Abstract
The CHRNA4 gene is known to be associated with individual differences in attention. However, its associations with other cognitive functions remain to be elucidated. In the present study, we investigated the effects of genetic variations in CHRNA4 on rapid scene categorization by 100 healthy human participants. In Experiment 1, we also conducted the Attention Network Test (ANT) in order to examine whether the genetic effects could be accounted for by attention. CHRNA4 was genotyped as carrying the TT, CT, or CC allele. The scene categorization task required participants to judge whether the category of a scene image (natural or man-made) was consistent with a cue word displayed at the response phase. The target–mask stimulus onset asynchrony (SOA) ranged from 13 to 93 ms. In comparison with CC-allele carriers, CT- and TT-allele carriers responded more accurately at the long SOA (93 ms) only during natural-scene categorization. In contrast, we observed no consistent association between CHRNA4 and the ANT, and no intertask correlation between scene categorization and the ANT. To validate our natural-scene categorization results, Experiment 2, carried out with an independent sample of 100 participants and a different stimulus set, successfully replicated the association between CHRNA4 genotypes and natural-scene categorization accuracy at long SOAs (67 and 93 ms). Our findings demonstrate, for the first time, that genetic variations in CHRNA4 can moderately contribute to individual differences in natural-scene categorization performance.
Similar content being viewed by others
Human cortical neurotransmitters (e.g., acetylcholine, dopamine, and serotonin) have been linked to many cognitive brain functions (Everitt & Robbins, 1997; Voytko, 1996). Thus, variation in the genes that encode neurotransmitters and their receptors may lead to individual differences in cognitive functions. Studies using the gene–cognition approach have usually explored a link between cognitive performance and genetic variation arising from single-nucleotide polymorphisms (SNPs; Fossella et al., 2002; Greenwood, Fossella, & Parasuraman, 2005). SNPs are defined as DNA sequence variations that occur when a single nucleotide (A, T, C, or G) differs among individuals. These SNPs, also known as point mutations, can lead to differences in protein coding. If the protein-coding variation influences receptor sensitivity or enzyme activity, the efficacy of cortical neurotransmission may be affected. Recently, SNPs in healthy individuals have been shown to strongly affect cognitive performance (Fossella et al., 2002; Greenwood, Sunderland, Friz, & Parasuraman, 2000). The genetic effects of polymorphisms can be more reliably detected among healthy individuals than within a patient population because a larger sample size can reduce Type I errors (Greenwood & Parasuraman, 2003; Ioannidis, Ntzani, Trikalinos, & Contopoulos-Ioannidis, 2001).
Previous studies have shown that the C1545T polymorphism (rs#1044396) in Exon 5 of the cholinergic receptor, nicotinic, alpha-4 (CHRNA4) gene, which encodes the alpha-4 subunit in nicotinic acetylcholine receptors (nAChRs), is associated with attentional orienting performance in healthy human individuals (Parasuraman, Greenwood, Kumar, & Fossella, 2005). CHRNA4 was genotyped as bearing the TT, CT, or CC allele. An increased C-allele dose (TT < CT < CC) was found to result in greater costs in attentional reorienting with invalid endogenous cues and decreased benefit in orienting with valid endogenous cues. This association was successfully replicated (Espeseth et al., 2006). A multiple-object tracking (MOT) task study revealed an association between CHRNA4 and sustained attention (Espeseth, Sneve, Rootwelt, & Laeng, 2010). Besides single-gene effects, synergistic effects of nicotinic and muscarinic cholinergic receptor genes (CHRNA4 and CHRM2) on attentional scaling have been reported (Greenwood, Lin, Sundararajan, Fryxell, & Parasuraman, 2009). Cognitive genetic studies using neuroimaging techniques have further confirmed the relationship between CHRNA4 and attention. An increased C-allele dose was shown to reduce the blood-oxygen-level-dependent signal in the anterior cingulate and parietal cortices, indicating increased neural efficiency with valid cues (Winterer et al., 2007). Similarly, an event-related potential (ERP) study showed that SNPs of the CHRNA4 gene affected the latency or amplitude of the ERP components for visual attention, N1 and P1 (Espeseth, Endestad, Rootwelt, & Reinvang, 2007). With regard to the physiological background, it is known that the nicotinic receptors are associated with a superfamily of ligand-gated ion channels that modulate fast signal transmission at synapses. The nicotinic receptor is thought to function as an excitatory element in the central nervous system, and mutations of CHRNA4 would affect neuronal excitation (Kuryatov, Gerzanich, Nelson, Olale, & Lindstrom 1997; Weiland, Witzemann, Villarroel, Propping, & Steinlein, 1996). All of these studies have focused on the same rs#1044396 mutation in Exon 5 of the CHRNA4 gene. Accordingly, a strong link between CHRNA4 and spatial attention has been proposed.
SNPs are frequently associated with multiple cognitive and physiological functions. Thus, elucidation of the complex relationships between SNPs and their phenotypic functions is a crucial step toward understanding the biophysical basis of cognitive processes (for a review, see Green et al., 2008). An association between CHRNA4 and working memory capacity has been demonstrated (Greenwood et al., 2008; Markett, Montag, & Reuter, 2009). This association was not attributed to attentional amplification in the working memory, because the task required no additional attentional requirements. Recently, CHRNA4 has also been reported to be related to negative emotions (Markett, Montag, & Reuter, 2011).
It is well known that CHRNA4 is a nicotine-receptor-related gene. A previous study in humans demonstrated that T-allele carriers of CHRNA4 showed a protective effect against nicotine dependence (Feng et al., 2004). In addition, it was proposed that nicotine-related cognitive functions such as attention are associated with CHRNA4 (e.g., Greenwood et al., 2005). Thus, CHRNA4 could influence nicotine signaling in humans. It has been suggested that the nicotine signal is also associated with enhanced detection of visual stimuli (Disney, Aoki, & Hawken, 2007) and with scene perception in rats (Goard & Dan, 2009). Taken together, these findings indicate that nicotine signaling is important in the visual processing of scene perception, and that nicotine signals of CHRNA4 may influence scene perception performance in humans. However, to our knowledge, the effect of nicotine on the processing of natural images in healthy humans has not previously been investigated, despite a long tradition of studying the visual processing of natural images using rapid scene categorization tasks (e.g., Loschky et al., 2007; Potter, 1975). Rapid scene categorization studies are focused on superordinate-level categorization. According to the spatial envelope model, the superordinate-level natural-versus-man-made distinction operates on the basis of the global statistical properties within a scene (Oliva & Torralba, 2001). For example, curvilinear orientations may indicate the natural category, whereas rectangular properties are indicative of the man-made category. Meanwhile, a longer reaction time (RT) is required for basic-level categorization (e.g., forest, mountain, or street) than for superordinate-level categorization (Greene & Oliva, 2009). One possible interpretation is the “coarse-to-fine” hypothesis (Hughes, Nozawa, & Kitterle, 1996; Macé, Joubert, Nespoulous, & Fabre-Thorpe, 2009; Schyns & Oliva, 1994), which states that a coarse view of a scene provides sufficient information for superordinate-level scene categorization, whereas basic-level scene categorization requires a finer view of the scene. Therefore, in comparison with superordinate-level categorization, basic-level scene categorization may require more detailed properties of local objects (Joubert, Rousselet, Fize, & Fabre-Thorpe, 2007). This indicates that basic-level categorization includes additional processing of scene information. Thus, superordinate-level and basic-level categorizations may be mediated by distinctive processes. Investigation of the effects of CHRNA4 on rapid superordinate-level scene categorization would provide useful information on associated brain networks, neurochemical innervation for scene perception, and later cognitive processes such as attention (Torralba, Oliva, Castelhano, & Henderson, 2006), object recognition (Davenport & Potter, 2004), or long-term memory within scenes (Brewer & Treyens, 1981). Therefore, in the present study, we examined the role of the nicotinic receptor in natural-image processing, by investigating the potential association between CHRNA4 gene polymorphisms and rapid scene categorization performance in healthy humans.
