Summary
Significant advances have been made in our understanding of the clinical pharmacokinetics of pethidine (meperidine) since it was reviewed by Mather and Meffin in 1978. This review attempts to evaluate recent information and assess the potential clinical implications.
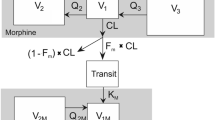
Pethidine is eliminated mainly via biotransformation — norpethidine, norpethidinic acid and pethidinic acid being the major metabolites. Blood clearance of this drug is approximately 800 to 900ml/min in healthy volunteers (elimination half-life 3 to 8 hours). Clearance appears to be reduced postoperatively in surgical patients by approximately 25% but is reduced by about 50 % in patients with cirrhosis. Oral bioavailability averages 56 % in healthy subjects, but increases to 80 to 90% in cirrhosis. The free fraction of pethidine is 0.2 to 0.4, the plasma concentration of α1-acid glycoprotein being its most important determinant.
Studies (primarily in patients following abdominal surgery) suggest that blood pethidine concentrations of 0.5 to 0.7 μg/ml are required for analgesia. The minimum analgesic concentration appears to be quite consistent within an individual, but varies between patients. Importantly, the concentration-analgesic response relationship for pethidine is extremely steep, with concentration differences as small as 0.05 μg/ml representing the difference between no analgesia and complete suppression of pain. Dosage regimens of pethidine which produce variable or fluctuating blood concentrations can be expected to achieve inconsistent relief of pain. Therefore, the use of continuous intravenous infusions of pethidine appears to be a rational new approach to acute pain management. Further studies assessing the effect of metabolites, protein binding, age, personality and source of pain on the pethidine concentrationanalgesic response relationship are needed.
Similar content being viewed by others
References
Aronsen, K.F.; Ekelund, G.; Kindmark, C.O. and Laurell, C.B.: Sequential changes of plasma proteins after surgical trauma. Scandinavian Journal of Clinical and Laboratory Investigation 29 (Suppl 124): 127–136 (1972).
Asatoor, A.M.; London, D.R.; Milne, M.D. and Simenhoff, M.L.: The excretion of pethidine and its derivatives. British Journal of Pharmacology 20: 285–298 (1963).
Austin, K.L.; Stapleton, J.V. and Mather, L.E.: Multiple intramuscular injections: A major source of variability in analgesic response to meperidine. Pain 8: 47–62 (1980a).
Austin, K.L.; Stapleton, J.V. and Mather, L.E.: Pethidine clearance during continuous intravenous infusions in postoperative patients. British Journal of Clinical Pharmacology 8: 25–30 (1981a).
Austin L.K.; Stapleton, J.V. and Mather, L.E.: Relationship between blood meperidine concentrations and analgesic response: A preliminary report. Anesthesiology 53: 460–466 (1980b).
Austin, K.L.; Stapleton, J.V. and Mather, L.E.: Rate of formation of norpethidine from pethidine. British Journal of Anaesthesia 53: 255–257 (1981b).
Belfrage, P.; Boreus, L.O.; Hartvig, P.; Irestedt, L. and Raabe, N.: Neonatal depression after obstetrical analgesia with pethidine. The role of the the injection-delivery time interval and of the plasma concentrations of pethidine and norpethidine. Acta Obstetrica et Gynecologica Scandinavica 60: 43–49 (1981).
Borga, O.; Piafsky, K.M. and Nilsen, O.G.: Plasma protein binding of basic drugs. I. Selective displacement from α1-acid glycoprotein by tris (2-butoxyethyl) phosphate. Clinical Pharmacology and Therapeutics 22: 539–544 (1977).
Burns, J.J.; Berger, B.L.; Lief, P.A.; Wollack, A.; Papper, E.M. and Brodie, B.B.: The physiological disposition and fate of meperidine (Demerol) in man and a method for its estimation in plasma. Journal of Pharmacology and Experimental Therapeutics 114: 259–298 (1955).
Chan, K.: The effects of physiochemical properties of pethidine and its basic metabolites on their buccal absorption and renal elimination. Journal of Pharmacy and Pharmacology 31: 672–675 (1979).
Clark, B. and Thompson, J.W.: Analysis of the inhibition of pethidine N-demethylation by monoamine oxidase inhibitors and some other drugs with special reference to drug interactions in man. British Journal of Pharmacology 44: 89–99 (1972).
Clements, J.A.; Heading, R.C.; Nimmo, W.S. and Prescott, L.F.: Kinetics of acetaminophen absorption and gastric emptying in man. Clinical Pharmacology and Therapeutics 24: 420–431 (1978).
Crawford, J.S. and Rudofsky, S.: The placental transfer of pethidine. British Journal of Anaesthesia 37: 929–933 (1965).
Dahlstrom, B.E.; Paalzow, L.K.; Lindberg, C. and Bogentoft, C.: Pharmacokinetics and analgesic effect of pethidine (meperidine) and its metabolites in the rat. Drug Metabolism and Disposition 7: 108–112 (1979).
Edwards, D.J.; Lalka, D.; Cerra, F. and Slaughter, R.: AIpha1-acid glycoprotein concentration and protein binding in trauma. Clinical Pharmacology and Therapeutics 31: 62–67 (1982).
