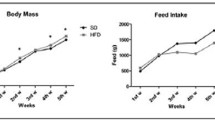
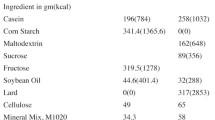
Abstract
This study compared the effects on bone metabolism and morphology of pathological obesity induced by excessive fat intake in a non-hibernator (mice) versus healthy obesity due to pre-hibernation fattening in a hibernator (ground squirrels). Kunming mice were fed a high-fat diet to provide a model of pathological obesity (OB group). Daurian ground squirrels fattened naturally in their pre-hibernation season (PRE group) were used as a healthy obesity model. Micro-computed tomography (micro-CT) and three-point bending tests were used to determine the microstructure and mechanical properties of bone. Western blots were used to analyze protein expression levels related to bone metabolism (Runt-related transcription factor 2 (RunX2), osteocalcin (OCN), alkaline phosphatase (ALP), osteoprotegerin (OPG), receptor activator of nuclear factor-κB ligand (RANKL), cathepsin K, matrix metallopeptidase 9 (MMP9), patched protein homolog 1 (Ptch1), phosphorylated β-catenin (P-β-catenin), and glycogen synthase kinase-3β (GSK-3β)). Compared with controls, there was no obvious bone loss in the OB mice, and the stiffness of the femur was increased significantly. Compared with summer active squirrels, bone formation was enhanced but the mechanical properties did not change in the PRE group squirrels. In OB mice, western blots showed significantly increased expression levels of all proteins except RunX2, OPG, and Ptch1. PRE ground squirrels showed significantly increased expression of most proteins except OCN and Ptch1, which decreased significantly, and P-β-catenin and OPG, which did not change. In conclusion, for non-hibernating mice, moderate obesity had a certain protective effect on bones, demonstrating two-way regulation, increasing both bone loss and bone formation. For pre-hibernating ground squirrels, the healthy obesity acquired before hibernation had a positive effect on the microstructure of bones, and also enhanced the expression levels of proteins related to bone formation, bone resorption, and Wnt signaling.
概要
本研究比较了非冬眠动物(昆明小鼠)过度摄入脂肪引起的病理性肥胖与冬眠动物(达乌尔黄鼠)冬眠前自然育肥导致的健康肥胖对骨代谢和形态的影响。研究将高脂饮食喂养的昆明小鼠作为病理性肥胖模型(OB组),冬眠前自然育肥的达乌尔黄鼠作为健康性肥胖模型(PRE组)。使用微计算机断层扫描和三点弯曲实验确定骨的微观结构和力学性能,并用蛋白质印迹分析与骨代谢相关的蛋白质(Runt相关转录因子2(Runt-related transcription factor 2,RunX2)、骨钙素(osteocalcin,OCN)、碱性磷酸酶(alkaline phosphatase,ALP)、骨保护素(osteoprotegerin,OPG)、核因子κB受体活化因子配体(receptor activator of nuclear factor-κB ligand,RANKL)、组织蛋白酶K(cathepsin K)、基质金属蛋白酶9(matrix metallopeptidase 9,MMP9)、蛋白修补同源1(patched protein homolog 1,Ptch1)、磷酸化β-连环蛋白(phosphorylated β-catenin,P-β-catenin)和糖原合酶激酶-3β(glycogen synthase kinase-3β,GSK-3β))的表达水平。结果显示,与对照(CON)组相比,OB组小鼠没有明显的骨质流失,但股骨的刚度明显增加。与夏季活跃(SA)组黄鼠相比,PRE组黄鼠的骨形成增强,但骨骼力学性能没有变化。在OB组小鼠中,蛋白质印迹显示除RunX2、OPG和Ptch1之外的所有蛋白质的表达水平显著增加。PRE组黄鼠大多数蛋白质的表达水平显著增加,但OCN和Ptch1显著下降,P-β-catenin和OPG没有变化。综上所述,对于非冬眠小鼠,适度肥胖对骨骼有一定的保护作用,表现为双向调节,既增加了骨质流失,又增加了骨形成。对于冬眠前黄鼠,冬眠前育肥对骨骼的微观结构有积极的影响,同时也提高了与骨形成、骨吸收和Wnt信号相关蛋白质的表达水平。
Similar content being viewed by others
References
Baron R, Kneissel M, 2013. Wnt signaling in bone homeostasis and disease: from human mutations to treatments. Nat Med, 19(2):179–192. https://doi.org/10.1038/nm.3074
Bettis T, Kim BJ, Hamrick MW, 2018. Impact of muscle atrophy on bone metabolism and bone strength: implications for muscle-bone crosstalk with aging and disuse. Osteoporos Int, 29(8):1713–1720. https://doi.org/10.1007/s00198-018-4570-1
Bhattarai T, Bhattacharya K, Chaudhuri P, et al., 2014. Correlation of common biochemical markers for bone turnover, serum calcium, and alkaline phosphatase in postmenopausal women. Malays J Med Sci, 21(1):58–61.