It is important to clarify whether an association exists between CHRNA4 and rapid scene categorization, and if so, whether it can be accounted for by the known associations of the receptor with other cognitive processes, such as spatial attention. The role of attention in rapid scene categorization is controversial. Humans can effortlessly categorize images with extremely short viewing times, such as 20 ms (Thorpe, Fize, & Marlot, 1996), even when the stimuli appear at unattended locations in peripheral vision (Fize, Fabre-Thorpe, Richard, Doyon, & Thorpe, 2005; Li, VanRullen, Koch, & Perona, 2002). This finding indicates that rapid scene categorization may be possible without the activation of attentional processes. Conversely, Codispoti, Ferrari, Junghöfer, and Schupp (2006) used ERPs to examine the effects of selective attention on rapid scene categorization. Early ERP activity (the P1 component) was observed in the posterior cortex while subjects were categorizing scene images, suggesting that the P1 component reflects the effects of attention in rapid scene categorization. Moreover, during the scene categorization task, the P1 component was also observed in the central–parietal region (Kincses, Chadaide, Varga, Antal, & Paulus, 2006). Thus, CHRNA4 may affect scene categorization in a manner that is either dependent or independent of its effects on selective attention.
To address this issue, we conducted the Attention Network Test (ANT; Fan et al., 2007) in conjunction with a rapid scene categorization task in Experiment 1. This task allowed us to measure three independent attentional components—alerting, orienting, and executive—with a single task. Given that attention can be broken into these three independent components (Posner & Petersen, 1990), we considered the ANT to be an appropriate task for productively and comprehensively investigating the association of attention with rapid scene categorization. We hypothesized that, if the effect of CHRNA4 on rapid scene categorization performance was a by-product of its association with attention, the CHRNA4 polymorphism would be associated with performance on both the ANT and the rapid scene categorization task, and, therefore, performance on the two behavioral tasks would be significantly correlated. By contrast, if the effects of CHRNA4 on rapid scene categorization were independent of attention, performance on the two behavioral tasks would not be correlated, and only a single task would be associated with the CHRNA4 polymorphism. In addition, in order to ensure that Experiment 1’s results were not obtained by chance, Experiment 2 was conducted as a replication.
Experiment 1
Method
Participants
A sample of 100 healthy participants (59 women, 41 men; mean age 20.9 years, SD = 1.92), was recruited from among undergraduate and graduate students at Kyoto University. All of the participants had normal vision and no neuropsychological deficits, and all provided written informed consent prior to the experiment. The experimental protocol was approved by the local Institutional Review Board.
Materials
Rapid scene categorization
A total of 432 gray scale photographs (1,280 pixels × 1,024 pixels) from the Oliva and Torralba (2001) database were drawn from four categories—two natural (beach and mountain) and two man-made (city center and highway)—with 108 images in each category. The images were presented on a 21-in. EIZO FlexScan F980 monitor (75-Hz refresh rate), with the viewing distance set at 68 cm by a chinrest. The images were manipulated to subtend a visual angle of 12.7º × 12.7º. Participants’ responses were collected using a response box (Cedrus RB-520), and the entire experiment was conducted in a dark room.
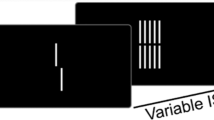
Mask images were created using full-phase randomization, while keeping the spatial frequency amplitude spectra and mean luminance identical to those of the original images (Fig. 1). The modifications were based on a fast Fourier transformation and the random image structure evolution (RISE) algorithm (Sadr & Sinha, 2004).
Schematic illustration of a trial in the rapid scene categorization task. The trial included a full-phase randomization image as a mask stimulus. The participant’s task was to judge whether or not the cue word category was consistent with the target scene category. ISI, interstimulus interval; SOA, stimulus onset asynchrony
Attention Network Test
The stimuli consisted of a row of five horizontal black lines, with arrowheads pointing either left or right, and a gray background. The individual arrow size was 0.58º of visual angle, and each arrow was separated from adjacent arrows by 0.06º of visual angle. The target appeared as a leftward or rightward arrow at the center, flanked on each side by two arrows pointing in either the same direction (congruent condition) or the opposite direction (incongruent condition). The stimuli (all five arrows) subtended 3.27º of visual angle. The row of five arrows was presented horizontally and displayed 1.06º above or below the fixation cross. Figure 2 illustrates the stimulus configuration of the ANT. We created three cue conditions: no cue, center cue (indicated by an asterisk that appeared on the fixation point), and spatial cue (indicated by an asterisk that appeared either above or below the fixation point). The attentional effects (alerting, orienting, and executive) were computed on the basis of the RT differences between experimental conditions, as follows: alerting effect = RT(no cue) – RT(center cue); orienting effect = RT(center cue) – RT(spatial cue); and executive effect = RT(incongruent) – RT(congruent).
Schematic illustration of a trial in the Attention Network Test (ANT). A cue condition (no cue, center cue, or spatial cue) appeared for 200 ms. A variable time later (300–1,450 ms, average 550 ms), the target was displayed until participants responded. Participants had to judge within 2,000 ms whether the target (the center arrow) pointed to the left or right, either with or against the flanker arrows (congruent or incongruent). The duration from the beginning of the target display to the next trial was varied from 3,000 to 4,200 ms (average 3,300 ms)
The stimuli were presented on a 21-in. EIZO FlexScan F980 monitor (75-Hz refresh rate), with the viewing distance set at 68 cm by a chinrest. Participants’ responses were collected using a response box (Cedrus RB-520). The entire experiment was conducted in a dark room.
