Abstract
Coffee is among the most drunk beverages in the world and its consumption produces massive amounts of waste. Valorization strategies of coffee wastes include production of carbon materials for electrochemical energy storage devices such as batteries, supercapacitors, and fuel cells.
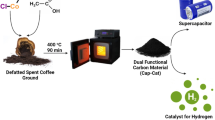
Coffee is one of the most consumed beverages in the world. In the linear model adopted so far, its consumption is associated with huge amounts of waste and spent coffee grounds. These wastes, instead, are very interesting secondary raw materials for several circular economy concepts. Nano-structured porous carbon materials obtained by coffee waste are emerging as active materials for electrochemical energy storage devices like supercapacitors and batteries. The major results achieved in the last decade in this high-value exploitation strategy of coffee wastes are summarized to suggest a new sustainable use of coffee waste in the empowerment of the ongoing transition toward a green, electrified, and happier coffee-drinking society.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Discussion
-
Coffee waste-derived carbon materials for energy storage has been proved, but further investigations to optimize and identify key metrics of the process are required to evaluate the consistency of materials and devices performances at a commercially relevant scale.
-
The logistics associated with the coffee wastes supply chain need to be carefully evaluated to provide economic and environmental sustainability along the entire life cycle of coffee wastes used as raw materials.
Coffee consumption worldwide
Coffee follows water and tea in the list of the most consumed beverages in the world. Brazil firmly holds the first place among exporting countries, while the European Union is the first consumer with roughly 6 kg per capita per year.1,2 According to the International Coffee Organization (ICO), the consumption in 2020–2021 reached 166.34 million bags—one bag corresponding to 60 kg of coffee—(Fig. 1) divided between the Arabica (close to 100 million bags) and the Robusta (70 million bags) varieties. The selection of coffee varieties, the specific coffee blends, and the drivers that guide consumers have been the object of a large number of studies recently reviewed.3 Ethical and environmental concerns are increasing their impact on the consumption and purchasing behaviors, and this is particularly relevant for coffee, which is considered as a pioneering industry for sustainability certification schemes.3 Yet, sustainability along the coffee value chain is still a challenge.4
Given the pervasive presence in our daily lives and its economic, social, and environmental impacts, coffee has attracted the interest of the scientific community in a wide number of fields, spanning from humanities and social science to natural and life science, well beyond the study of production and consumption behaviors. As a direct consequence of the global coffee consumption, the waste and byproducts generated along the entire value chain (from the agricultural production, through industrial transformation, until the brewing and the final user consumption) can enter in a circular development model for a resilient and sustainable society; hence, employing the abundant coffee waste stream represents a challenge for engineers, chemists, and material scientists. Herein, we reviewed the emerging application of coffee waste, especially spent coffee grounds, as raw material for electrochemical energy conversion and storage devices.
Byproducts from coffee value chain and recycling options
It is important to examine the whole value chain of coffee for a complete overview of the mitigation strategies of the impact associated with coffee production and consumption. A study on environmental impacts of food consumptions in Europe considered coffee among the basket of products selected to examine possible impacts and future consumption scenarios. Coffee has the highest emissions to air and water among the agriculture products, estimated at 4 kg CO2 eq. per year per person in the EU5 as consequence of intensive use of fertilizers and a strong impact on logistics due to the geographical position of exporting countries. Considering water footprint coffee scores 140 L of virtual water content per standard cup, almost five times the one of tea. With less than 0.5% for the processing, almost the entire water consumption is associated to plant cultivation and is in charge to the producing countries rather than to the consuming one.6,7 Transportation is responsible for the majority of the coffee Carbon footprint; for instance, the exportation from Brazil or Vietnam to the UK accounts for more than 70% of kg CO2 equivalent per kg of Arabica coffee.8 In addition to the environmental impact and resource consumptions, different kinds of byproducts and waste are generated along the entire value chain of coffee, from production, through transformation, to the final user consumption.
The coffee value chain is schematically presented in Fig. 2. Coffee direct byproducts and waste are mainly generated during processing and consumption.
Pulp, husk, and skin of the coffee cherries (or berries) represent roughly the 45% in weight of the fruit9 and are removed under either wet or dry conditions to obtain the green coffee beans. The following roasting process produces the coffee silver skin, a minor byproduct, accounting for 1–2% in weight of the dry coffee cherry, rich in antioxidant polyphenol compounds and in cellulose and hemicellulose (almost 60%). As the coffee bean accounts for almost 50% of the cherry, the main byproduct of the whole coffee value chain is spent coffee grounds (SCG) generated by the industrial production of soluble coffee or by the final consumption of roasted coffee. For every ton of green coffee (or dried berry), the byproducts of the process account for 500 kg of coffee pulp, 180 kg of coffee husk, and 650 kg of spent coffee grounds.9,10 along with other minor byproducts such as coffee silver skin, coffee parchment, and mucilage.11
Besides secondary waste (e.g., packaging), the major direct byproduct of the entire coffee value chain is generated at the final consumer level, and this aspect must be considered for circular design strategies and waste management in the coffee industry. The byproducts illustrated In Fig. 2 have gained attention as sources of high-value chemicals and materials as reviewed elsewhere.9,10,11
Spent coffee grounds have attracted the interest as secondary raw materials for several different products (Fig. 3). The most diffuse current applications of SCGs are relatively low-value applications, as substrate for mushroom growth or for composting and vermi-composting.10 Another investigated application is energy recovery through biofuel production. This approach includes the use of SCGs as low-cost solid fuels, for bioethanol and biogas production through fermentation and for biodiesel by oil extraction and chemical processing.12 An additional wide class of studies has been focusing on further value recovery from SCGs especially through bulk and specialty chemicals and substances, addressing the concept of biorefinery beyond the sole energy recovery target. Besides valuable caffeine extraction, carbohydrates, non-protein nitrogenous compounds, lipids, phenolic compounds, and melanoidins have been highlighted as possible products attainable from SCGs.13 Alternatively to its direct use, the fractionation of SCGs, with the extraction of different classes of compounds, is reported to allow multiple valorization strategies. Next to the already mentioned use of antioxidant polyphenol fraction, the oil fraction has been adopted not only for high-purity biodiesel, but also proposed for cosmetic industry (UV protection, antioxidant, skin hydration) and for bioplastic production, with examples of high productivities obtained for polyhydroxyalkanoates (PHAs), a class of high-performance polyesters produced by bacterial fermentation.13,14 The carbohydrate fraction has been instead used to isolate hemicellulose and galactomannans with useful application in cosmetics and healthcare.13 The hydrolysate produced with the hemicellulose isolation has been applied also in PHA production, as well as for yeast fermentation substrate, e.g., to produce high-value carotenoids.14
Another interesting conceptualization of spent coffee ground biorefinery was based on different approaches to make products, separating primary products which are obtained by the already discussed fractionation, and secondary products obtained by their transformation through biotechnological, chemical, and thermochemical routes. These secondary products include bioprocessing products (lactic acid, PHAs, biogas, triglycerides from yeast, bioethanol), biodiesel, fertilizers, animal feeds, formulations of polysaccharides and antioxidants (mainly for cosmetics), biosorbents, and biodegradable films for food packaging and construction materials.15 A critical point for the economic viability of a spent coffee biorefinery is definitely the logistics for the supply and recovery of SCGs. Moreover, many of the processes must take into account the presence of contaminants and the overall lifecycle of the processing to ensure a low impact of the biorefinery product. Coupling high added value chemical isolations with thermochemical valorization for energy and materials recovery seems to emerge as a reasonable approach to be further explored and investigated.10,11,12,13,14,15,16 Thermochemical valorization leads to mainly bio-oils and bio-chars, and minor quantities of biogas through hydrothermal or pyrolytic processes. Clearly the process conditions are varied to maximize the desired output of oil, char, and gas for the intended circular economy approach.15,16 As better detailed in the next section, the solid carbon fraction (char) obtained by thermal treatment of spent coffee grounds can be further chemically or physically activated at high temperatures to produce carbon materials with high surface area and controlled porosity (activated carbon) with key properties as active materials for electrochemical energy storage and conversion devices. This is one of the most intriguing emerging applications to re-use coffee waste and specifically spent coffee grounds by valorizing their intrinsic chemical and physical properties.