Buchet R, Millán JL, Magne D, 2013. Multisystemic functions of alkaline phosphatases. In: Millán J (Ed.), Phosphatase Modulators. Methods in Molecular Biology, Vol. 1053. PressHumana, Totowa, NJ, p.27–51. https://doi.org/10.1007/978-1-62703-562-0_3
Busutil R, Espallardo O, Torres A, et al., 2017. The impact of obesity on health-related quality of life in Spain. Health Qual Life Outcomes, 15:197. https://doi.org/10.1186/s12955-017-0773-y
Cao JJ, Picklo MJ, 2014. N-Acetylcysteine supplementation decreases osteoclast differentiation and increases bone mass in mice fed a high-fat diet. J Nutr, 144(3):289–296. https://doi.org/10.3945/jn.113.185397
Cao JJ, Gregoire BR, Michelsen KG, et al., 2020. Increasing dietary fish oil reduces adiposity and mitigates bone deterioration in growing C57BL/6 mice fed a high-fat diet. J Nutr, 150(1):99–107. https://doi.org/10.1093/jn/nxz215
Chen FL, Wang Y, Wang HW, et al., 2019. Flaxseed oil ameliorated high-fat-diet-induced bone loss in rats by promoting osteoblastic function in rat primary osteoblasts. Nutr Metab (Lond), 16:71. https://doi.org/10.1186/s12986-019-0393-0
Chen HL, Li JF, Wang Q, 2018. Associations between bone-alkaline phosphatase and bone mineral density in adults with and without diabetes. Medicine (Baltimore), 97(17):e0432. https://doi.org/10.1097/md.jzus010432
Chen JR, Lazarenko OP, Wu XL, et al., 2010. Obesity reduces bone density associated with activation of PPARγ and suppression of Wnt/β-catenin in rapidly growing male rats. PLoS ONE, 5(10):e13704. https://doi.org/10.1371/journal.pone.0013704
Chung AWY, Hsiang YN, Matzke LA, et al., 2006. Reduced expression of vascular endothelial growth factor paralleled with the increased angiostatin expression resulting from the upregulated activities of matrix metalloproteinase-2 and -9 in human type 2 diabetic arterial vasculature. Circ Res, 99(2):140–148. https://doi.org/10.1161/01.RES.0000232352.90786.fa
Clark EM, Ness AR, Tobias JH, 2006. Adipose tissue stimulates bone growth in prepubertal children. J Clin Endocrinol Metab, 91(7):2534–2541. https://doi.org/10.1210/jc.2006-0332
Dallas SL, Prideaux M, Bonewald LF, 2013. The osteocyte: an endocrine cell … and more. Endocr Rev, 34(5):658–690. https://doi.org/10.1210/er.2012-1026
Datta HK, Ng WF, Walker JA, et al., 2008. The cell biology of bone metabolism. J Clin Pathol, 61(5):577–587. https://doi.org/10.1136/jcp.2007.048868
Day TF, Yang YZ, 2008. Wnt and hedgehog signaling pathways in bone development. J Bone Joint Surg Am, 90(Suppl 1):19–24. https://doi.org/10.2106/jbjs.g.01174
Ducy P, Desbois C, Boyce B, et al., 1996. Increased bone formation in osteocalcin-deficient mice. Nature, 382(6590):448–452. https://doi.org/10.1038/382448a0
Fang ZY, Lin R, Yuan BX, et al., 2007. Tanshinone IIA inhibits atherosclerotic plaque formation by down-regulating MMP-2 and MMP-9 expression in rabbits fed a high-fat diet. Life Sci, 81(17–18):1339–1345. https://doi.org/10.1016/j.lfs.2007.08.043
Feng YM, Wan PB, Yin LL, et al., 2020. The inhibition of microRNA-139-5p promoted osteoporosis of bone marrow-derived mesenchymal stem cells by targeting Wnt/beta-catenin signaling pathway by NOTCH1. J Microbiol Biotechnol, 30(3):448–458. https://doi.org/10.4014/jmb.1908.08036