Procedure
Rapid scene categorization
An experimental trial is schematically represented in Fig. 1; this design was chosen to replicate Experiment 3 from Loschky et al. (2007). Each trial began with a fixation cross displayed for 1,000 ms. Either the natural or man-made stimulus (i.e., the target) was displayed for 13 ms—approximately the minimum duration required for above-chance gist recognition performance, as had been determined by previous studies (Bacon-Macé, Macé, Fabre-Thorpe, & Thorpe, 2005). This was followed by an interstimulus interval (ISI)—a blank screen display—of variable duration (0, 27, 54, or 80 ms), after which a mask stimulus was shown for 53 ms, making a strong (4:1) mask:target duration to show any spatial masking effects. The stimulus onset asynchrony (SOA) from a scene picture to mask stimulus was 13, 40, 67, or 93 ms. The short SOAs (13, 40, or 67 ms) were chosen in order to focus on the early processing stage of scene recognition. The long SOA (93 ms) was selected because an SOA close to 100 ms generally reaches asymptote (Potter, 1976). Following the mask image, a blank screen was displayed for 750 ms, after which the response screen was presented. During the response phase, either “Natural” or “Man-Made” was displayed at the center of the screen. Participants were asked to judge whether or not the displayed category was consistent with the target category, and accuracy and RTs were recorded. During practice trials, either CORRECT or INCORRECT was displayed for 1,000 ms as feedback after the response. In the experimental block, this feedback was not provided. The number of practice trials was 36, and the number of experimental trials was 432 (12 blocks of 36 trials per block).
Attention Network Test
Figure 2 illustrates the timeline of the ANT, which is a replication of the experiment conducted by Fan et al. (2007). Each trial began with a cue display (no cue, center cue, or spatial cue) presented for 200 ms. A variable time later (300–1,450 ms, average 550 ms), the target was displayed until participants responded. The maximum time allowed for the response was 2,000 ms. The target validity was 100 % (i.e., the target always appeared at the cued location). The duration from the beginning of the target display to the next trial varied from 3,000 to 4,200 ms (average 3,300 ms). A fixation cross was displayed in the center of the screen throughout each trial. Participants were instructed to respond as quickly and accurately as possible, with the left button of the Cedrus response box if the central arrow of the target was directed to the left, and with the right button of the response box if the central arrow was directed right. Prior to the experimental block, a practice block was conducted. In practice trials, CORRECT or INCORRECT was displayed for 1,000 ms as feedback after the response. In the experimental block, this feedback was not provided. The number of practice trials was 30, and the number of experimental trials was 648 (six blocks of 108 trials per block).
Genotyping
All participants were genotyped for the C1545T polymorphism (rs#1044396) in Exon 5 of the target gene, CHRNA4. Genomic DNA was extracted from buccal cells (Buccal Amp DNA Extraction Kit, Epicentre Biotechnologies, Madison, WI). Each buccal sample yielded 2 μl of DNA. Taq polymerase, the polymerase chain reaction–restriction fragment length polymorphism (PCR-RFLP) buffer, and deoxyribonucleotide triphosphates were acquired from Nacalai Tesque (Kyoto, Japan) and used at the manufacturer’s recommended concentrations for a 20 μl PCR reaction. PCR reactions and RFLP digits were carried out in a programmed thermal cycler (Nacalai Tesque, Kyoto, Japan). PCR products were separated by gel electrophoresis (3 % agarose), followed by staining with SYBR Green (Nacalai Tesque, Kyoto, Japan) to resolve and visualize the DNA fragments. Two primers were employed to identify T-to-C polymorphisms in the alpha 4 nicotinic receptor subunit (i.e., no C alleles = TT, one C allele = CT, two C alleles = CC). Forward (5′-ACCAGGGCTGGCCAAAGCCAGG-3′) and reverse (5′-GTGCTTTGGTGCTGCGGGTC-3′) primers were utilized and digested with the HhaI enzyme (Steinlein et al., 1997). The T allele yielded 152-base-pair (bp) and 138-bp bands, whereas the C allele resulted in 152-bp, 105-bp, and 33-bp bands. Using these methods, the sample of 100 participants was subdivided into the TT genotype (n = 5), CT genotype (n = 47), and CC genotype (n = 48). Thus, the T-allele frequency was 28.5 %. These groups resulted in the Hardy–Weinberg equilibrium (p > .05 in χ 2 tests). The three SNPs from Exon 5 of CHRNA4 were selected from rs#1044396. The cohort was grouped into CHRNA4 T-allele carriers (CT and TT genotypes) and CC genotypes, because of the low number of TT carriers.
Results and discussion
Rapid scene categorization
The mean proportions of correct responses were analyzed for each experimental condition and for each CHRNA4 genotype. A 2 (genotype) × 2 (image category) × 4 (SOA) mixed-design analysis of variance (ANOVA) revealed significant effects of image category [F(1, 98) = 114.72, p < .001, η 2 = .1430] and SOA [F(3, 98) = 156.56, p < .001, η 2 = .1522]. No significant effect of CHRNA4 genotype on rapid scene categorization emerged (F < 1, n.s., η 2 = 0.0010), but a three-way interaction of this factor with image category and SOA was shown [F(3, 294) = 3.02, p = .04, η 2 = .0400]. To clarify this interaction, a 2 (genotype) × 2 (image category) ANOVA was carried out for each SOA condition. Only the 93-ms SOA condition showed a significant interaction [F(1, 294) = 5.59, p = .04, η 2 = .0551]. Planned comparisons showed that T carriers (CT/TT) performed the natural-scene categorization task with greater accuracy (.83, SE = .01) than did the CC homozygotes (.78, SE = .02) (see Fig. 3). By contrast, with man-made images, we observed no significant difference in performance between T carriers and CC homozygotes. To our knowledge, this is the first report demonstrating that CHRNA4 is a gene that modestly modulates the behavioral variation of rapid scene categorization.
Proportions of correct responses given by T carriers and CC homozygotes, as a function of stimulus onset asynchrony (SOA). a Natural-scene categorization conditions. b Man-made-scene categorization conditions. A significant difference was observed between the CHRNA4 genotypes at the 93-ms SOA in natural-scene categorization. Standard error bars are shown
To confirm that our results for the proportions of correct responses were not affected by speed–accuracy trade-offs, we evaluated the RT data. We observed a main effect of SOA [F(3, 294) = 6.19, p < .001, η 2 = .0530], with shorter mean RTs under longer SOA conditions, but no main effect of genotype and category, and no interactions (F < 1, η 2 < .001). For example, for natural-scene categorization at the 93-ms SOA, the RT of the CC genotype (580 ms) was significantly longer than that of the CT/TT genotype (513 ms). Greater accuracy was associated with faster RTs, and therefore no speed–accuracy trade-off was evident.