Electrochemical energy storage introduction
“In Europe, within this decade, where it is technologically and economically viable, everything that can be electrified will be electrified.”17 This statement opens the European “Strategic Research Agenda for batteries” recognizing that the present industrial transition is massively shifting from a conversion by combustion of carbon-based fuels toward the use of electrical energy, or in other words a flow of electrons. The electrification switch does not per se guarantee a short-term decarbonization of energy production. Regardless, besides energy production and usage, a key role in this inflection point we are facing toward a truly electrified society will be played by the so-called “energy vectors,” or in other terms, the systems that will be adopted to store and, in some cases, transport the energy from the production to the usage point.
The most widespread technology for energy storage is stationary and portable electrochemical devices such as batteries and supercapacitors. Less common but under further developments are the technologies for hydrogen production such as electrolyzers, hydrogen-storage systems, and fuel cells to transport and finally convert back hydrogen into water and electricity.
Despite the specific nature of the technologies adopted or of the combination thereof, they all rely on electrodes and electrochemical interfaces to convert electric energy into chemical one and vice versa.
Lithium-ion batteries (LIBs) are gaining a dominant role in the market of portable energy storage systems due to the exponential growth of electric vehicles and battery-powered personal electronic devices; thus, a raising issue associated with LIBs is their overall sustainability.19,20 The main active materials and components of a conventional lithium-ion battery are assessed in Europe as “critical raw materials,” i.e., materials of economic importance having risk of supply, raising global environmental and geo-political issues.21 Considering the growing request of energy portable systems in the next years, LIBs global consumption is expected to increase by 18% per year22,23 and materials recycling is an utmost relevant topic in the short to medium period. Even more important is the advent of a “post-lithium” era that will address the sustainability of diffuse energy portable systems by substituting lithium with more abundant metal ions such as sodium, potassium, magnesium, zinc, and aluminum and the relevant chemistry.24,25 A comprehensive overview on present and developing materials and technologies for greener batteries has been recently published by Dühnen et al.,18 with a focus on different critical issues for state-of-the-art LIBs as shown in Fig. 4.
State-of-the-art lithium battery technology and major critical aspects. Reproduced from Ref. 18.
Commonly, natural graphite is used as anode material in state-of-the-art LIBs; this carbon form has already been listed as critical raw material.21 Recently, alternative to graphite as electroactive carbon materials obtained from waste biomass and agricultural byproducts have been proposed.18,26,27 Coffee waste is included as a possible carbon precursor. The intimate intercalation of lithium ions in the ordered structure of graphite is currently yet unreached by any other carbon substitute, including coffee waste-derived one. Nonetheless, bio-based and coffee-derived carbon (CDC) materials for electrochemical energy storage, still in their infancy, can find a closer commercial application in the class of supercapacitors.
Supercapacitors as high-power green alternatives
Supercapacitors or ultracapacitors are a class of energy storage devices complementary to batteries. Batteries rely on faradic, redox, reactions to store electrons while supercapacitors exploit the electrostatic charge accumulation at the electrodes balanced by a counter-ion absorption. This energy storage mechanism results in high power densities, i.e., high rate of charge and discharge of the electrode, with low energy densities, i.e., a low quantity of energy accumulated in the device.27,28,29,30,31,32,33 Supercapacitors use highly porous carbon-based electrodes possessing high surface area in place of the metal electrodes used in conventional capacitors. Furthermore, supercapacitors replace the solid dielectric of conventional capacitors with the electrical double layer (supercapacitors are also known as electrical double layer capacitors or EDLC). Supercapacitors are very effective in complementing batteries in stationary applications for peak current storage or in electric vehicles. They are becoming a key technology for miniaturized devices in applications as wearable electronics or Internet of Things (IoT) devices, sensors, and actuators.30,31
Supercapacitors are classified as symmetric or asymmetric based on the device configuration and as purely capacitive, pseudocapacitive, and hybrid, based on the charge storage mechanism (see Fig. 5). Supercapacitors are intrinsically greener devices than batteries as they use less critical materials and have a manufacturing process with lower environmental impact; the majority of commercially supercapacitors employs low-cost and largely available bio-based carbon materials, such as activated carbon derived from coconut shells.32,33,34 Other bio-based nanostructured carbon materials have been investigated in the last decades for using waste biomass or agro-food byproducts to increase the sustainability and circularity of supercapacitors.34,35
Energy storage faradaic mechanism in batteries (a) and differences between faradaic, capacitive, and hybrid electrodes (b) Symmetric capacitors employ the same electrode material both at the anode and at the cathode.28,32 Typical materials which provide a pseudocapacitive behavior are metal oxides such as MnO2 and RuO2 that combine the capacitive charge storage mechanism with the redox reactions of metal ions.29 Alternatively a capacitive electrode can be coupled with redox-active species in the electrolyte, with the so-called a “redox mediator electrode” configuration.29,32 A hybrid device can present one electrode typically made of purely capacitive porous carbon and the other made of a metal-ion-intercalating material such as graphite.28,29,30,32
Electroactive carbon materials are produced by controlled hydrothermal or pyrolytic carbonization followed by chemical or physical activation, leading to amorphous carbon with a highly porous structure. Specific treatments have been proposed for bio-based controlled nanostructures as graphene or carbon nanofibers.36,37 Researchers have been focusing also on the manufacturing process of commercial devices that require polymeric binders to process the powder carbon materials onto the metallic current collectors. Water-soluble natural binders have proven to be a promising alternative.38,39 The binder and the processing of the carbon material are also crucial to achieve sufficient mass loading while maintaining specific performances, in order to get viable devices for practical commercial applications.40,41,42
Within this broad context, one of the most intriguing bio-based precursor for a circular design of green supercapacitor materials is coffee byproducts and waste, and particularly spent coffee grounds.
