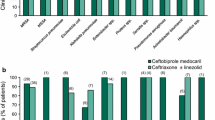
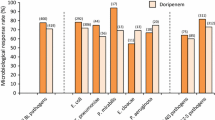
Summary
Cefotaxime, a third generation cephalosporin, is used throughout the world over a wide range of doses. The purpose of this paper is to discuss the rationale for determination of the optimal dosage and of adequate modes of administration. Among the factors determining in vivo activity, the most important are: (1) the time dependence of the antibacterial effect of cephalosporins, (2) the limited effect of increasing the drug concentration in contact with the bacteria and (3) the absence of a significant post-antibiotic effect. Combined with the rather short elimination half-life of cefotaxime, these factors argue for the use of a unitary dose of 1g in adult patients and for a 6-or 8-hour interval between doses. Information obtained from various animal models of infection are discussed. Clinical and bacteriological studies published in the international literature report a high rate of cure (between 80 and 100% ) according to the type of infection and to the criteria of efficacy, with daily doses ranging from 2 to 4g bid or aid. The results obtained with the lowest doses are detailed, particularly for infections permitting the use of a low dosage. The necessity for increasing the dose is discussed in the following situations: (1) in specific infections requiring high local drug concentrations such as meningitis and endocarditis, (2) against micro-organisms exhibiting moderate susceptibility to cefotaxime (MIC ⩾1 mg/L) and (3) in immunocompromised patients.
It is now well established that third generation cephalosporins have to be combined with other antimicrobial agents (e.g. aminoglycosides) for the treatment of patients with infections caused by bacteria able to become resistant. For susceptible strains, it has not been established that a Synergistic effect of cefotaxime with another agent allows a reduction of the dosage of each member of the combination.
Similar content being viewed by others
References
Bassaris HP, Lianon PE, Volta EG, Papavasition JTh. Effects of subinhibitory concentrations of cefotaxime on adhesion and polymorphonuclear leucocyte function with Gram-negative bacteria. Journal of Antimicrobial Chemotherapy 14 (Suppl. B): 91–96, 1984
Bundtzen RW, Gerber AV, Cohn DL, Craig WA. Post antibiotic suppression of bacterial growth. Review of Infectious Diseases 3: 28–37, 1981
Carbon C, Manuel Ch. L’évaluation de la posologie des antibiotiques. Journal de Pharmacologie 17 (Suppl. 1): 51–52, 1986
Carbon C, Auboyer C, Becq-Giraudon B, Motin JP, Vachon F, et al. Cefotaxime vs cefotaxime + amikacin in the treatment of septicaemia due to enterobacteria: a multicentric prospective study. Fifth Mediterranean Congress of Chemotherapy, Cairo. Abstract no. 01, 1986
Carmine AA, Brogden RN, Heel RC, Speight TM, Avery GS. Cefotaxime: a review of its antibacterial activity, pharmacological properties and therapeutic use. Drugs 25: 223–289, 1983
Cherubin CE, Corrado ML, Nai SR, Gombert ME, Landsman S, et al. Treatment of Gram-negative bacillary meningitis: role of the new cephalosporin antibiotics. Reviews of Infectious Diseases 4 (Suppl.): S453–S464, 1982
Craig WA, Vogelman B. The post-antibiotic effect. Annals of Internal Medicine 106: 900–902, 1987
Durack DT, Beeson PB. Experimental bacterial endocarditis. II. Survival of a bacteria in endocardial vegetations. British Journal of Experimental Pathology 53: 50–53, 1972
Eagle H, Fleischman R, Levy M. ‘Continuous’ vs ‘discontinuous’ therapy with penicillin. The effect of the interval between injections on therapeutic efficacy. New England Journal of Medicine 248: 481–488, 1953
Eng RHK, Cherubin C, Smith SM, Buccini F. Examination of Gram-negative bacilli from meningitis patients who failed or relapsed on moxalactam therapy. Antimicrobial Agents and Chemotherapy 26: 850–856, 1984
Frimodt-Möller N, Bentzon WM, Thomsen VF. Experimental infection with Streptococcus pneumoniae in mice: correlation of in vitro activity and pharmacokinetic parameters with in vivo effect for 14 cephalosporins. Journal of Infectious Diseases 154: 511–517, 1986
Gengo FM, Mannion TW, Nightingale CH, Schentag JJ. Integration of pharmacokinetics and pharmacodynamics of methicillin in curative treatment of experimental endocarditis. Journal of Antimicrobial Chemotherapy 14: 619–621, 1984
Ings RM, Fillastre JP, Godin M, Leroy A, Humbert G. The pharmacokinetics of cefotaxime and its metabolites in subjects with normal and impaired renal function. Reviews of Infectious Diseases 4 (Suppl.); S379–S391, 1982
Jenkinson SG, Briggs MS, Bryn RD. Cefotaxime in the treatment of pneumococcal pneumonia. Journal of Antimicrobial Chemotherapy 6 (Suppl. A): 177–180, 1980