Fung, D.L.; Asling, J.H.; Eisele, J.H. and Martucci, R.: A comparison of alphaprodine and meperidine pharmacokinetics. Journal of Clinical Pharmacology 20: 37–41 (1980).
Gilbert, P.E. and Martin, W.R.: Antagonism of the convulsant effects of heroin, d-propoxyphene, meperidine, normeperidine and thebaine by naloxone in mice. Journal of Pharmacology and Experimental Therapeutics 192: 538–541 (1975).
Goolkasian, D.L.; Slaughter, R.L.; Edwards, D.J. and Lalka, D.: Displacement of lidocaine from serum α1,-acid glycoprotein binding sites by basic drugs. Clinical Research (In press 1982).
Hodgkinson, R.; Bhatt, M. and Wang, C.N.: Double-blind comparison of the neurobehavior of neonates following the administration of different doses of meperidine to the mother. Canadian Anaesthetists’ Society Journal 25: 405–411 (1978).
Jackson, A.J.: Determination of meperidine blood to plasma concentration ratio (γ) — Effect of temperature and pH. Research Communications in Chemical Pathology and Pharmacology 32: 365–368 (1981).
Klotz, U.; McHorse, T.S.; Wilkinson, G.R. and Schenker, S.: The effect of cirrhosis on the disposition and elimination of meperidine in man. Clinical Pharmacology and Therapeutics 16: 667–675 (1974).
Koska, A.J.; Kramer. W.G.; Romagnoli, A.; Keats, A.S. and Sabawala, P.B.: Pharmacokinetics of high-dose meperidine in surgical patients. Anesthesia and Analgesia 60: 8–11 (1981).
Kuhnert, B.R.; Kuhnert, P.M.; Tu, AL. and Lin, D.C.K.: Meperidine and normeperidine levels following meperidine administration during labor. II. Fetus and neonate. American Journal of Obstetrics and Gynecology 133: 909–914 (1979).
Kuhnert, B.R.; Kuhnert, P.M.; Prochaska, A.L. and Sokol, R.J.: Meperidine disposition in mother, neonate and non-pregnant females. Clinical Pharmacology and Therapeutics 27: 486–491 (1980).
Lindberg, C.; Bondesson, U. and Hartvig, P.: Investigation of the urinary excretion of pethidine and five of its metabolites in man using selected ion monitoring. Biomedical Mass Spectrometry 7: 88–92 (1980).
Mather, L.E. and Tucker, G.T.: Systemic availability of orally administered meperidine. Clinical Pharmacology and Therapeutics 20: 535–540 (1976).
Mather, L.E. and Meffin, P.J.: Clinical pharmacokinetics of pethidine. Clinical Pharmacokinetics 3: 352–368 (1978).
Mather, L.E.; Tucker, G.T.; Pflug, A.E.; Lindop, M.J. and Wilkerson, C.: Meperidine kinetics in man. Intravenous injection in surgical patients and volunteers. Clinical Pharmacology and Therapeutics 17: 21–30 (1975a).
Mather, L.E.; Lindop, M.J.; Tucker, G.T. and Pflug, A.E.: Pethidine revisited: Plasma concentrations and effects after intramuscular injection. British Journal of Anaesthesia 47: 1269–1275 (1975b).
McHorse, T.S.; Wilkinson, G.R.; Johnson, R.F. and Schenker, S.: Effect of acute viral hepatitis in man on the disposition and elimination of meperidine. Gastroenterology 68: 775–780 (1975).
Miller, J.W. and Anderson, H.H.: The effect of N-demethylation on certain pharmacological actions of morphine, codeine and meperidine in the mouse. Journal of Pharmacology and Experimental Therapeutics 112: 191–196 (1954).
Moore, J.; McNabb, T.G. and Glynn, J.P.: The placental transfer of pentazocine and pethidine. British Journal of Anaesthesia 45 (Suppl.): 798–801 (1973).
Morrison, J.C.; Wiser, W.L.; Rosser, S.I.; Gayden, J.O.; Bucovaz, E.T.; Whybrew, W.D. and Fish, S.A.: Metabolites of meperidine related to fetal depression. American Journal of Obstetrics and Gynecology 115: 1132–1137 (1973).
Morrison, J.C.; Whybrew, W.D.; Rosser, S.I.; Bucovaz, E.T.; Wiser, W.L. and Fish, S.A.: Metabolites of meperidine in the fetal and maternal serum. American Journal of Obstetrics and Gynecology 126: 997–1002 (1976).
Morselli, P.L. and Rovei, V.: Placental transfer of pethidine and norpethidine and their pharmacokinetics in the newborn. European Journal of Clinical Pharmacology 18: 25–30 (1980).
Nation, R.L.: Meperidine binding in maternal and fetal plasma. Clinical Pharmacology and Therapeutics 29: 472–479 (1981).
Neal, E.A.; Meffin, P.J.; Gregory, P.B. and Blaschke, T.F.: Enhanced bioavailability and decreased clearance of analgesics in patients with cirrhosis. Gastroenterology 77: 96–102 (1979).