Coffee-based materials for electrochemical energy conversion and storage devices
Coffee has attracted interest as precursor of electroactive materials in supercapacitors, batteries, and fuel cells given its large availability. Coffee waste chemical composition also provides a beneficial natural heteroatom doping for carbon material as better described later in the text. Finally, spent coffee grounds are usually recovered after hot water extraction, the brewing process, and are, thus, already washed and rich in humidity, useful for conventional thermal treatment to obtain porous carbon materials. As shown in the following paragraphs, CDC has often outperformed other materials in electrochemical energy conversion and storage applications (Fig. 6).
Main routes to produce activated carbons from coffee waste as electroactive materials for electrochemical energy conversion and storage devices with real SEM images of carbonized coffee (a) and activated carbon (b) adapted from Ref. 49.
The common procedure to produce porous activated carbon from biomass is carbonization followed by activation at high temperatures, often in the presence of activating or porogenic chemical agents. A number of different carbonization and activation protocols have been developed aiming to a controlled hierarchical porous structure, a large surface area, a good electrical conductivity, and to a maximized carbon yields.34,35
Supercapacitors
Due to their higher intrinsic sustainability, supercapacitors have attracted more attention as a final application for CDC in energy storage in a circular economy perspective. Nonetheless, even within this same device category, it is quite difficult to compare the specific properties of electrode materials in different reported works as the overall performance in energy storage applications is strictly dependent on the device configuration and on the combination of different components and their processing.
Supercapacitors are high-power devices and their performance is assessed by the energy density figure of merit defined as follows:
The useful operating voltage window V depends on the type of electrolytic solution; in case of electrolytes in aqueous solution, it is less than or equal to 1.2 V while reaches 2.7 V for electrolytes in organic solvents.32 The specific capacitance C depends on the nature of the carbon material used in the device; the correlation between the structure and the properties of porous carbon materials is still under debate,28,29,30,31,32,33,43 but it is widely accepted that a large surface area and hierarchical porosity are beneficial to increase the specific charge storage capacity of the carbon material expressed in F g−1 for the gravimetric capacitance.
The value of the specific capacitance is also influenced by the charge and discharge rate of the capacitor, though for the final application, this is less relevant, as typical charge/discharge rates of supercapacitors are generally 2–3 orders of magnitude higher than batteries.32 In the studies reported here, charge and discharge rates typically range from 0.1 to 20 A g−1 with common values being 0.5 or 1 A g−1. The main influence on the carbon structure, especially in the case of a quite homogenous class of a biomass precursor as in the case of coffee waste, is due to the carbonization and activation procedure, and a number of conditions and different chemical and physical activating agents have been proposed.
An overview of coffee waste-derived supercapacitors is given in Table 1 presented in Appendix 1, where the main strategies and results for different materials and devices are presented. The papers collected in the table often do not indicate the carbon yield of the activation process. Another important aspect that is often missing is the mass loading per surface unit of the active carbon material. These two aspects are indeed crucial for the commercial exploitation of coffee waste, and more in general of any biomass, for the preparation of electrochemically active porous carbon materials.
The pioneering studies in the field focused on the challenging importance of fine-tuning activation processes in terms of porogen/activating agents and thermal conditions in order to control the pore distribution in bio-based carbon materials.44 This aspect is crucial for ion penetration and consequently for the specific capacitance of the material, and it set the ground for the following investigations.
Spent coffee grounds as raw material for electrodes in supercapacitors were firstly used to prepare activated carbon by thermal activation in 2008.44 The studies mainly focused on the role of the porosity and micro- and meso-pore distribution in the final material, and thus, different activating agents were investigated like ZnCl2, FeCl3, and MgCl2.44,45
The benefic role of meso-pores in ion transport at fast charge was also reported by the authors, who further explored the contribution of micro-pore and meso-pore surface areas to the specific capacitance of activated CDC and reported a high specific capacitance on the micro-pore surfaces at 0.05 Ag−1 decreasing at higher current loads.46
Carbonization followed by CO2 activation was also adopted for used coffee grounds to produce a high surface area (1867 m2 g−1) and highly porous (1.52 cm3 g−1 total pore volume) carbon material with a good specific capacitance in organic electrolytes.47
Yang et al. reported later a very high surface area of 2558 m2 g−1 for CDC produced by KOH activation at 800 °C preceded by carbonization at 600 °C in an inert atmosphere. The study focused on the correlation between the pore size in the carbon material and the dimension of the desolvated ions in the organic electrolyte. Contrary to expectations, it was proposed that bare ions with a dimension bigger than the material pore size could still penetrate into the pores due to ion distortion and pore expansion caused by voltage-induced forces at high voltage value.48
Unprecedented high values in terms of surface area (3549 m2 g−1) and total pore volume (2.41 cm3 g−1) and very good electrochemical properties (440 F g−1 at 0.5 A g−1) have been very recently reported by Liu and coworkers. They achieved such results by performing a catalytic pyrolysis of coffee biomass in the presence of FeCl3 before activation with KOH. This first carbonization step produced an ordered meso-porous carbon in quite high yield (42.5%) and led to a hierarchical porous carbon with a large volume of meso- and macro-pores (1.63 cm3 g−1). The larger pores were considered responsible for the fast ion transport/diffusion ions which gave a good retention of the specific capacitance at high current densities (67% at 20 A g−1).49
The ratio between KOH and carbon was confirmed to have a strong impact on the activation procedure also by Lee et al. who very recently obtained hierarchical porous carbon with a 3065 m2 g−1 surface area and a good specific capacity of 127.8 F g−1 in sodium sulfate electrolyte with a good stability over 2500 cycles, by employing a KOH/carbon ratio of 8:1 in the activation step.50
It is interesting to note as a recent work by Gadipelli et al. highlighted the high performances of highly porous CDC for electrochemical energy storage applications compared to other nanoporous materials, as pinecones-derived nanoporous carbons, graphene oxide, and metal–organic frameworks. Besides electrical charge storage, CO2, H2, and water storage multifunctional properties were also examined. Coffee grounds highly hierarchical porous carbon obtained by KOH activation outperformed other materials both in CO2 and H2 absorption and in specific capacitance, thanks to an exceptionally high surface area of 2656 m2 g−1 and total pore volume of 1.13 cm3g−1, with a calculated percentage of 59% of sub-nm pores.51