Attention Network Test
The mean RTs were computed for each condition. A 2 (genotype) × 3 (cue) × 2 (target condition) mixed-design ANOVA revealed no significant effect of genotype [F(1, 98) = 0.03, n.s., η 2 = .0003] A two-way interaction was observed between cue and target condition [F(2, 196) = 1500.00, p < .001, η 2 = .2957], but not between genotype and cue [F(2, 196) = 0.55, n.s., η 2 = 0.0083] or between genotype and target condition [F(1, 98) = 0.14, n.s., η 2 = .0001] No three-way interaction was observed [F(2, 196) = 0.93, n.s., η 2 = .0002]
In order to confirm the lack of a significant difference in attention between CHRNA4 genotypes, a planned comparison was conducted for each attentional effect. As is shown in Fig. 4, none of the attentional effects differed significantly between the CHRNA4 genotypes [alerting, t(98) = 1.02; orienting, t(98) = 1.87; and executive, t(98) = 0.25]. The effect-size (d) values for the unequal sample sizes of the genotype subgroups for the alerting, orienting, and executive components were 0.24, 0.26, and 0.10, respectively. Accuracy in the ANT exceeded 94 % across all experimental conditions, and therefore, no speed–accuracy trade-off was evident.
Our findings are inconsistent with those of Parasuraman et al. (2005), who demonstrated that orienting showed CHRNA4 genotype effects. This discrepancy may have arisen because of the use of invalid trials by Parasuraman et al.; the ANT did not include invalid trials. Invalid trials in spatial cueing tasks may require more distributed attention, because participants need to distribute attention to two possible locations (left and right). This proposal is consistent with the previously reported distributed attention and CHRNA4 association for the MOT task (Espeseth et al., 2010). Therefore, although the spatial cueing task of Parasuraman et al. was originally assumed to be a selective attention task, it is possible that distributed attention was included, and this may explain the absence of CHRNA4 genotype effects in the ANT in this study.
Intertask correlations
Intertask correlations were analyzed for each condition of the rapid scene categorization task and ANT. The accuracy of natural-scene categorization at the longest SOA (93 ms) correlated with the alerting, orienting, and executive effects (−0.01, 0.04, and 0.14, respectively) of the ANT. In this case, the effect-size index reflects the population product moment correlation (Cohen, 1988). Accordingly, the effect-size indices are small. No correlation was observed with any other condition.
Taken together, the results of our ANT and intertask correlations indicate that the association of rapid scene categorization with CHRNA4 may not be a by-product of any attentional components in the ANT (alerting, orienting, or executive). To our knowledge, this is the first report demonstrating that CHRNA4 has moderate genetic effects contributing to the behavioral variation in rapid scene categorization. However, the observed effect might reflect a single anomalous data point (an error rate increase from 17 % to 22 % by CC genotype), which implies that the results from Experiment 1 could have been obtained by chance. To validate our results, we replicated Experiment 1 with an independent sample of 100 participants (Exp. 2). Moreover, in order to confirm that our results were not stimulus-specific, we included an independent stimulus set with additional basic-level scene categories (forest and street) in Experiment 2.
Experiment 2
Method
Participants
A sample of 100 healthy participants (37 women, 63 men; mean age 21.0 years, SD = 2.47) was recruited from undergraduate and graduate students at Kyoto University. All of the participants had normal vision and no neuropsychological deficits, and all provided written informed consent prior to the experiment. The experimental protocol was approved by the local Institutional Review Board.
Materials
A total of 648 gray scale photographs (1,280 pixels × 1,024 pixels) from the Oliva and Torralba (2001) database were drawn from six basic-level categories (forest and street were added)—three natural (beach, mountain, and forest) and three man-made (city center, highway, and street)—with 324 images in each superordinate-level category. Mask images were created with the same algorithm (RISE) as in Experiment 1.
The images were presented on a 17-in. EIZO FlexScan T561 monitor (75-Hz refresh rate), with the viewing distance set at 56 cm by a chinrest. Images were manipulated to subtend a visual angle of 12.7º × 12.7º. Participants’ responses were recorded using the Cedrus RB-520 response box. The entire experiment was carried out in a dark room.
Procedure
The basic experimental sequence was the same as in Experiment 1 (see Fig. 1), but images of two new basic-level categories (forest and street) were added to previous data set. The number of practice trials was 36, and the number of experimental trials was 648 (12 blocks of 54 trials per block).
Genotyping
All participants were genotyped using the same procedure (PCR) as in Experiment 1. The sample of 100 participants was subdivided among the TT (n = 6), CT (n = 43), and the CC (n = 51) genotypes. Thus, the T-allele frequency was 27.5 %. These groups resulted in the Hardy–Weinberg equilibrium (p > .05 in χ 2 tests). As in Experiment 1, the cohort was grouped into CHRNA4 T-allele carriers (CT and TT genotypes) and CC genotypes, because of the low number of TT carriers.
Results and discussion
The mean proportions of correct responses were analyzed for each experimental condition. A 2 (genotype) × 2 (image category) × 4 (SOA) mixed-design ANOVA revealed a significant interaction between image category and SOA [F(3, 294) = 42.91, p < .001, η 2 = .03045]. We observed no significant effect of CHRNA4 genotype (F < 1, η 2 = .0176), but a three-way interaction of this factor with image category and SOA did emerge [F(3, 294) = 2.75, p = .04, η 2 = 0.0273]. To clarify this interaction, a 2 (genotype) × 4 (SOA) ANOVA was carried out for each image category. Only the natural category showed a significant interaction between CHRNA4 genotype and SOA [F(3, 588) = 2.86, p = .04, η 2 = .0332]. To interpret this interaction, we analyzed the genotype effect for the natural category. We revealed a significant main effect of genotype for the 67 ms [F(1, 784) = 4.77, p = .03, η 2 = .0568] and 93 ms [F(1, 784) = 3.86, p = .04, η 2 = .0442] SOAs. Planned comparisons showed that T carriers (CT/TT) performed the natural-scene categorization with greater accuracy than did the CC homozygotes for the 67-ms SOA (.84, SE = .02 vs. .79, SE = .01; Fig. 5) and the 93-ms SOA (.86, SE = .02 vs. .81, SE = .01; Fig. 5).
