Abstract
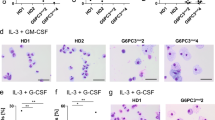
Severe congenital neutropenia (SCN) is a primary immunodeficiency disease in which a number of underlying gene defects are responsible for abnormalities in neutrophil development. The HCLS1-associated protein X1 (HAX1) mutation is associated with an autosomal-recessive form of SCN. Considering the potential of gene therapy approaches for the treatment of monogenic disorders, in this study we aimed to develop retroviral vectors expressing coding sequences (CDS) to be used for the removal of the genetic blockade in deficient hematopoietic cells. Following amplification of CDS with primers containing appropriate restriction sites, HAX1 CDS was cloned into an intermediate vector using TA-cloning. The sequence was transferred into a retroviral vector, followed by retroviral packaging in Plat-A cells. To show HAX1 protein expression, HEK293T cells were exposed to 10 multiplicity of infection (MOI) of retroviral particles and HAX1 expression was confirmed in these cells, using indirect intracellular flow cytometry. This vector was applied for in vitro transduction of hematopoietic stem cell with HAX1 mutation; after 11 days, cultured cells were analyzed for CD66acde and CD177 (neutrophil surface markers) expression. Increased neutrophil production in HAX1 viral vector-expressing hematopoietic cells was observed as compared to control vector transduced cells. Hence, according to the results, this type of therapy could be considered a potential treatment protocol for the disease.
Similar content being viewed by others
References
Klein C. Genetic defects in severe congenital neutropenia: emerging insights into life and death of human neutrophil granulocytes. Annu Rev Immunol 2011; 29(1): 399–413, https://doi.org/www.annualreviews.org/doi/10.1146/annurev-immunol-030409-101259.
Klein C, Grudzien M, Appaswamy G, et al. HAX1 deficiency causes autosomal recessive severe congenital neutropenia (Kostmann disease). Nat Genet 2007; 39(1): 86–92.
Krüger K, Mooren FC. Exercise-induced leukocyte apoptosis. Exerc Immunol Rev 2014; 20: 117–34.
Summers C, Rankin SM, Condliffe AM, Singh N, Peters AM, Chilvers ER. Neutrophil kinetics in health and disease. Trends Immunol 2010; 31(8): 318–24, https://doi.org/10.1016/j.it.2010.05.006.
Skokowa J, Dale DC, Touw IP, Zeidler C, Welte K. Severe congenital neutropenias. Nat Rev Dis Prim 2017; 3: 17032.
Fadeel B, Grzybowska E. HAX-1: a multifunctional protein with emerging roles in human disease. Biochim Biophys Acta 2009; 1790(10): 1139–48, https://doi.org/10.1016/j.bbagen.2009.06.004.
Lees DM, Hart IR, Marshall JF. Existence of multiple isoforms of HS1-associated protein X-1 in murine and human tissues. J Mol Biol 2008; 379(4): 645–55.
Suzuki Y, Demoliere C, Kitamura D, Takeshita H, Deuschle U, Watanabe T. HAX-1, a novel intracellular protein, localized on mitochondria, directly associates with HS1, a substrate of Src family tyrosine kinases. J Immunol 1997; 158(6): 2736–44, https://doi.org/www.jimmunol.org/cgi/reprint/158/6/2736%5Cnpapers2://publication/uuid/46027181-F6A0-4118-AF33-ED181D815FAA.
Klein C. Kostmann’s disease and HCLS1-associated protein X-1 (HAX1). J Clin Immunol 2017; 37(2): 117–22, https://doi.org/10.1007/s10875-016-0358-2.
Touw IP, Palande K, Beekman R. Granulocyte colony-stimulating factor receptor signaling. Implications for G-CSF responses and leukemic progression in severe congenital neutropenia. Hematol Oncol Clin North Am 2013; 27(1): 61–73, https://doi.org/10.1016/j.hoc.2012.10.002.
Fioredda F, Iacobelli S, Biezen A, et al. Stem cell transplantation in severe congenital neutropenia: an analysis from the European society for blood and marrow transplantation. Blood 2015; 126(16): 1885–93.