Together with coffee waste source, carbon produced from other industrial residues of coffee was adopted. Jisha et al. reported on activated carbon produced from coffee shells. ZnCl2 was used as activating agent leading to activated carbon materials with 842 m2 g−1 surface area and 156 F g−1 specific capacitance.52 Another industrial residue of coffee, i.e., coffee endocarp (husk), was proposed to obtain low-cost bio-based activated carbon fibers (ACFs), with the major advantage of eliminating the binder in the electrode preparation. Monolithic electrodes were prepared by pressing the carbonized fibers activated by CO2 exposure at 800 °C reaching values of specific capacitance between 149 and 176 F g−1.53 Besides the studies on coffee solid lignocellulosic residues, a huge coffee industry waste is represented by waste sludge from beverage manufacturers. Very recently, this stream was exploited for the generation of active materials for energy storage devices by Jung et al. Activated hydro-char (AHC) was produced by hydrothermal carbonization of waste coffee sludge (230 °C, 4 h) and subjected to conventional KOH activation. AHC was reported to have mainly an amorphous structure, but the partial graphitization of the glucose ring from the highly cellulosic fraction of waste sludge was shown. A specific capacitance of 140 F g−1 retained at 97% after 1500 cycles was recorded.54
Another aspect that attracted the attention of the scientific community is the possibility to enrich the CDC with heteroatoms to improve the material performance. The phosphorus enrichment of activated CDC was reported by Huang et al. Despite a lower surface area, total pore volume, and specific capacitance, the activation with phosphoric acid led to an increase in the operating voltage window and consequently an overall increased energy density of the related device at high current densities, also with a good stability (retaining 82% capacity after 10,000 cycles).55
In 2015, Yun et al. reported on activated carbon obtained from waste coffee grounds by KOH activation, after a wet exfoliation in DMF aided by ultrasonication. The process originated hierarchically porous carbon nanosheets with high aspect ratio nanostructures along with amorphous carbon containing different heteroatoms (N, O, S).56 The presence of such heteroatoms was further exploited to prepare functionalized meso-porous carbon nanosheets (FM-CNS) suitable to be used in asymmetric hybrid capacitors. As a matter of fact, it was found that the heteroatom content aided charge storage and their combinations could act as redox center for lithium-ion storage.57 These heteroatom-rich carbon nanosheets exhibited also a high specific capacitance up to 438.5 F g−1 in symmetric supercapacitors operating in neutral and basic aqueous electrolytes.58 Afterwards, the authors extended the use of such materials to sodium-intercalating hybrid energy storage devices by effectively combining a FM-CNS cathode with a hierarchically porous carbon nanowebs anode prepared from bacterial cellulose.59 Based on these results, the carbon materials obtained by 800 °C pyrolysis were further treated thermally up to 2800 °C to extend the graphitization level and used in sodium-ion batteries with a specific energy of 240 Wh kg−1 and good stability over 200 cycles.60
Following the pioneering works by Hulicova-Jurcakova and colleagues, Wang et al. further investigated coffee waste-based carbon in symmetric supercapacitors. They obtained a very good specific capacitance of 475 F g−1 at 0.1 A g−1 in 6 M KOH aqueous electrolyte and excellent stability after 10,000 cycles even at high current densities. These superior performances were attributed to the controlled distribution of micro- and meso-pore surface areas (890 and 732 m2 g−1) and to the presence of N-containing functional groups.61
The effectiveness of activated carbon doping by heteroatoms was further explored by Ma et al. who reported a one-step activation procedure for the preparation of S-doped activated carbon (SAC). They added magnesium sulfate as dopant obtaining a surface area of 1079 m2 g−1 with a high electrical conductivity (1026 S cm−1). SACs also showed a very good retention of initial capacitance (97.6%) at high current densities of 6 A g−1 after 10,000 cycles. The mass loading of the carbon material was pushed up to 7.5 mg cm−2. The increase of the mass loading of the electroactive material is expected to proportionally increase the specific areal capacitance. On the other hand, the high mass loading can lead to a more difficult diffusion of the electrolyte within the porous film, thus limiting the overall device efficiency in storing charges and ion penetration in pores. They reported still a high specific capacitance value of 200 F g−1, of utmost importance for transferring lab results to commercially available supercapacitor devices.62
A dual doping by sulfur and nitrogen was achieved by using cysteine in a multi-step activation procedure. After a first carbonization of coffee waste and ZnCl2 activation, cysteine was added to the thermal treatment to obtain N/S dual-doped carbon materials. The low weight ratio of cysteine/carbon was preferred, as the dual N/S heteroatoms were suggested to reduce the surface area due to pore damage. At 30% cysteine loading, the resulting N/S-doped carbon showed a 66% increase in specific capacitance.63
Additional examples of dual and single heteroatom doping were reported by Ramasahayam and coworkers, where P and N co-doping were achieved by microwave-induced activation of coffee grounds mixed with ammonium polyphosphate at different weight ratios,64 and by Choi and coworkers, where N-doping was obtained by performing the carbonization of commercial ground coffee powder mixed with melamine as nitrogen donor prior to conventional KOH activation.65
In the most recent report on the use of coffee waste for electrochemical supercapacitors, CO2-assisted thermal activation was described as an effective method to also preserve the natural N-doping of activated carbon owing to the presence of proteins as natural N-sources. The activation temperatures adopted (600–750 °C) were lower than in the case of past CO2 activation studies, obtaining a carbon material with > 3%/w of nitrogen. A very good distribution of micro- and meso-pores (47 and 53%, respectively) was obtained with a simple chemical-agent-free activation that resulted in a specific capacitance of 190 F g−1 at 1 A g−1 well retained after 2000 cycles.66
As aforementioned, a common approach to increase the specific capacitance of activated carbon supercapacitors is to add to the electrodes metal oxides with a pseudocapacitive behavior thanks to redox reactions at the metal centers. This was the case for ZnCl2-activated carbon prepared from coffee husks subsequently combined by wet ball milling in ethanol with CeO2 nanoparticles. The composite material with a 30% CeO2 loading presented a surface area of 270 m2 g−1 reaching a specific capacitance up to 62.4 F g−1 when used as anode in Li-ion cells, much higher than carbon alone.67
A slightly different approach was proposed for composites of coffee bean waste with iron waste in the form of Fe2O3. Fe-decorated nano-porous carbon (PC/Fe) was obtained by adopting a mix of carbonized coffee bean waste with iron oxide and by means of a carbothermic reduction. By washing out Fe with hydrochloric acid, the obtained PC showed an increase in surface area (252.11 m2 g−1) with a specific capacitance of 27 F g−1 retained over 5000 cycles.68
Very recently, carbon nanotubes and graphene nanosheets have been used to create a hierarchical porous structure on a 3D skeleton derived from coffee waste,69 while SnO2 and Ru composites with CDC have been proposed for hybrid capacitors.70,71
As already mentioned, the binder, usually an electrically inactive material, can be crucial not only for the processing of the carbon to prepare the relevant electrode, but also for the overall performance of the material and device. Reduced graphene oxide (RGO) was used as a conductive binder for activated CDC, in place of the conventional PVDF. RGO at 2%/w was shown to preserve the high surface area and porosity of the activated carbon, improve the conductivity of the material, and allow a nearly four times increase in the specific capacitance compared to PVDF-based electrodes, both in aqueous and ionic liquid electrolyte. Moreover, such high initial capacitance was retained for more than 90% after 5000 cycles in KOH electrolyte and for more than 80% after 10,000 cycles in ionic liquid electrolyte.72