Gao XL, Wang SQ, Zhang J, et al., 2021. Differential bone remodeling mechanism in hindlimb unloaded rats and hibernating Daurian ground squirrels: a comparison between artificial and natural disuse. J Comp Physiol B, 191(4):793–814. https://doi.org/10.1007/s00360-021-01375-9
Gautam J, Choudhary D, Khedgikar V, et al., 2014. Microarchitectural changes in cancellous bone differ in female and male C57BL/6 mice with high-fat diet-induced low bone mineral density. Br J Nutr, 111(10):1811–1821. https://doi.org/10.1017/s0007114514000051
Glass DA II, Bialek P, Ahn JD, et al., 2005. Canonical Wnt signaling in differentiated osteoblasts controls osteoclast differentiation. Dev Cell, 8(5):751–764. https://doi.org/10.1016/j.devcel.2005.02.017
Gong YQ, Slee RB, Fukai N, et al., 2001. LDL receptor-related protein 5 (LRP5) affects bone accrual and eye development. Cell, 107(4):513–523. https://doi.org/10.1016/s0092-8674(01)00571-2
Gu Y, Wang ZR, Shi JW, et al., 2017. Titanium particle-induced osteogenic inhibition and bone destruction are mediated by the GSK-3β/β-catenin signal pathway. Cell Death Dis, 8(6):e2878. https://doi.org/10.1038/cddis.2017.275
Halade GV, Rahman M, Williams PJ, et al., 2010. High fat diet-induced animal model of age-associated obesity and osteoporosis. J Nutr Biochem, 21(12):1162–1169. https://doi.org/10.1016/j.jnutbio.2009.10.002
Halade GV, el Jamali A, Williams PJ, et al., 2011. Obesity-mediated inflammatory microenvironment stimulates osteoclastogenesis and bone loss in mice. Exp Gerontol, 46(1):43–52. https://doi.org/10.1016/j.exger.2010.09.014
Halade GV, Jin YF, Lindsey ML, 2013. Matrix metalloproteinase (MMP)-9: a proximal biomarker for cardiac remodeling and a distal biomarker for inflammation. Pharmacol Ther, 139(1):32–40. https://doi.org/10.1016/j.pharmthera.2013.03.009
Hill TP, Später D, Taketo MM, et al., 2005. Canonical Wnt/β-catenin signaling prevents osteoblasts from differentiating into chondrocytes. Dev Cell, 8(5):727–738. https://doi.org/10.1016/j.devcel.2005.02.013
Hoeppner LH, Secreto FJ, Westendorf JJ, 2009. Wnt signaling as a therapeutic target for bone diseases. Expert Opin Ther Targets, 13(4):485–496. https://doi.org/10.1517/14728220902841961
Hwang JH, Cha PH, Han G, et al., 2015. Euodia sutchuenensis dode extract stimulates osteoblast differentiation via Wnt/β-catenin pathway activation. Exp Mol Med, 47(3):e152. https://doi.org/10.1038/emm.2014.115
Ionova-Martin SS, Do SH, Barth HD, et al., 2010. Reduced size-independent mechanical properties of cortical bone in high-fat diet-induced obesity. Bone, 46(1):217–225. https://doi.org/10.1016/j.bone.2009.10.015
Ionova-Martin SS, Wade JM, Tang S, et al., 2011. Changes in cortical bone response to high-fat diet from adolescence to adulthood in mice. Osteoporos Int, 22(8):2283–2293. https://doi.org/10.1007/s00198-010-1432-x
Kamine A, Shimozuru M, Shibata H, et al., 2012. Changes in blood glucose and insulin responses to intravenous glucose tolerance tests and blood biochemical values in adult female Japanese black bears (Ursus thibetanus japonicus). Jpn J Vet Res, 60(1):5–13.