Proportions of correct responses given by T carriers and CC homozygotes, as a function of stimulus onset asynchrony (SOA; Exp. 2). a Natural-scene categorization conditions. b Man-made-scene categorization conditions. A significant difference was observed between the CHRNA4 genotypes at the 67-ms and 93-ms SOAs in the natural-scene categorization. Standard error bars are shown
To confirm that our results for the proportions of correct responses were not affected by speed–accuracy trade-offs, we evaluated the RT data. We observed a main effect of SOA [F(3, 294) = 22.03, p < .001, η 2 = .1835], with shorter mean RTs under the longer SOA conditions. We also showed a three-way interaction [F(3, 294) = 3.00, p = .03, η 2 = .0297]. To clarify this interaction, we carried out a 2 (genotype) × 4 (SOA) ANOVA. This revealed a significant interaction between genotype and SOA for man-made-scene categorization [F(3, 588) = 3.16, p = .02, η 2 = .0284]. By contrast, we found no significant genotype effects for man-made-scene categorization in any of the SOA conditions (F < 1, η 2 = .0024, for 93 ms). Greater accuracy was associated with faster RTs, and therefore no speed–accuracy trade-off was evident.
Our successful replication of Experiment 1 confirmed the validity of our results, which suggests that our findings are more likely to reflect an actual effect than an anomaly. The data from Experiment 2 also strongly suggest that CHRNA4 is a gene contributing to behavioral variation in rapid scene categorization. Moreover, because CHRNA4 is known to be a nicotinic acetylcholine-receptor-related gene (Pilz, Willer, Povey, & Abbott, 1992), it is possible that human acetylcholine function is associated with superordinate-level scene categorization process.
General discussion
In the present study, we investigated the effects of polymorphism in the CHRNA4 gene (which mediates binding of the nAChR alpha-4 subunit) on performance in rapid scene categorization task. We observed that participants with the CT/TT genotypes recognized natural-scene images with higher accuracy than did those with the CC genotype. To our knowledge, this is the first report demonstrating that a CHRNA4 polymorphism has a moderate effect on scene categorization performance. By contrast, we observed no significant effect of polymorphism on behavioral performance in the ANT, and no significant correlation between the performance scores in scene categorization and the ANT. Moreover, the effect of the polymorphism was observed only with natural-scene categories. Such content specificity is inconsistent with accounts based on general attentional orienting, because orienting is presumably independent of the content of the attended stimulus.
In Experiment 2, we validated the relationship between rapid scene categorization and CHRNA4 polymorphism. We successfully replicated the results of Experiment 1; that is, individuals with the CT/TT genotypes showed significantly greater accuracy in natural-scene categorization at the 67-ms and 93-ms SOAs than did those with the CC genotype. Thus, we confirmed the association between rapid scene categorization and CHRNA4 polymorphism.
Taken together, the present study offers evidence of a modest effect of CHRNA4 on scene categorization. Although our finding raises many questions, it does add to the body of knowledge that will eventually map very specific genetic contributions to cognitive functions.
Possible mechanisms underlying the effect of CHRNA4 on scene categorization
The functional properties of the rs#1044396 polymorphism in the CHRNA4 gene and its specific relationship with scene categorization performance remain to be elucidated. There is no known biochemical background to link this polymorphism to regulation of mRNA or protein structure. This lack of biochemical knowledge prevents us from providing a definitive explanation for our present findings. Nevertheless, we can propose some plausible theories on the basis of related studies of primates and humans.
Firstly, knowledge of functionality differences between CHRNA4 SNPs has been derived using mutational analysis of Xenopus oocytes models. Xenopus oocytes are frequently used for electrophysiological recording and for induction of distinct functional modifications of a receptor’s (ion channel) transmembrane ion channel by genetic alterations. Winterer et al. (2011) performed mutational analysis of Xenopus oocytes models and showed that Exon 5 CHRNA4 SNPs (i.e., TT/CT and CC) exert opposite functional effects on the sensitivity of the nicotinic alpha-4 beta-2 receptor to acetylcholine. In the high-affinity state, the TT/CT SNP resulted in higher receptor sensitivity than did the CC SNP. Generally, in the high-affinity state, a relatively low concentration of acetylcholine or nicotine is required in order to occupy its receptor and elicit a physiological response. If the nicotine receptor is sensitive to acetylcholine, the synapse channels open much more readily than if a receptor is less sensitive. This generally results in greater ion flow across the cell membrane, which alters the release of acetylcholine and changes signaling between neurons, thereby increasing the membrane potential concerning particular events. For instance, it is known that epilepsy is caused by excessive excitation of part or all of the gray matter around related brain regions (e.g., the temporal lobe). In particular, a study of autosomal dominant nocturnal frontal lobe epilepsy has demonstrated that CHRNA4 mutations increased the sensitivity of the receptor to acetylcholine, thereby enhancing neuronal excitability (Steinlein, 2004). Thus, because of higher nicotine receptor sensitivity, the membrane potential of specific neurons showed a greater rise with the TT/CT genotypes than with the CC genotype. Furthermore, using a neuroimaging technique (magnetic resonance imaging, or MRI), excessive gray matter excitation was identified in epilepsy-related brain regions (Feng & Walsh, 2001). Similarly, functional MRI (fMRI; Winterer et al., 2007) and ERP studies (Espeseth et al., 2007) have indicated that T-allele carriers of CHRNA4 exhibited enhanced regional brain activity to unexpected events in an oddball task. Furthermore, in behavioral experiments, T-allele carriers exhibited greater slowing of responses for invalid cues (Parasuraman et al., 2005). Greenwood et al. (2009) speculated that the higher nicotine receptor sensitivity of T-allele carriers might be a factor in the consistency between the brain and cognitive measurements. Thus, in the present study, varying nicotine receptor sensitivity by CHRNA4 SNPs may have contributed to differences in behavioral performance.
Secondly, a rodent study has yielded supporting evidence for nicotinic gene effects on scene perception. The visual receptive field response of rats to natural images was enhanced by nicotine injection into the nucleus basalis of the basal forebrain (Goard & Dan, 2009). Moreover, Disney et al. (2007) suggested that nicotinic input to area V1 in macaque monkeys mediated the detection of visual stimuli. Nicotine was thought to induce greater encoding and visual information processing of the natural scene. Similarly, other rodent studies have shown the effects of glutamatergic synaptic transmission enhancement by nicotinic receptor activation in the hippocampus (Giocomo & Hasselmo, 2005; Radcliffe & Dani, 1998). If we extrapolate the results of these rodent studies to humans, the nicotine receptor of CHRNA4 would be expected to play a critical role in encoding. Thus, CHRNA4 may modulate the encoding sensitivity for visual information processing in natural-scene categorization.
The content-specific effect of CHRNA4 on scene categorization may be accounted for by the differences in spatial components between natural and man-made images. Oliva and Torralba (2001) suggested that superordinate-level categorization includes a basis of global statistical properties. For example, man-made images are generally structured with straight horizontal and vertical lines, whereas natural images consist of textured zones and undulating contours. Thus, natural category images may be more similar to those used in previous animal studies (Disney et al., 2007; Goard & Dan, 2009) and may contain a broader range of orientation components. Because of this broader range of spatial components, natural category images may be more sensitive tools for detecting the effect of CHRNA4 on visual processing.