Insights on the difference of activating agents in a one-step carbonization and activation of waste coffee ground were provided more recently by Chiu and Lin. In a typical procedure, KOH, NaOH, HCl, H3PO4, ZnCl2, and FeCl3 were adsorbed on coffee grounds by wet impregnation, and the resulting powders were then heated at 700 °C under nitrogen atmosphere. In terms of electrochemical energy storage properties, KOH was shown the best activating agent in the explored conditions, as it provided a carbon material with a specific capacitance 30 to 60% higher than the other activating agents and with an optimal retention in aqueous electrolyte after 8000 cycles.73
Different activation procedures were also compared for producing micro-porous carbon from coffee husk (CH) precursor. Steam, KOH and K2CO3 were used to activate carbonized CH, resulting in high surface area and porous carbon materials. Despite the highest surface area and total pore volume obtained with KOH, steam and potassium carbonate activation led to higher specific capacitance materials. The lowest electrochemical performances of KOH activated CH were explained by a more disordered, low graphitic structure with a lower electrical conductivity, and a reduced penetrability of the electrolyte in the active layer and, thus, a reduced ion diffusion into micro-pores. Among the three methods, K2CO3 activation was considered the best for producing an activated CH with a good specific capacitance (129 F g−1) with a green route and higher carbon yield (18.9%). The better electrochemical performance was ascribed to a higher crystallinity of the materials, leading to a higher electrical conductivity, and level of surface functionalization, especially oxygen-containing functional groups, with beneficial effects for pseudocapacitive contribution to the overall capacitance and reduced charge-transfer resistance in aqueous electrolyte.74
CH were also used as starting material for further studies on different activation conditions, exploring the effect of different activation temperatures (600–700 °C) and different weight ratios (100–150%) in the impregnation by H3PO4 used as chemical activating agent. The best results in terms of higher surface area and electrochemical properties were obtained at higher impregnation proportions and temperatures, with a specific capacitance of 186 F g−1 and a surface area of 734 m2 g−1.75
To maximize the value recovery from spent coffee grounds, Adan-Mas and coworkers performed a hydro-alcoholic extraction of valuable polyphenols before the carbonization and activation of coffee waste. They studied different activation methods (physical and chemical-KOH) and temperatures between 700 and 850 °C. A higher temperature activation led to higher surface area and porosity, though a very low carbon yield was achieved (4–5%/w) compared to the starting precursor. KOH activation also gave slightly better results in terms of specific capacitance, though physically activated carbon showed a superior retention of initial capacitance. Chemically activated samples showed a higher disorder (lower graphitization) and open porosity facilitating the electrolyte penetration and thus reducing the resistance of the whole system. In this work, Na2SO4 was used as a non-toxic and inexpensive neutral electrolyte.76
Coffee waste has been used not only to prepare activated carbon by direct controlled carbonization, but also different nanostructures and composites suitable for electrochemical energy storage devices, through a few different approaches reported below.
An attempt to prepare 2D carbon nanostructures from coffee waste to be used in electrochemical energy storage application is represented by the work of Wang et al. who prepared graphene-sheet fibers (GSF) by microwave plasma irradiation, increasing the areal capacitance to 1293.33 mF cm−2 compared to bare Pt and glassy carbon electrodes (223.93 and 71.05 mF cm−2, respectively). Such results are interesting for the application of bio-based carbon materials in the fabrication of miniaturized supercapacitors for disposable micro-electronics and IoT devices.77
A very high specific capacitance of 1428 F g−1 was reported when combining in a composite electrode 2D nanostructured carbon from coffee waste with polyaniline (PANI) and silver nanoparticles. The carbon material used in this work was described as coffee extracted porous graphene oxide (CEPG) and mixed with aniline and silver nitrate to produce a CEPG-PANI-Ag nanocomposite by an in-situ polymerization process.78
Beyond the number of reports on supercapacitors and also other applications of CDC for electrochemical energy storage and conversion have been studied. Two promising directions are the use in batteries, mainly lithium- and sodium-ion secondary batteries, and in fuel cells.
Batteries (lithium and post-lithium)
It is important to mention that coffee waste-derived carbon materials have been proposed both as a component for cathode composites and as a substitute for natural graphite in lithium-ion, advanced lithium-ion, and post-lithium-ion batteries. Despite its bio-based nature, the impact of using biomass-derived carbon in batteries has a lower impact on the overall device sustainability.18 Therefore, here we will just briefly comment on the major inflection points in the development of CDC concept cathodes and anodes for batteries. (Appendix 1, Table 2).
Although the first contributions were reported in early 2000s, showing promising potential for coffee shells and beans-derived carbon,79,80,81 more recently coffee grounds got back attention as carbon precursor for lithium- and sodium-ion batteries. Improvements in anode performances were reported thanks to the porous honeycomb structure,82 and both micro-porous83 and non-porous structures84 were demonstrated to be effective over 100 cycles.
Very recently, intrinsic coffee N-doping in graphene-like structures were reported to contribute to the high reversible capacity and stability.85 In addition, also external nitrogen-doping sources were used, as in the case of hexamethylenetetramine86 or urea.87 Many of these materials were proposed as anode for both lithium- and sodium-ion batteries, while few studies focused specifically on the latter class of devices showing promising results.60,88,89
Coffee waste was also used as composite material for advanced Li and Na batteries cathodes. Recently, the feasibility of CDC was indeed demonstrated for Li–S and Li–Se cathodes with a good stability after few hundreds cycles.90,91 NaCrO2 was effectively combined with N-doped CDC to produce a composite with 73% capacity retention after 500 cycles.92 Guo et al. reported an even superior cycling stability (2000 cycles) achieved in advanced cathodes for Na-S batteries, owing to the ultramicropores in the carbon structure which can suppress the formation of polysulfides, responsible for the poor cycling performances of room temperature.93
Finally, some studies on CDC are notable for future directions focusing on both high performance and device sustainability. Following the aforementioned studies of Jin and colleagues,57,59 coffee waste was powdered with graphene oxide and used to prepare both hard carbons and activated carbon in order to increase the device energy density at high power densities. The two different materials were used to assemble an asymmetric Li-ion capacitor which showed considerable results in terms of energy density, power, and cyclability.94
Another approach to increase the sustainability of batteries is electrode processing. Following this direction, the use of coffee waste-derived hard carbon as green material for electrochemical energy storage was coupled with greener binders in the fabrication of anodes for both Li- and Na-ion batteries. Specifically, alginate, carboxy-methyl-cellulose, and poly-acrylic acid were used, and when compared with commercial fluorinated binders (PVDF), they showed remarkable improvements in terms of both reversible discharge capacity and cyclability.95 Hence, the efforts in using bio-based waste coffee materials and green additives and processes represent a promising strategy not only to improve the overall sustainability of electrochemical energy storage devices, but also their performances.