Kato M, Patel MS, Levasseur R, et al., 2002. Cbfa1-independent decrease in osteoblast proliferation, osteopenia, and persistent embryonic eye vascularization in mice deficient in Lrp5, a Wnt coreceptor. J Cell Biol, 157(2):303–314. https://doi.org/10.1083/jcb.200201089
Kim JM, Yang YS, Park KH, et al., 2020. A RUNX2 stabilization pathway mediates physiologic and pathologic bone formation. at Commun, 11:2289. https://doi.org/10.1038/s41467-020-16038-6
Kim MS, Yang YM, Son A, et al., 2010. RANKL-mediated reactive oxygen species pathway that induces long lasting Ca2+ oscillations essential for osteoclastogenesis. J Biol Chem, 285(10):6913–6921. https://doi.org/10.1074/jbc.M109.051557
Koch FP, Merkel C, Al-Nawas B, et al., 2011. Zoledronate, ibandronate and clodronate enhance osteoblast differentiation in a dose dependent manner—a quantitative in vitro gene expression analysis of Dlx5, Runx2, OCN, MSX1 and MSX2. J Cranio-Maxillofac Surg, 39(8):562–569. https://doi.org/10.1016/j.jcms.2010.10.007
Komori T, 2005. Regulation of skeletal development by the Runx family of transcription factors. J Cell Biochem, 95(3):445–453. https://doi.org/10.1002/jcb.20420
Komori T, 2011. Signaling networks in RUNX2-dependent bone development. J Cell Biochem, 112(3):750–755. https://doi.org/10.1002/jcb.22994
Koshihara Y, Hoshi K, 1997. Vitamin K2 enhances osteocalcin accumulation in the extracellular matrix of human osteoblasts in vitro. J Bone Miner Res, 12(3):431–438. https://doi.org/10.1359/jbmr.1997.12.3.431
Kurgan N, Bott KN, Helmeczi WE, et al., 2019. Low dose lithium supplementation activates Wnt/β-catenin signalling and increases bone OPG/RANKL ratio in mice. Biochem Biophys Res Commun, 511(2):394–397. https://doi.org/10.1016/j.bbrc.2019.02.066
Lam AP, Gottardi CJ, 2011. β-Catenin signaling: a novel mediator of fibrosis and potential therapeutic target. Curr Opin Rheumatol, 23(6):562–567. https://doi.org/10.1097/BOR.0b013e32834b3309
Lampropoulos CE, Papaioannou I, D’Cruz DP, 2012. Osteoporosis—a risk factor for cardiovascular disease? Nat Rev Rheumatol, 8(10):587–598. https://doi.org/10.1038/nrrheum.2012.120
Lanaspa MA, Kuwabara M, Andres-Hernando A, et al., 2018. High salt intake causes leptin resistance and obesity in mice by stimulating endogenous fructose production and metabolism. Proc Natl Acad Sci USA, 115(12):3138–3143. https://doi.org/10.1073/pnas.1713837115
Lee NK, Sowa H, Hinoi E, et al., 2007. Endocrine regulation of energy metabolism by the skeleton. Cell, 130(3):456–469. https://doi.org/10.1016/j.cell.2007.05.047
Leonard MB, Shults J, Wilson BA, et al., 2004. Obesity during childhood and adolescence augments bone mass and bone dimensions. Am J Clin Nutr, 80(2):514–523. https://doi.org/10.1093/ajcn/80.2.514
Liu CT, Broe KE, Zhou YH, et al., 2017. Visceral adipose tissue is associated with bone microarchitecture in the framingham osteoporosis study. J Bone Miner Res, 32(1):143–150. https://doi.org/10.1002/jbmr.2931
Liu XL, Liang YZ, Xia N, et al., 2021. Decrease in leptin mediates rat bone metabolism impairments during high-fat diet-induced catch-up growth by modulating the OPG/RANKL balance. 3 Biotech, 11:103. https://doi.org/10.1007/s13205-021-02658-2
Martin SA, Souder DC, Miller KN, et al., 2018. GSK3β regulates brain energy metabolism. Cell Rep, 23(7):1922–1931.e4. https://doi.org/10.1016/j.celrep.2018.04.045
Minematsu A, Nishii Y, Sakata S, 2018. High-fat/high-sucrose diet results in higher bone mass in aged rats. Bone Rep, 8:18–24. https://doi.org/10.1016/j.bonr.2018.01.001
Moorer MC, Riddle RC, 2018. Regulation of osteoblast metabolism by Wnt signaling. Endocrinol Metab, 33(3):318–330. https://doi.org/10.3803/EnM.2018.33.3.318