Jones RD, Thornsberry C. Cefotaxime: a review of in vitro antimicrobial properties. Reviews of Infectious Diseases 4 (Suppl.): S316–S325, 1982
Kawada Y, Shimizu Y, Nishiura T. Comparative studies of cefotaxime and sulbenicillin in complicated urinary tract infections. Journal of Antimicrobial Chemotherapy 6 (Suppl. A): 213–218, 1980
Kermarec J, Sauvaget J. Le céfotaxime dans le traitement des affections bronchopulmonaires. Nouvelle Presse Médicale 10: 650–653, 1981
Klastersky J, Glauser MP, Schimpff SC, Zinner SH, Gaya H. Prospective randomized comparison of three antibiotic regimens for empirical therapy of suspected bacteremic infection in febrile granulocytopenic patients. Antimicrobial Agents and Chemotherapy 29: 263–270, 1986
Kunin CC. Dosage schedules of antimicrobial agents: a historical review. Review of Infectious Diseases 3: 4–11, 1981
Lavoie GY, Bergeron MG. Influence of four modes of administration on penetration of aztreonam, cefuroxime, and ampicillin into interstitial fluid and fibrin-clots, and on in vivo efficacy against Haemophilus influenzae. Antimicrobial Agents and Chemotherapy 28: 404–412, 1985
Ludwig G, Knebel L. Cefotaxime in urinary tract infections. Comparative clinical studies with gentamicin and with cefotaxime. Journal of Antimicrobial Chemotherapy 6 (Suppl. A): 207–221, 1980
Maesen FPV, Davies BI, Drenth BMH, Elfers H. Treatment of acute exacerbations of chronic bronchitis.with cefotaxime: a controlled clinical trial. Journal of Antimicrobial Chemotherapy 6 (Suppl. A): 187–192, 1980
Mattie H, Kunst MW. Animal models for the assessment of potentiation of antibiotics by probenecid and by host resistance. Infection 6 (Suppl. 1): S36–S37, 1978
Miki F, Shiota K. Cefotaxime in lower respiratory tract infections compared to cefazolin. Journal of Antimicrobial Chemotherapy 6 (Suppl. A): 169–175, 1980
Newson SWB, Mattews J, Pearce VR. Cefotaxime for urinary tract infections. Journal of Antimicrobial Chemotherapy 6 (Suppl. A): 199–203, 1980
Nordbring F. Current practice in penicillin dosing. Journal of Antimicrobial Chemotherapy 8 (Suppl. C): 1–6, 1981
Pangon B, Joly V, Vallois JM, Abel L, Buré A, et al. Comparative efficacy of cefotiam, cefmenoxime, and ceftriaxone in experimental endocarditis and correlation with pharmacokinetics and in vitro efficacy. Antimicrobial Agents and Chemotherapy 4: 518–522, 1987
Parker RH. Effect of frequency of administration on therapeutic efficacy of cefotaxime. Clinical Therapeutics 6: 488–499, 1984
Perkins RL. Clinical trials of cefotaxime for the treatment of bacterial infections of the lower respiratory tract. Reviews of Infectious Diseases 4 (Suppl.): S421–S431, 1982
Prat V, Horcickova M, Halala M. Single dose treatment with netilmicin for different clinical forms of urinary tract infections. Infection 12: 99–104, 1984
Prat V, Horcickova M, Milotova Z, Matousovic K. Single dose cefotaxime in the treatment of urinary tract infection with 1g of cefotaxime. In Neu (Ed.) Cefotaxime: a selection of papers presented at the 14th International Congress of Chemotherapy, Kyoto, pp. 112–115, Excerpta Medica, 1985
Repetto HA, MacLoughlin GJF. Single-dose cefotaxime in the treatment of urinary tract infections in children: a randomized clinical trial. Journal of Antimicrobial Chemotherapy 14 (Suppl. B): 307–310, 1984
Sheehan G, Marding GKM, Ronald AR. Advances in the treatment of urinary tract infection. American Journal of Medicine 76 (Suppl. 5A): 141–147, 1984
Smith CR, Ambinder R, Lipsky JJ, Petty BG, Israel E, et al. Cefotaxime compared with nafcillin plus tobramycin for serious bacterial infections. Annals of Internal Medicine 101: 469–477, 1982
Sow AB. Cefotaxime treatment of meningitis in children. Journal of Antimicrobial Chemotherapy 14 (Suppl. B): 191–194, 1984
Stolz E, Oney L, Van Joost T, Michel MF. Treatment of non complicated urogenital, rectal and oropharyngeal gonorrhea with intramuscular cefotaxime l.0g and cefuroxime 1.5g. Journal of Antimicrobial Chemotherapy 14 (Suppl. B): 295–299, 1984
Tauber MG, Sande MA. Principles in the treatment of bacterial meningitis. American Journal of Medicine 76 (Suppl. 5A): 224–230, 1984
Thornsberry C, Jones RN, Barry AL, Fuchs PC. Antimicrobial susceptibility tests with cefotaxime and correlation with clinical bacteriologic response. Reviews of Infectious Diseases 4 (Suppl.): S316–S324, 1982
Viladrich PF, Pallares R, Ariza J, Rufi G, Gudial F. Four days of penicillin therapy for meningitis. Archives of Internal Medicine 146: 2380–2382, 1986
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Simon, A., d’Aubrac, C.A., Safran, C. et al. Cefotaxime Optimal Dosage in Adult Patients. Drugs 35 (Suppl 2), 221–230 (1988). https://doi.org/10.2165/00003495-198800352-00049
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-198800352-00049