Superordinate-level and basic-level categorizations are assumed to involve distinct processes, and therefore, basic-level categorization is likely to provide us with aspects others than those presented with superordinate categorization. In comparison with superordinate scene categorization, basic-level scene categorization may require more detailed properties of local objects (Joubert et al., 2007). Therefore, the CHRNA4 contribution to local objects within a scene could be clarified using a basic-level scene categorization task. In order to address this issue, basic-level scene categorization (e.g., beach, forest, or mountain) performance between CHRNA4 genotypes should be compared in future research.
Role of attention in rapid scene categorization
The results of the present study favor the view that CHRNA4 affects rapid scene categorization in a manner that is independent of its effect on selective attention. Nevertheless, this hypothesis is based on null results (e.g., our behavioral correlation data), and therefore is inconclusive. It is also inconclusive because the genetic effect was found only for long SOAs (93 ms for Exp. 1, and 67 and 93 ms for Exp. 2), which is precisely the condition under which attentional influences are likely, as opposed to the automatic processes at short SOAs (Bachmann, Luiga, & Põder, 2005; Loftus & Ginn, 1984; Michaels & Turvey, 1979). Thus, attentional mediation could be the reason why the genetic association was found only at long and not at short SOAs. In addition, the ANT is just one type of test and may not cover all attentional aspects; therefore, further experiments using attentional manipulations will be required.
Distributed attention may be one possible factor that is not covered by the ANT. For instance, a previous study employing an MOT (multiple object tracking) task, which involved distributed attention (Alvarez & Scholl, 2005), revealed that CHRNA4 interacts with processing capacity (Espeseth et al., 2010). In the MOT task, participants monitored the random motion of multiple objects, for which superior distributed attention would appear to be beneficial. Espeseth et al. (2010) further suggested that T carriers of CHRNA4 could track objects more accurately than could C carriers. This trend is compatible with our present findings. Treisman (2006) stated that distributed attention is required for recognizing a scene as a whole; moreover, superordinate-level categorization can be processed using the scene as a whole (Greene & Oliva, 2009; Oliva & Torralba, 2001). Therefore, distributed attention may be essential for superordinate-level scene categorization, and superior distributed-attention performance by T carriers could explain why T carriers performed better with natural-scene categorization in our study. However, to clarify this attentional role issue, further studies should address the interaction between superordinate categorization and various attentional tasks (e.g., MOT or visual search). The notion of distributed attention may also explain the discrepancy observed between a previous spatial cueing task (Parasuraman et al., 2005) and the present ANT. A critical difference between the two studies was the use of invalid trials; our ANT did not include invalid trials. Without invalid trials, a spatial cue probably induces a highly focused attentional state. If the effect of CHRNA4 polymorphism is observed during distributed attention, it would be unlikely to be induced by the ANT used in the present study. By contrast, the validity manipulation in the spatial cueing task (Parasuraman et al., 2005) may have led participants to distribute attention to possible target locations, resulting in the polymorphism effect. In the spatial cueing task, participants need to distribute attention between two possible locations (left and right); in the ANT, they need to attend to a cued location only. This difference in attentional distribution may explain the faster overall RTs and smaller individual differences for the ANT than for the spatial cueing task. For instance, the mean RT for the spatial-cue condition in the ANT was much faster (437.8 ms) and showed less variance (SE = 6.1 ms) than did that for the valid condition in Parasuraman et al.’s spatial cueing task (592.3 ms). Thus, attention distribution may be critical for the lack of genetic effects shown in our ANT. This distributed-attention hypothesis is highly speculative and will require validation through more direct investigation.
Future studies should employ additional manipulations of attentional mechanisms for a comprehensive evaluation of attention effects. For example, the use of a dual-task paradigm (e.g., Li, VanRullen, Koch, & Perona, 2005) might help to elucidate the generality of our present findings. Moreover, investigations of brain activity by using fMRI and electroencephalography and/or magnetoencephalography (EEG/MEG) will help clarify the nature of top-down processing.
Future directions and extensions
For a complete understanding of the role of acetylcholine in rapid scene categorization and its relation to attention, further studies will be required. Firstly, because scene categorization relates to various brain regions and neural processes, cognitive investigations employing fMRI and EEG/MEG will be essential. In particular, the locus of the CHRNA4 polymorphism effect in the brain will provide important clues for understanding its relationship with attentional effects. For example, the genotype effect in early visual areas such as V1 favors the view that this effect is independent of attention. By contrast, the effect in the parietal lobe suggests that it may be a by-product of the attention effect. Secondly, it will be important to investigate a potential interactive effect of SNPs on CHRNA4 and CHRNA7, or on CHRM2. For example, CHRNA7 has an SNP affecting the nicotinic receptor subunit alpha 7. CHRM2 is also a gene for a type of acetylcholine receptor, a muscarinic receptor that is known to enhance the response of the receptive field to natural images (Goard & Dan, 2009). Investigations into the SNP interaction will require much larger sample sizes than those used in the present study (Greenwood et al., 2009). Thirdly, it will be desirable to obtain evidence for the nicotinic acetylcholine receptor effect by means of other experimental manipulations. For example, the effects of smoking on rapid scene categorization may be worth pursuing. Furthermore, replicating our present findings by properly controlling for potentially contaminating factors such as smoking habit, gender, or age may be necessary.
In the present study, we have demonstrated for the first time that rapid scene categorization performance is related to the nicotinic receptor subunit gene CHRNA4. Our findings contribute to the knowledge on the effects of polymorphisms in neurotransmitter genes, with respect to individual differences in cognition (e.g., Markett et al., 2009, 2011; Parasuraman et al., 2005). The effect size (η 2) in our study was moderately large (.0568), and therefore our data should be capable of being successfully replicated. We further revealed that the effect of the cholinergic receptor subunit gene CHRNA4 is not limited to attention. Elucidation of the effect of a novel polymorphism in a single gene will enhance our understanding of specific differences in basic research, thereby providing useful information for the development of clinical treatments (e.g., schizophrenia, epilepsy) and also for future experimental research.
References
Alvarez, G. A., & Scholl, B. J. (2005). How does attention select and track spatially extended objects? New effects of attentional concentration and amplification. Journal of Experimental Psychology, 134, 461–476.
Bachmann, T., Luiga, I., & Põder, E. (2005). Variations in backward masking with different masking stimuli: II. The effects of spatially quantised masks in the light of local contour interaction, interchannel inhibition, perceptual retouch, and substitution theories. Perception, 34, 139–153.