Fuel cells
Despite at an earlier stage than supercapacitors and batteries, a different exploitation strategy for coffee and CDC materials has been proposed in the field of the “hydrogen economy” with their use in fuel cells. Although fuel cells are electrochemical energy conversion device, they are worth being mentioned as alternative to batteries and supercapacitors for an electrified society.
Fuel cell technologies and their role in an electrified society have been extensively reviewed elsewhere,96,97 so here we just provide an overview on a few recent examples of CDC material as potential substitute in cathodes for the oxygen reduction reaction (ORR). Besides the use in fuel cell cathodes, the use of CDC in anodes for direct methanol fuel cells (DMFC) and Microbial Fuel Cells (MFC) has been also recently explored.98,99
The use of CDC as fuel cell cathode materials has been proposed to both improve performances in ORR and to face the problematic use of platinum-group metals and relevant alloys in these devices. Platinum-based electrodes despite their outstanding catalytic properties in ORR, suffer from high cost, scarcity, and low performance over time. The main reason for this reduced durability is usually associated with the agglomeration of Pt nanostructures and the poisoning from methanol and CO traces.100 Hence, many efforts have been spent to design Pt-free electrode materials and between different alternatives heteroatoms-doped porous carbon and especially biomass-derived one has shown many advantages.100,101,102 Spent coffee grounds have been firstly used for ORR by Kumar et al. who reported on the microwave-assisted preparation of carbon co-doped with P and N atoms by ammonium phosphate. The oxygen evolution performances in alkaline solutions in terms of onset potential and current density were found to be comparable to those of commercial 20% Pt/C electrodes.103 Even better performance in terms of kinetics and electrochemical stability than state-of-the-art Pt/C ORR electrocatalyst has been reported in case of intrinsically N-doped porous carbon derived from coffee bean waste.104 The same intrinsic N-doping strategy was adopted by Chung et al. who synthesized hierarchical N-doped porous carbon from coffee waste by ZnCl2 activation, confirming the improvements with respect to Pt-containing electrodes in oxygen reduction.105 Further insights on N and P doped CDC were provided by Sahu and coworkers by using melamine and triphenylphosphine. The N–P-co-doped carbon showed the best performance especially in terms of stability of the ORR half-wave potential over thousands of cycles when compared with the commercial 20% Pt/C catalyst.106 Another report from the same group focused on N and F co-doping, achieved by triazine and ammonium fluoride, reaching again very good ORR performances in terms of electrode stability over 10,000 cycles in both acidic and alkaline media.107
General considerations on coffee-derived carbons for energy storage applications
The use of coffee waste and byproducts as raw materials for electroactive carbon for energy storage devices is not yet commercially exploited. Three main applications examined for coffee-derived carbon in electrochemical energy storage and conversion are supercapacitors, metal-ion batteries (lithium and beyond), and fuel cells (Fig. 7).
Most studies in this field focus on supercapacitor applications. The use of activated carbon from waste biomass sources for this class of devices is already technologically mature and originated commercial products; thus, coffee represents a valid alternative in this context and few general considerations can be made considering the state-of-the-art supercapacitors.
A first consideration is related to the material properties that have been discussed above. As highlighted, due to differences in the preparation of materials and devices (e.g., activating agents and processes, electrode mass loadings, electrolytes, and separators) and in the characterization parameters (e.g., cell setup, charge/discharge rates, and cycles), it is almost impossible to get an absolute comparison of different carbon material performances, and thus, a comparison of the final devices is usually preferred. Nonetheless, it is useful to compare the main figure of merit, the specific gravimetric capacitance (F/g) for coffee-derived carbons with similar alternatives. A comprehensive recent overview of different bio-waste-derived ACs has shown that fruit peels generally resulted in carbons with higher values of capacitance in common aqueous electrolytes such as concentrated sulfuric acid and potassium hydroxide solutions. As a reference, AC from peanut shell (activated by ZnCl2) and orange peel (activated by KOH) showed in 1 M H2SO4 a 340 and 460 F/g specific capacitance, respectively.109 Table 1 presents coffee-derived carbons specific capacitance is within this range: 368 F/g in 1 M H2SO4 with ZnCl2 activation,44 or 475 F/g in 6 M KOH with KOH activation.61
Besides the material intrinsic properties, techno-economic considerations can also be put forth to evaluate the potentiality of coffee waste valorization through energy storage applications. Wang et al. recently reported on the quantification of environmental and economic impacts of AC production from lignocellulosic biomass.110 For a 3 ton/day plant, lignocellulosic feedstock accounts for 4.4% of the total annual operating costs. Although comparing coffee waste as direct drop-in in such a process could be quite a rough approximation, this study helps understand the major costs of AC commercial production from biomass. Interestingly with a CAPEX of USD 6.66 million, a 15.45 million OPEX was projected, with the activating agent (KOH) as the major cost accounting for 81.53% of total annual operating costs. These figures clearly show how coffee waste as raw materials would have a minimal effect on the overall process costs. Conversely, it could be very interesting to evaluate in the same conditions of this report the overall AC yield and premium properties by switching the raw materials to coffee waste, with a market price of AC that can range between 17 and 22 USD/kg, corresponding to a decrease of the discounted payback period (DPB) from 10 to less than 2 years.110 In addition to these technological and economic considerations, it is worth to comment LCA aspects for coffee-derived carbons with state-of-the-art activated carbons for energy storage. Wang et al. reported that the AC production (carbonization and activation) accounted for a contribution of 95.8 to 99.6% in all the ten environmental impact categories considered (e.g., 96.7% of GHG emission occurred in the process of carbon production).110 Thus, it can be expected that using coffee waste as raw material for activated carbon in place of other lignocellulosic feedstock would end in minor effects on the overall LCA results.
A final consideration is related to the risks of AC production from coffee waste, mainly associated to the low technological maturity. The main risk for the commercial application of this valorization strategy is associated with the lack of a pilot study for the production process. The AC yields, physical and chemical properties, and their consistency at a pilot scale production are essential data for the actual exploitation of coffee waste. In this context, another intrinsic risk is represented by the quality of the input biomass that can be variate depending on the specific area of collection and storage. In this perspective, a pilot study would be crucial to identify key metrics (e.g., surface area, heteroatom doping, electrical conductivity) to implement necessary quality control (QC) strategies. Considering all these aspects, a pilot production study is needed to lay out the technical risk mitigation strategy. On the techno-economical side, in analogy with other coffee waste biorefinery concepts, the supply chain faces several risks (e.g., volume and price fluctuations, contaminations, competing alternative uses), and although complete analysis models are lacking, preliminary evidence showed that, despite more complex logistics, a centralized collection and transformation of coffee waste are to be preferred to small, decentralized AC productions.15
Conclusions with comments on key trends
Despite still in a relatively early research stage, coffee waste-derived carbon represents a promising material for electrochemical energy conversion and storage devices. Interestingly, it has been demonstrated to be effective in different classes of devices, namely metal-ion batteries, supercapacitors, and fuel cells which are expected to be complementary key players for stationary and portable energy conversion and storage systems in the next decades.