Nimmo, W.S.; Wilson, J. and Prescott, L.F.: Narcotic analgesics and delayed gastric emptying during labour. Lancet I: 890–892 (1975).
Plotnikoff, N.P.; Way, E.L. and Elliot, H.W.: Biotransformation products of meperidine excreted in the urine of man. Journal of Pharmacology and Experimental Therapeutics 117: 414–419 (1956).
Pond, S.M. and Kretschzmar, K.M.: Effect of Phenytoin on meperidine clearance and normeperidine formation. Clinical Pharmacology and Therapeutics 30: 680–686 (1981b).
Pond, S.M.; Tong, T.; Benowitz, N.L. and Jacob, P.: Enhanced bioavailability of pethidine and pentazocine in patients with cirrhosis of the liver. Australia-New Zealand Journal of Medicine 10: 515–519 (1980).
Pond, S.M.; Tong, T.; Benowitz, N.L.; Jacob, P. and Rigod, J.: Presystemic metabolism of meperidine to normeperidine in normal and cirrhotic subjects. Clinical Pharmacology and Therapeutics 30: 183–188 (1981).
Rigg, J.R.A.; Llsley, A.H. and Vedig, A.E.: Relationship of ventilatory depression to steady-state blood pethidine concentrations. British Journal of Anaesthesia 53: 613–619 (1981).
Shih, A.P.L.; Robinson, K. and Au, W.Y.W.: Determination of therapeutic serum concentrations of oral and parenteral meperidine by gas liquid chromatography. European Journal of Clinical Pharmacology 9: 451–456 (1976).
Shnider, S.M. and Moya, F.: Effects of meperidine on the newborn infant. American Journal of Obstetrics and Gynecology 89: 1009–1014 (1964).
Stambaugh, J.E. and Wainer, I.W.: Drug interaction: Meperidine and chlorpromazine, a toxic combination. Journal of Clinical Pharmacology 71: 140–146 (1981).
Stambaugh, J.E., Wainer, I.W.; Stanstead, J.K. and Hemphill, D.M.: The clinical pharmacology of meperidine — comparison of routes of administration. Journal of Clinical Pharmacology 16: 245–256 (1976).
Stambaugh, J.E.; Wainer, I.W., Hemphill, D.M. and Schwartz, I.: A potentially toxic drug interaction between pethidine (meperidine) and phenobarbitone. Lancet 1: 398–399 (1977).
Stambaugh, J.E.; Wainer, I.W. and Schwartz, I.: The effect of phenobarbital on the metabolism of meperidine in normal volunteers. Journal of Clinical Pharmacology 18: 482–490 (1978).
Stapleton, J.V.; Austin, K.L. and Mather, L.E.: A pharmacokinetic approach to post-operative pain: Continuous infusion of pethidine. Anaesthesia and Intensive Care 7: 25–32 (1979).
Szeto, H.H.; Inturrisi, C.E.; Houde, R.; Saal, S.: Cleigh, J. and Reidenberg, M.M.: Accumulation of normeperidine, an active metabolite of meperidine, in patients with renal failure or cancer. Annals of Internal Medicine 86: 738–741 (1977).
Szeto, H.H.; Clapp, J.F.; Abrams, R.; Inturrisi, C.E.; Kaiko, R.F.; Larrow, R.W. and Mann, L.I.: Brain uptake of meperidine in the fetal lamb. American Journal of Obstetrics and Gynecology 138: 528–533 (1980).
Tamsen, A.; Hartvig, P.; Fagerlund, C. and Dahlstrom, B.: Patient-controlled analgesic therapy, Part I: Pharmacokinetics of pethidine in the per- and post-operative periods. Clinical Pharmacokinetics 7: 149–163 (1982a).
Tamsen, A.; Hartvig, P.; Fagerlund, C. and Dahlstrom, B.: Patient-controlled analgesic therapy. Part II: Individual analgesic demand and analgesic plasma concentrations of pethidine in postoperative pain. Clinical Pharmacokinetics 7: 164–175 (1982b).
Verbeeck, R.K.; Branch, R.A. and Wilkinson, G.R.: Meperidine disposition in man: Influence of urinary pH and route of administration. Clinical Pharmacology and Therapeutics 30: 619–628 (1981).
Wilkinson, G. and Schenker, S.: Pharmacokinetics of meperidine in man. Clinical Pharmacology and Therapeutics 19: 486–488 (1976).
Yeh, S.Y. and Krebs, H.A.: TLC identification and GLC determination of meperidine and its metabolites in biological fluids. Journal of Pharmaceutical Sciences 70: 482–486 (1981).
Yeh, S.Y.; Chang, B.L.; Krebs, H.A. and Gorodetsky, C.W.: Potentiation of the analgesia of meperidine in the rat by pargyline. Journal of Pharmacology and Experimental Therapeutics 209: 125–131 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Edwards, D., Svensson, C.K., Visco, J.P. et al. Clinical Pharmacokinetics of Pethidine: 1982. Clin Pharmacokinet 7, 421–433 (1982). https://doi.org/10.2165/00003088-198207050-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-198207050-00003