Bacon-Macé, N., Macé, M. J.-M., Fabre-Thorpe, M., & Thorpe, S. J. (2005). The time course of visual processing: Backward masking and natural scene categorisation. Vision Research, 45, 1459–1469. doi:10.1016/j.visres.2005.01.004
Brewer, W. F., & Treyens, J. C. (1981). Role of schemata in memory for places. Cognitive Psychology, 13, 1207–1230.
Codispoti, M., Ferrari, V., Junghöfer, M., & Schupp, H. T. (2006). The categorization of natural scenes: Brain attention networks revealed by dense sensor ERPs. NeuroImage, 32, 583–591. doi:10.1016/j.neuroimage.2006.04.180
Cohen, J. (1988). Statistical power analysis for the behavioral sciences (2nd ed.). Hillsdale: Erlbaum.
Davenport, J. L., & Potter, M. C. (2004). Scene consistency in object and background perception. Psychological Science, 15, 559–564. doi:10.1111/j.0956-7976.2004.00719.x
Disney, A. A., Aoki, C., & Hawken, M. J. (2007). Gain modulation by nicotine in macaque V1. Neuron, 56, 701–713.
Espeseth, T., Endestad, T., Rootwelt, H., & Reinvang, I. (2007). Nicotine receptor gene CHRNA4 modulates early event-related potentials in auditory and visual oddball target detection tasks. Neuroscience, 147, 974–985.
Espeseth, T., Greenwood, P. M., Reinvang, I., Fjell, A. M., Walhovd, K. B., Westyle, L. T., & Parasuraman, R. (2006). Interactive effects of APOE and CHRNA4 on attention and white matter volume in healthy middle-aged and older adults. Cognitive, Affective, & Behavioral Neuroscience, 6, 31–43. doi:10.3758/CABN.6.1.31
Espeseth, T., Sneve, M. H., Rootwelt, H., & Laeng, B. (2010). Nicotinic receptor gene CHRNA4 interacts with processing load in attention. PLoS One, 5, e14407. doi:10.1371/journal.pone.0014407
Everitt, B. J., & Robbins, T. W. (1997). Central cholinergic systems and cognition. Annual Review of Psychology, 48, 649–684.
Fan, J., Byrne, J., Worden, M. S., Guise, K. G., McCandliss, B. D., Fossella, J., & Posner, M. I. (2007). The relation of brain oscillations to attentional networks. Journal of Neuroscience, 27, 6197–6206. doi:10.1523/JNEUROSCI.1833-07.2007
Feng, Y., Niu, T., Xing, H., Xu, X., Chen, C., Peng, S., & Xuet, X. (2004). A common haplotype of the nicotine acetylcholine receptor α4 subunit gene is associated with vulnerability to nicotine addiction in men. American Journal of Human Genetics, 75, 112–121.
Feng, Y., & Walsh, C. A. (2001). Protein–protein interactions, cytoskeletal regulation and neuronal migration. Nature Reviews Neuroscience, 2, 408–416.
Fize, D., Fabre-Thorpe, M., Richard, G., Doyon, B., & Thorpe, S. J. (2005). Rapid categorization of foveal and extrafoveal natural images: Associated ERPs and effects of lateralization. Brain and Cognition, 59, 145–158.
Fossella, J., Sommer, T., Fan, J., Wu, Y., Swanson, J. M., Pfaff, D. W., & Posner, M. I. (2002). Assessing the molecular genetics of attention networks. BMC Neuroscience, 3, 1–11.
Giocomo, L. M., & Hasselmo, M. E. (2005). Nicotinic modulation of glutamatergic synaptic transmission in region CA4 of the hippocampus. European Journal of Neuroscience, 22, 1349–1356.
Goard, M., & Dan, Y. (2009). Basal forebrain activation enhances cortical coding of natural scenes. Nature Neuroscience, 12, 1444–1449.
Green, A. E., Munafò, M. R., DeYoung, C. G., Fossella, J. A., Fan, J., & Gray, J. R. (2008). Using genetic data in cognitive neuroscience: From growing pains to genuine insights. Nature Reviews Neuroscience, 9, 710–720.
Greene, M. R., & Oliva, A. (2009). The briefest of glances: The time course of natural scene understanding. Psychological Science, 20, 464–472. doi:10.1111/j.1467-9280.2009.02316.x
Greenwood, P. M., Fossella, J. A., & Parasuraman, R. (2005). Specificity of the effect of a nicotinic receptor polymorphism on individual differences in visuospatial attention. Journal of Cognitive Neuroscience, 17, 1611–1620.
Greenwood, P. M., Lin, M.-K., Sundararajan, R., Fryxell, K. J., & Parasuraman, R. (2009). Synergistic effects of genetic variation in nicotinic and muscarinic receptors on visual attention but not working memory. Proceedings of the National Academy of Sciences, 106, 3633–3638. doi:10.1073/pnas.0807891106
Greenwood, P. M., & Parasuraman, R. (2003). Normal genetic variation, cognition, and aging. Behavioral and Cognitive Neuroscience Reviews, 2, 278–306.
Greenwood, P. M., Sundararajan, R., Lin, M.-K., Kumar, R., Fryxell, K. J., & Parasuraman, R. (2008). Both a nicotinic SNP and a noradrenergic SNP modulate working memory performance when attention is manipulated. Journal of Cognitive Neuroscience, 21, 2139–2153. doi:10.1162/jocn.2008.21164
Greenwood, P. M., Sunderland, T., Friz, J. L., & Parasuraman, R. (2000). Genetics and visual attention: Selective deficits in healthy adult carriers of the varepsilon 4 allele of the apolipoprotein E gene. Proceedings of the National Academy of Sciences, 97, 11661–11666.
Hughes, H. C., Nozawa, G., & Kitterle, F. (1996). Global precedence, spatial frequency channels, and the statistics of natural images. Journal of Cognitive Neuroscience, 8, 197–230. doi:10.1162/jocn.1996.8.3.197
Ioannidis, J. P., Ntzani, E. E., Trikalinos, T. A., & Contopoulos-Ioannidis, D. G. (2001). Replication validity of genetic association studies. Nature Genetics, 29, 306–309.
Joubert, O., Rousselet, G., Fize, D., & Fabre-Thorpe, M. (2007). Processing scene context: Fast categorization and object interference. Vision Research, 47, 3286–3297.
Kincses, T. Z., Chadaide, Z., Varga, E. T., Antal, A., & Paulus, W. (2006). Task-related temporal and topographical changes of cortical activity during ultra-rapid visual categorization. Brain Research, 1112, 191–200.
Kuryatov, A., Gerzanich, V., Nelson, M., Olale, F., & Lindstrom, J. (1997). Mutation causing autosomal dominant nocturnal frontal lobe epilepsy alters Ca2+ permeability, conductance, and gating of human α4β2 nicotinic acetylcholine receptors. Journal of Neuroscience, 17, 9035–9047.