Novinec M, Lenarčič B, 2013. Cathepsin K: a unique collagenolytic cysteine peptidase. Biol Chem, 394(9):1163–1179. https://doi.org/10.1515/hsz-2013-0134
Núñez NP, Carpenter CL, Perkins SN, et al., 2007. Extreme obesity reduces bone mineral density: complementary evidence from mice and women. Obesity, 15(8):1980–1987. https://doi.org/10.1038/oby.2007.236
Penrose HM, Heller S, Cable C, et al., 2017. High-fat diet induced leptin and Wnt expression: RNA-sequencing and pathway analysis of mouse colonic tissue and tumors. Carcinogenesis, 38(3):302–311. https://doi.org/10.1093/carcin/bgx001
Petit MA, Beck TJ, Shults J, et al., 2005. Proximal femur bone geometry is appropriately adapted to lean mass in overweight children and adolescents. Bone, 36(3):568–576. https://doi.org/10.1016/j.bone.2004.12.003
Picke AK, Sylow L, Møller LLV, et al., 2018. Differential effects of high-fat diet and exercise training on bone and energy metabolism. Bone, 116:120–134. https://doi.org/10.1016/j.bone.2018.07.015
Pineda N, Owen M, Tucker C, et al., 2017. Hibernating little pocket mice show few seasonal changes in bone properties. Anat Rec, 300(12):2175–2183. https://doi.org/10.1002/ar.23676
Pitynski-Miller D, Ross M, Schmill M, et al., 2017. A high salt diet inhibits obesity and delays puberty in the female rat. Int J Obes, 41(11):1685–1692. https://doi.org/10.1038/ijo.2017.154
Pollock NK, Laing EM, Hamrick MW, et al., 2011. Bone and fat relationships in postadolescent black females: a pQCT study. Osteoporos Int, 22(2):655–665. https://doi.org/10.1007/s00198-010-1266-6
Proietto J, 2020. Obesity and bone. F1000Research, 9(Faculty Rev):1111. https://doi.org/10.12688/f1000research.20875.1
Reinhold MI, Naski MC, 2007. Direct interactions of Runx2 and canonical Wnt signaling induce FGF18. J Biol Chem, 282(6):3653–3663. https://doi.org/10.1074/jbc.M608995200
Rigano KS, Gehring JL, Hutzenbiler BDE, et al., 2017. Life in the fat lane: seasonal regulation of insulin sensitivity, food intake, and adipose biology in brown bears. J Comp Physiol B, 187(4):649–676. https://doi.org/10.1007/s00360-016-1050-9
Savvidis C, Tournis S, Dede AD, 2018. Obesity and bone metabolism. Hormones, 17(2):205–217. https://doi.org/10.1007/s42000-018-0018-4
Shapses SA, Sukumar D, 2012. Bone metabolism in obesity and weight loss. Annu Rev Nutr, 32:287–309. https://doi.org/10.1146/annurev.nutr.012809.104655
Takagi Y, Sugimoto T, Kobayashi M, et al., 2018. High-salt intake ameliorates hyperglycemia and insulin resistance in WBN/Kob-Leprfa/fa rats: a new model of type 2 diabetes mellitus. J Diabetes Res, 2018:3671892. https://doi.org/10.1155/2018/3671892
Takahashi N, Maeda K, Ishihara A, et al., 2011. Regulatory mechanism of osteoclastogenesis by RANKL and Wnt signals. Front Biosci (Landmark Ed), 16(1):21–30. https://doi.org/10.2741/3673
Tauer JT, Boraschi-Diaz I, al Rifai O, et al., 2021. Male but not female mice with severe osteogenesis imperfecta are partially protected from high-fat diet-induced obesity. Mol Genet Metab, 133(2):211–221. https://doi.org/10.1016/j.ymgme.2021.03.014
Vijay GV, Zhao N, den Hollander P, et al., 2019. GSK3β regulates epithelial-mesenchymal transition and cancer stem cell properties in triple-negative breast cancer. Breast Cancer Res, 21:37. https://doi.org/10.1186/s13058-019-1125-0
Vimalraj S, 2020. Alkaline phosphatase: structure, expression and its function in bone mineralization. Gene, 754:144855. https://doi.org/10.1016/j.gene.2020.144855
Wang HW, Chen FL, Li JX, et al., 2020. Vaspin antagonizes high fat-induced bone loss in rats and promotes osteoblastic differentiation in primary rat osteoblasts through Smad-Runx2 signaling pathway. Nutr Metab (Lond), 17:9. https://doi.org/10.1186/s12986-020-0429-5
