi.doe.gov
The Phred/Phrap/Consed software package. http://www.phrap.com
Zerbino DR, Birney E. Velvet: algorithms for de novo short read assembly using de Bruijn graphs. Genome Res 2008; 18:821–829. PubMed http://dx.doi.org/10.1101/gr.074492.107
Han C, Chain P. Finishing repeat regions automatically with Dupfinisher. In: Proceeding of the 2006 international conference on bioinformatics & computational biology. Arabnia HR, Valafar H (eds), CSREA Press. June 26–29, 2006: 141–146.
Lapidus A, LaButti K, Foster B, Lowry S, Trong S, Goltsman E. POLISHER: An effective tool for using ultra short reads in microbial genome assembly and finishing. AGBT, Marco Island, FL, 200845. J Bacteriol 1940; 40:619–635.
Hyatt D, Chen GL, LoCascio PF, Land ML, Larimer FW, Hauser LJ. Prodigal: prokaryotic gene recognition and translation initiation site identification. BMC Bioinformatics 2010; 11:119. PubMed http://dx.doi.org/10.1186/1471-2105-11-119
Pati A, Ivanova NN, Mikhailova N, Ovchinnikova G, Hooper SD, Lykidis A, Kyrpides NC. GenePRIMP: a gene prediction improvement pipeline for prokaryotic genomes. Nat Methods 2010; 7:455–457. PubMed http://dx.doi.org/10.1038/nmeth.1457
Markowitz VM, Ivanova NN, Chen IMA, Chu K, Kyrpides NC. IMG ER: a system for microbial genome annotation expert review and curation. Bioinformatics 2009; 25:2271–2278. PubMed http://dx.doi.org/10.1093/bioinformatics/btp393
Acknowledgements
We would like to gratefully acknowledge the help of Regine Fähnrich (DSMZ) for growing R. slithyformis cultures. This work was performed under the auspices of the US Department of Energy Office of Science, Biological and Environmental Research Program, and by the University of California, Lawrence Berkeley National Laboratory under contract No. DE-AC02-05CH11231, Lawrence Livermore National Laboratory under Contract No. DE-AC52-07NA27344, and Los Alamos National Laboratory under contract No. DE-AC02-06NA25396, UT-Battelle and Oak Ridge National Laboratory under contract DE-AC05-00OR22725, as well as German Research Foundation (DFG) INST 599/1-2.
Author information
Authors and Affiliations
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Copeland, A., Zhang, X., Misra, M. et al. Complete genome sequence of the aquatic bacterium Runella slithyformis type strain (LSU 4T). Stand in Genomic Sci 6, 145–154 (2012). https://doi.org/10.4056/sigs.2475579
Published:
Issue Date:
DOI: https://doi.org/10.4056/sigs.2475579