Abstract
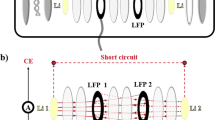
Although the three-electrode electrochemical cell is considered to be a powerful cell configuration for electrochemical impedance measurements, many researchers have extensively used the two-electrode electrochemical cell for the sake of convenience. In this work, the impedance of the lithium metal counter electrode that is frequently used to explore the electrochemical properties of the working electrode containing active materials in the field of lithium batteries was measured on the basis of a threelectrode electrochemical cell, and its contribution to the full cell impedance was carefully investigated. In particular, the effect of the counter electrode on the full cell impedance was quantitatively analyzed in the cases of cell aging and a low cell temperature. From the experimental results, it was demonstrated that inappropriate conclusions might be drawn in the case where a two-electrode electrochemical cell configuration is used to explore the impedance behavior of the active materials.
Similar content being viewed by others
References
Y. H. Oh, D. G. Ahn, S. H. Nam, C. J. Kim, J. G. Lee, and B. W. Park, Electron. Mater. Lett. 4, 103 (2008).
E. Barsoukov and J. R. Macdonald, Impedance Spectroscopy Theory, Experiment, and applications 2 nd ed., John Wiley & Sons, Inc., Hoboken, New Jersey (2005).
J. Fan and S. Tan, J. Electrochem. Soc. 153, A1081 (2006).
D. P. Abraham, J. L. Knuth, D. W. Dees, I. Bloom, and J. P. Christophersen, J. Power Sources 170, 465 (2007).
D. P. Abraham, J. R. Heaton, S.-H. Kang, D. W. Dees, and A. N. Jansen, J. Electrochem. Soc. 155, A41 (2008).
J. Huang and Z. Jiang, Electrochim. Acta 53, 7756 (2008).
D.-K. Kang and H.-C. Shin, J. Solid State Electrochem. 11, 1405 (2007).
A. Blyr, C. Sigala, G. Amatucci, D. Guyomard, Y. Chabre, and J.-M. Tarascon, J. Electrochem. Soc. 145, 194 (1998).
M. Dolle, F. Orsini, A.S. Gozdz, and J.-M. Tarascon, J. Electrochem. Soc. 148, A851 (2001).
H.-M. Cho and H.-C. Shin, Analysis of Cell Impedance for the Design of High-Power Lithium-Ion Battery in Recent Research and Progress in Lithium Batteries (ed., F. Columbus), Nova Science, New York (2010) (in press).
H.-M. Cho, Y. J. Park, and H.-C. Shin, J. Electrochem. Soc. 157(1), A8 (2010).
D. A. Harrington, and B. E. Conway, Electrochim. Acta. 32, 1703 (1987).
C. Gabrielli, P. Mocoteguy, H. Perrot, D. Nieto-Sanz, and A. Zdunek, J. Appl. Electrochem. 38, 457 (2008).
M. D. Levi, G. Salitra, B. Markovsky, H. Teller, D. Aurbach, U. Heider and L. Heider, J. Electrochem. Soc. 146, 1279 (1999).
Y.-M. Choi, S.-I. Pyun, J.-S. Bae, and S.-I. Moon, J. Power Sources 56, 25 (1995).
E. Barsoukov and J. R. Macdonald, Impedance Spectroscopy Theory, Experiment, and applications 2 nd ed., p. 307, John Wiley & Sons, Inc., Hoboken, New Jersey (2005).
C. Ho, I. D. Raistrick, and R. A. Huggins, J. Electrochem. Soc. 127, 343 (1980).
M. D. Levi and D. Aurbach, J. Phys. Chem. B 101, 4641 (1997).
T. Pajkossy and L. Nyikos, Electrochim. Acta. 30, 1533 (1985).
S. H. Liu, Phys. Rev. Lett. 55, 529 (1985).
B. Sapoval, Solid State Ionics 23, 253 (1987).
T. Pajkossy and L. Nyikos, J. Electrochem. Soc. 133, 2061 (1986).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cho, HM., Park, Y.J., Yeon, JW. et al. In-depth investigation on two- and three-electrode impedance measurements in terms of the effect of the counter electrode. Electron. Mater. Lett. 5, 169–178 (2009). https://doi.org/10.3365/eml.2009.12.169
Issue Date:
DOI: https://doi.org/10.3365/eml.2009.12.169