Li, F. F., VanRullen, R., Koch, C., & Perona, P. (2002). Rapid natural scene categorization in the near absence of attention. Proceedings of the National Academy of Sciences, 99, 9596–9601.
Li, F. F., VanRullen, R., Koch, C., & Perona, P. (2005). Why does natural scene categorization require little attention? Exploring attentional requirements for natural and synthetic stimuli. Visual Cognition, 12, 893–924.
Loftus, G. R., & Ginn, M. (1984). Perceptual and conceptual masking of pictures. Journal of Experimental Psychology: Learning, Memory, and Cognition, 10, 435–441.
Loschky, L. C., Sethi, A., Simons, D. J., Pydimarri, T. N., Ochs, D., & Corbeille, J. L. (2007). The importance of information localization in scene gist recognition. Journal of Experimental Psychology. Human Perception and Performance, 33, 1431–1450. doi:10.1037/0096-1523.33.6.1431
Macé, M. J.-M., Joubert, O. R., Nespoulous, J.-L., & Fabre-Thorpe, M. (2009). The time-course of visual categorizations: You spot the animal faster than the bird. PLoS One, 4, e5927. doi:10.1371/journal.pone.0005927
Markett, S. A., Montag, C., & Reuter, M. (2009). The association between dopamine DRD2 polymorphisms and working memory capacity is modulated by a functional polymorphism on the nicotinic receptor gene CHRNA4. Journal of Cognitive Neuroscience, 22, 1944–1954.
Markett, S., Montag, C., & Reuter, M. (2011). The nicotinic acetylcholine receptor gene CHRNA4 is associated with negative emotionality. Emotion, 11, 450–455.
Michaels, C. F., & Turvey, M. T. (1979). Central sources of visual masking: Indexing structures supporting seeing at a single, brief glance. Psychological Research, 41, 1–61.
Oliva, A., & Torralba, A. (2001). Modeling the shape of the scene: A holistic representation of the spatial envelope. International Journal of Computer Vision, 42, 145–175.
Parasuraman, R., Greenwood, P. M., Kumar, R., & Fossella, J. (2005). Beyond heritability: Neurotransmitter genes differentially modulate visuospatial attention and working memory. Psychological Science, 16, 200–207. doi:10.1111/j.0956-7976.2005.00804.x
Pilz, A. J., Willer, E., Povey, S., & Abbott, C. M. (1992). The genes coding for phosphoenolpyruvate carboxykinase-1 (PCK1) and neuronal nicotinic acetylcholine receptor α4 subunit (CHRNA4) map to human chromosome 20, extending the known region of homology with mouse chromosome 2. Annals of Human Genetics, 56, 289–293.
Posner, M. I., & Petersen, S. E. (1990). The attention system of the human brain. Annual Review of Neuroscience, 13, 25–42. doi:10.1146/annurev.ne.13.030190.000325
Potter, M. C. (1975). Meaning in visual scenes. Science, 187, 965–966. doi:10.1126/science.1145183
Potter, M. C. (1976). Short-term conceptual memory for pictures. Journal of Experimental Psychology: Human Learning and Memory, 2, 509–522.
Radcliffe, K. A., & Dani, J. A. (1998). Nicotinic stimulation produces multiple forms of increased glutamatergic synaptic transmission. Journal of Neuroscience, 18, 7075–7083.
Sadr, J., & Sinha, P. (2004). Object recognition and random image structure evolution. Cognitive Science, 28, 259–287.
Schyns, P. G., & Oliva, A. (1994). From blobs to boundary edges: Evidence for time- and spatial-scale-dependent scene recognition. Psychological Science, 5, 195–200. doi:10.1111/j.1467-9280.1994.tb00500.x
Steinlein, O. K. (2004). Genetic mechanisms that underlie epilepsy. Nature Reviews Neuroscience, 5, 400–408.
Steinlein, O. K., Deckert, J., Nothen, M. M., Franke, P., Maier, W., Beckmann, H., & Propping, P. (1997). Neuronal nicotinic acetylcholine receptor α4 subunit (CHRNA4) and panic disorder: An association study. American Journal of Medical Genetics, 74, 199–201.
Thorpe, S., Fize, D., & Marlot, C. (1996). Speed of processing in the human visual system. Nature, 381, 520–552.
Torralba, A., Oliva, A., Castelhano, M. S., & Henderson, J. M. (2006). Contextual guidance of eye movements and attention in real-world scenes: The role of global features in object search. Psychological Review, 113, 766–786. doi:10.1037/0033-295X.113.4.766
Treisman, A. (2006). How the deployment of attention determines what we see. Visual Cognition, 14, 411–443. doi:10.1080/13506280500195250
Voytko, M. L. (1996). Cognitive functions of the basal forebrain cholinergic system in monkeys: Memory or attention? Behavioural Brain Research, 75, 13–25.
Weiland, S., Witzemann, V., Villarroel, A., Propping, P., & Steinlein, O. (1996). An amino acid exchange in the second transmembrane segment of a neuronal nicotinic receptor causes partial epilepsy by altering its desensitization kinetics. Federation of European Biochemical Societies Letters, 398, 91–96.
Winterer, G., Musso, F., Konrad, A., Vucurevic, G., Stoeter, P., Sander, T., & Gallinat, J. (2007). Association of attentional network function with exon 5 variations of the CHRNA4 gene. Human Molecular Genetics, 16, 2165–2174.
Winterer, G., Rujescu, D., Wagner, M., Gallinat, J., Maier, W., Wienker, T., . . . Steinlein, O. (2011, May). CHRNA4 genotype predicts nicotinic receptor and brain function as well as clinical outcome in schizophrenia. Paper presented at the Nicotinic Acetylcholine Receptors 2011 Conference, Hinxton, Cambridge, UK.
Author Note
This research was supported by Grant No. 21650056 from the Japan Society for the Promotion of Science, awarded to J.S., and by a Grant-in-Aid for Global COE (Centers of Excellence) Program (D07) funded by the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
Yuichiro Kikuno, Graduate School of Human and Environmental Studies, Kyoto University; Tetsuro Matsunaga, AJINOMOTO Integrative Research for Advanced Dieting, Graduate School of Agriculture, Kyoto University; Jun Saiki, Graduate School of Human and Environmental Studies, Kyoto University
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kikuno, Y., Matsunaga, T. & Saiki, J. Polymorphism in the CHRNA4 gene is associated with rapid scene categorization performance. Atten Percept Psychophys 75, 1427–1437 (2013). https://doi.org/10.3758/s13414-013-0486-1
Published:
Issue Date:
DOI: https://doi.org/10.3758/s13414-013-0486-1