Wosje KS, Khoury PR, Claytor RP, et al., 2009. Adiposity and TV viewing are related to less bone accrual in young children. J Pediatr, 154(1):79–85.e72. https://doi.org/10.1016/j.jpeds.2008.06.031
Yan L, Graef GL, Nielsen FH, et al., 2015. Soy protein is beneficial but high-fat diet and voluntary running are detrimental to bone structure in mice. Nutr Res, 35(6):523–531. https://doi.org/10.1016/j.nutres.2015.04.012
Yan X, Niu QH, Gao XL, et al., 2021. Differential protein metabolism and regeneration in gastrocnemius muscles in high-fat diet fed mice and pre-hibernation daurian ground squirrels: a comparison between pathological and healthy obesity. Zool Stud, 60:e6. https://doi.org/10.6620/zs.2021.60-06
Yanagihara GR, Shimano RC, Tida JA, et al., 2017. Influence of high-fat diet on bone tissue: an experimental study in growing rats. J Nutr Health Aging, 21(10):1337–1343. https://doi.org/10.1007/s12603-017-0871-x
Yang Y, Mallampati S, Sun BH, et al., 2013. Wnt pathway contributes to the protection by bone marrow stromal cells of acute lymphoblastic leukemia cells and is a potential therapeutic target. Cancer Lett, 333(1):9–17. https://doi.org/10.1016/j.canlet.2012.11.056
Yun TJ, Tallquist MD, Aicher A, et al., 2001. Osteoprotegerin, a crucial regulator of bone metabolism, also regulates B cell development and function. J Immunol, 166(3):1482–1491. https://doi.org/10.4049/jimmunol.166.3.1482
Zhong L, Yuan JH, Huang L, et al., 2020. RANKL is involved in Runx2-triggered hepatic infiltration of macrophages in mice with NAFLD induced by a high-fat diet. Biomed Res Int, 2020:6953421. https://doi.org/10.1155/2020/6953421
Zhou S, Wang GH, Qiao L, et al., 2018. Age-dependent variations of cancellous bone in response to ovariectomy in C57BL/6J mice. Exp Ther Med, 15(4):3623–3632. https://doi.org/10.3892/etm.2018.5839
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Nos. 31640072 and 31900338) and the Natural Science Basic Research Program of Shaanxi (No. 2020JM-428), China.
Author information
Authors and Affiliations
Contributions
Hui CHANG conceived and designed the experiments. Xuli GAO, Shenyang SHEN, Qiaohua NIU, Weilan MIAO, Yuting HAN, Ziwei HAO, Ning AN, Yingyu YANG, Yu ZHANG, and Han ZHANG performed the experiments. Xuli GAO, Shenyang SHEN, and Qiaohua NIU analyzed the data. Shenyang SHEN, Qiaohua NIU, and Kenneth B STOREY wrote the paper. All authors have read and approved the final manuscript, and therefore, have full access to all data relevant to the study and take responsibility for the integrity and security of such data.
Corresponding author
Additional information
Compliance with ethics guidelines
Xuli GAO, Shenyang SHEN, Qiaohua NIU, Weilan MIAO, Yuting HAN, Ziwei HAO, Ning AN, Yingyu YANG, Yu ZHANG, Han ZHANG, Kenneth B STOREY, and Hui CHANG declare that they have no conflict of interest.
All animal experiments were approved by the Experimental Animal Protection Committee of the Ministry of Health of the People’s Republic of China (Approval Number: SL-2012-42).
Materials and methods
Detailed methods are provided in the electronic supplementary materials of this paper.
Supplementary information
Materials and methods
Supplementary information
11585_2022_658_MOESM1_ESM.pdf
Differential bone metabolism and protein expression in mice fed a high-fat diet versus Daurian ground squirrels following natural pre-hibernation fattening
Rights and permissions
About this article
Cite this article
Gao, X., Shen, S., Niu, Q. et al. Differential bone metabolism and protein expression in mice fed a high-fat diet versus Daurian ground squirrels following natural pre-hibernation fattening. J. Zhejiang Univ. Sci. B 23, 1042–1056 (2022). https://doi.org/10.1631/jzus.B2100798
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2100798