Touzot F, Hacein-Bey-Abina S, Fischer A, Cavazzana M. Gene therapy for inherited immunodeficiency. Expert Opin Biol Ther 2014; 14(6): 789–98, https://doi.org/www.tandfonline.com/doi/full/10.1517/14712598.2014.895811.
Aiuti A, Slavin S, Aker M, et al. Correction of ADA-SCID by stem cell gene therapy combined with nonmyeloablative conditioning. Science 2002; 296(5577): 2410–3, https://doi.org/www.sciencemag.org/cgi/doi/10.1126/science.1070104%5Cnhttp://www.ncbi.nlm.nih.gov/pubmed/12089448.
Cavazzana-Calvo M, Hacein-Bey S, De Saint Basile G, et al. Gene therapy of human severe combined immunodeficiency (SCID)-X1 disease. Science 2000; 288(5466): 669–72.
Candotti F. Gene transfer into hematopoietic stem cells as treatment for primary immunodeficiency diseases. Int J Hematol 2014; 99(4): 383–92.
Aiuti A, Bacchetta R, Seger R, Villa A, Cavazzana-Calvo M. Gene therapy for primary immunodeficiencies: Part 2. Curr Opin Immunol 2012; 24(5): 585–91, https://doi.org/10.1016/j.coi.2012.07.012.
Naldini L. Gene therapy returns to centre stage. Nature 2015; 526(7573): 351–60.
Morishima T, Watanabe K, Niwa A, et al. Genetic correction of HAX1 in induced pluripotent stem cells from a patient with severe congenital neutropenia improves defective granulopoiesis. Haematologica 2014; 99(1): 19–27.
Lanini LLS, Prader S, Siler U, Reichenbach J. Modern management of phagocyte defects. Pediatr Allergy Immunol 2017; 28(2): 124–34.
Fioredda F, Calvillo M, Burlando O, et al. Infectious complications in children with severe congenital, autoimmune or idiopathic neutropenia: a retrospective study from the Italian neutropenia registry. Pediatr Infect Dis J 2013; 32(4): 410–2.
Rosenberg PS, Zeidler C, Bolyard AA, et al. Stable long-term risk of leukaemia in patients with severe congenital neutropenia maintained on G-CSF therapy. Br J Haematol 2010; 150(2): 196–9, https://doi.org/10.1111/j.1365-2141.2010.08216.x.
Bao P, Zheng Y, Wang C, Gu K, Jin F, Lu W. Lethal sepsis and malignant transformation in severe congenital neutropenia: report from the Italian neutropenia registry. Pediatr Blood Cancer 2009; 53: 13–6.
Connelly JA, Choi SW, Levine JE. Hematopoietic stem cell transplantation for severe congenital neutropenia. Curr Opin Hematol 2012; 19(1): 44–51.
Welte K, Zeidler C, Dale DC. Severe congenital neutropenia. Semin Hematol 2006; 43(3): 189–95.
Elhasid R, Rowe JM. Hematopoetic stem cell transplantation in neutrophil disorders: severe congenital neutropenia, leukocyte adhesion deficiency and chronic granulomatous disease. Clin Rev Allergy Immunol 2010; 38(1): 61–7.
Watts KL, Adair J, Kiem H-P. Hematopoietic stem cell expansion and gene therapy. Cytotherapy 2011; 13(10): 1164–71, https://doi.org/www.ncbi.nlm.nih.gov/pubmed/21999373%5Cnhttp://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=PMC3291512.
Cavazzana-calvo M, Thrasher A, Mavilio F. The future of gene therapy. Balancing the risks and benefits of clinical trials. Nature 2004; 427: 779–81.
Aiuti A, Biasco L, Scaramuzza S, et al. Lentiviral hematopoietic stem cell gene therapy in patients with wiskott-aldrich syndrome. Science 2013; 341(6148): 1233151.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Farajifard, H., Zavvar, M., Rajaei, T. et al. In vitro study of HAX1 gene therapy by retro viral transduction as a therapeutic target in severe congenital neutropenia. Eur Cytokine Netw 29, 146–152 (2018). https://doi.org/10.1684/ecn.2018.0419
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/ecn.2018.0419