One of the main aspects to be carefully evaluated, which is common to any circular economy model, is the logistics of the coffee waste supply, to actually provide an economic and environmental sustainability along the entire life cycle.
Concerning some key material science aspects emerging from this overview of the scientific literature, further investigations and improvements are foreseen in controlling the doping of the carbon, the hierarchical porosity of the structure, and maximizing carbon yield. A general method combining all these aspects, while controlling the sustainability of the overall process, is still missing and represents an interesting space for further investigations.
Moreover, along with current carbon material and precious metal substitution, the use of coffee waste-based carbon should be coupled with solvent-less or water-based processing, employing water soluble and possibly bio-based binders and additives, and designing eco-friendly devices and concepts with a focus on the end-of-life options like recycling vs. disposal and degradability.
Finally, a key aspect in the processing of carbon material for energy storage applications is the mass loading of carbon materials, especially for energy storage devices. While a thick electrode is highly desirable to increase the energy density of the device per unit of surface and volume, it is difficult to guarantee a good diffusion for ions and redox species to the active sites.
A comparison with technological, economic, and environmental benefits of alternatives in the strategies required for using recycled energy storage device materials is strongly needed. Nonetheless, despite the open challenges, the feasibility of using coffee waste for electrochemical energy storage represents an opportunity to create a second energizing life for coffee used in our daily life, by building new circular value chains and ecosystems.
References
British coffee association. https://www.britishcoffeeassociation.org/. Accessed 17 May, 2021
Coffee market report April 2021. http://www.ico.org/. Accessed 17 May, 2021
A. Samoggia, B. Riedel, Coffee consumption and purchasing behavior review: Insights for further research. Appetite 129, 70–81 (2018). https://doi.org/10.1016/j.appet.2018.07.002
S.L. Bager, E.F. Lambin, Sustainability strategies by companies in the global coffee sector. Bus Strateg Environ 29, 3555–3570 (2020). https://doi.org/10.1002/bse.2596
B. Notarnicola, G. Tassielli, P.A. Renzulli et al., Environmental impacts of food consumption in Europe. J Clean Prod 140, 753–765 (2017). https://doi.org/10.1016/j.jclepro.2016.06.080
M.M. Mekonnen, A.Y. Hoekstra, The green, blue and grey water footprint of crops and derived crop products. Hydrol Earth Syst Sci 15, 1577–1600 (2011). https://doi.org/10.5194/hess-15-1577-2011
A.K. Chapagain, A.Y. Hoekstra, The water footprint of coffee and tea consumption in the Netherlands. Ecol Econ 64, 109–118 (2007). https://doi.org/10.1016/j.ecolecon.2007.02.022
C. Nab, M. Maslin, Life cycle assessment synthesis of the carbon footprint of Arabica coffee: Case study of Brazil and Vietnam conventional and sustainable coffee production and export to the United Kingdom. Geo Geogr Environ 7, 1–19 (2020). https://doi.org/10.1002/geo2.96
P.S. Murthy, M. Madhava Naidu, Sustainable management of coffee industry by-products and value addition: A review. Resour Conserv Recycl 66, 45–58 (2012). https://doi.org/10.1016/j.resconrec.2012.06.005
B. Janissen, T. Huynh, Chemical composition and value-adding applications of coffee industry by-products: A review. Resour Conserv Recycl 128, 110–117 (2018). https://doi.org/10.1016/j.resconrec.2017.10.001
P. Esquivel, V.M. Jiménez, Functional properties of coffee and coffee by-products. Food Res Int 46, 488–495 (2012). https://doi.org/10.1016/j.foodres.2011.05.028
M. Kamil, K.M. Ramadan, A.G. Olabi et al., Comprehensive evaluation of the life cycle of liquid and solid fuels derived from recycled coffee waste. Resour Conserv Recycl 150, 104446 (2019). https://doi.org/10.1016/j.resconrec.2019.104446
R. Campos-Vega, G. Loarca-Piña, H.A. Vergara-Castañeda, B. Dave Oomah, Spent coffee grounds: A review on current research and future prospects. Trends Food Sci Technol 45, 24–36 (2015). https://doi.org/10.1016/j.tifs.2015.04.012
A. Kovalcik, S. Obruca, I. Marova, Valorization of spent coffee grounds: A review. Food Bioprod Process 110, 104–119 (2018). https://doi.org/10.1016/j.fbp.2018.05.002
J. Massaya, A. Prates Pereira, B. Mills-Lamptey et al., Conceptualization of a spent coffee grounds biorefinery: A review of existing valorisation approaches. Food Bioprod Process 118, 149–166 (2019). https://doi.org/10.1016/j.fbp.2019.08.010
R. Ktori, P. Kamaterou, A. Zabaniotou, Spent coffee grounds valorization through pyrolysis for energy and materials production in the concept of circular economy. Mater Today Proc 5, 27582–27588 (2018). https://doi.org/10.1016/j.matpr.2018.09.078
Strategic Research Agenda for batteries 2020, European Technology and Innovation Platform on Batteries – Batteries Europe, December 2020. https://ec.europa.eu/energy/sites/ener/files/documents/batteries_europe_strategic_research_agenda_december_2020__1.pdf. Accessed 17 May, 2021
S. Dühnen, J. Betz, M. Kolek et al., Toward green battery cells: Perspective on materials and technologies. Small Methods 4, 2000039 (2020). https://doi.org/10.1002/smtd.202000039
Battery 2030+ Roadmap, Inventing the sustainable batteries of the future: Research needs and future actions. https://battery2030.eu/digitalAssets/861/c_861008-l_1-k_roadmap-27-march.pdf. Accessed 17 May, 2021
Green Deal: pile e batterie sostenibili per un’economia circolare e climaticamente neutra, EU Commission, comunicato stampa 10 Dicembre 2020. https://ec.europa.eu/italy/news/20201210_green_deal_pile_e_batterie_sostenibili_per_un_economia_circolare_it. Accessed 17 May, 2021
EU Critical Raw Materials. https://ec.europa.eu/growth/sectors/raw-materials/specific-interest/critical_en. Accessed 17 May, 2021
H. Bae, Y. Kim, Technologies of lithium recycling from waste lithium ion batteries: a review. Mater Adv 2, 3234–3250 (2021). https://doi.org/10.1039/D1MA00216C
G. Harper, R. Sommerville, E. Kendrick et al., Recycling lithium-ion batteries from electric vehicles. Nature 575, 75–86 (2019). https://doi.org/10.1038/s41586-019-1682-5
J. Liu, J. Wang, C. Xu et al., Advanced energy storage devices: basic principles, analytical methods, and rational materials design. Adv Sci 5, 1700322 (2018). https://doi.org/10.1002/advs.201700322
M. Walter, M.V. Kovalenko, K.V. Kravchyk, Challenges and benefits of post-lithium-ion batteries. New J Chem 44, 1677–1683 (2020). https://doi.org/10.1039/C9NJ05682C
V. Hoffmann, M.P. Olszewski, K.M. Swiatek et al., Bio-based electric devices (Elsevier Inc, Amsterdam, 2020)
C. Liedel, Sustainable battery materials from biomass. Chemsuschem 13, 2110–2141 (2020). https://doi.org/10.1002/cssc.201903577
M. Salanne, B. Rotenberg, K. Naoi et al., Efficient storage mechanisms for building better supercapacitors. Nat Energy (2016). https://doi.org/10.1038/nenergy.2016.70
V. Augustyn, P. Simon, B. Dunn, Pseudocapacitive oxide materials for high-rate electrochemical energy storage. Energy Environ Sci 7, 1597 (2014). https://doi.org/10.1039/c3ee44164d
P. Simon, Y. Gogotsi, Perspectives for electrochemical capacitors and related devices. Nat Mater 19, 1151–1163 (2020). https://doi.org/10.1038/s41563-020-0747-z
N.A. Kyeremateng, T. Brousse, D. Pech, Microsupercapacitors as miniaturized energy-storage components for on-chip electronics. Nat Nanotechnol 12, 7–15 (2017). https://doi.org/10.1038/nnano.2016.196
Y. Shao, M.F. El-Kady, J. Sun et al., Design and mechanisms of asymmetric supercapacitors. Chem Rev 118, 9233–9280 (2018). https://doi.org/10.1021/acs.chemrev.8b00252
F. Béguin, V. Presser, A. Balducci, E. Frackowiak, Carbons and electrolytes for advanced supercapacitors. Adv Mater 26, 2219–2251 (2014). https://doi.org/10.1002/adma.201304137
Z. Bi, Q. Kong, Y. Cao et al., Biomass-derived porous carbon materials with different dimensions for supercapacitor electrodes: a review. J Mater Chem A 7, 16028–16045 (2019). https://doi.org/10.1039/C9TA04436A
K. Mensah-Darkwa, C. Zequine, P. Kahol, R. Gupta, Supercapacitor energy storage device using biowastes: a sustainable approach to green energy. Sustainability 11, 414 (2019). https://doi.org/10.3390/su11020414
T. Purkait, G. Singh, M. Singh et al., Large area few-layer graphene with scalable preparation from waste biomass for high-performance supercapacitor. Sci Rep 7, 15239 (2017). https://doi.org/10.1038/s41598-017-15463-w
M. Kumar, M. Hietala, K. Oksman, Lignin-based electrospun carbon nanofibers. Front Mater (2019). https://doi.org/10.3389/fmats.2019.00062
D. Bresser, D. Buchholz, A. Moretti et al., Alternative binders for sustainable electrochemical energy storage: the transition to aqueous electrode processing and bio-derived polymers. Energy Environ Sci 11, 3096–3127 (2018). https://doi.org/10.1039/C8EE00640G
H. Chen, M. Ling, L. Hencz et al., Exploring chemical, mechanical, and electrical functionalities of binders for advanced energy-storage devices. Chem Rev 118, 8936–8982 (2018). https://doi.org/10.1021/acs.chemrev.8b00241
L. Chang, Y.H. Hu, Breakthroughs in designing commercial-level mass-loading graphene electrodes for electrochemical double-layer capacitors. Matter 1, 596–620 (2019). https://doi.org/10.1016/j.matt.2019.06.016
Y. Dong, J. Zhu, Q. Li et al., Carbon materials for high mass-loading supercapacitors: filling the gap between new materials and practical applications. J Mater Chem A 8, 21930–21946 (2020). https://doi.org/10.1039/D0TA08265A
H. Sun, J. Zhu, D. Baumann et al., Hierarchical 3D electrodes for electrochemical energy storage. Nat Rev Mater 4, 45–60 (2019). https://doi.org/10.1038/s41578-018-0069-9
J. Yin, W. Zhang, N.A. Alhebshi et al., Synthesis strategies of porous carbon for supercapacitor applications. Small Methods 4, 1–31 (2020). https://doi.org/10.1002/smtd.201900853
T.E. Rufford, D. Hulicova-Jurcakova, Z. Zhu, G.Q. Lu, Nanoporous carbon electrode from waste coffee beans for high performance supercapacitors. Electrochem commun 10, 1594–1597 (2008). https://doi.org/10.1016/j.elecom.2008.08.022
T.E. Rufford, D. Hulicova-Jurcakova, E. Fiset et al., Double-layer capacitance of waste coffee ground activated carbons in an organic electrolyte. Electrochem commun 11, 974–977 (2009). https://doi.org/10.1016/j.elecom.2009.02.038
T.E. Rufford, D. Hulicova-Jurcakova, Z. Zhu, G.Q. Lu, A comparative study of chemical treatment by FeCl3, MgCl2, and ZnCl2 on microstructure, surface chemistry, and double-layer capacitance of carbons from waste biomass. J Mater Res 25, 1451–1459 (2010). https://doi.org/10.1557/jmr.2010.0186
D. Tashima, Y. Hamasuna, D. Mishima et al., Microporous activated carbons from used coffee grounds for application to electric double-layer capacitors. IEEJ Trans Electr Electron Eng 9, 343–350 (2014). https://doi.org/10.1002/tee.21978
C.M. Yang, H.J. Jung, Y.J. Kim, Anomalous cyclic voltammetric response from pores smaller than ion size by voltage-induced force. J Colloid Interface Sci 446, 208–212 (2015). https://doi.org/10.1016/j.jcis.2015.01.041
X. Liu, S. Zhang, X. Wen et al., High yield conversion of biowaste coffee grounds into hierarchical porous carbon for superior capacitive energy storage. Sci Rep 10, 1–12 (2020). https://doi.org/10.1038/s41598-020-60625-y
U.W. Lee, G. Yang, S.J. Park, Improvement of mesoporosity on supercapacitive performance of activated carbons derived from coffee grounds. Bull Korean Chem Soc 42, 748–755 (2021). https://doi.org/10.1002/bkcs.12251
S. Gadipelli, C.A. Howard, J. Guo et al., Superior multifunctional activity of nanoporous carbons with widely tunable porosity: enhanced storage capacities for carbon-dioxide, hydrogen, water, and electric charge. Adv Energy Mater 10, 1–14 (2020). https://doi.org/10.1002/aenm.201903649
M.R. Jisha, Y.J. Hwang, J.S. Shin et al., Electrochemical characterization of supercapacitors based on carbons derived from coffee shells. Mater Chem Phys 115, 33–39 (2009). https://doi.org/10.1016/j.matchemphys.2008.11.010
J.M. Valente Nabais, J.G. Teixeira, I. Almeida, Development of easy made low cost bindless monolithic electrodes from biomass with controlled properties to be used as electrochemical capacitors. Bioresour Technol 102, 2781–2787 (2011). https://doi.org/10.1016/j.biortech.2010.11.083
H. Jung, J. Kang, I. Nam, Graphitic porous carbon derived from waste coffee. Material 13, 3972 (2020)
C. Huang, T. Sun, D. Hulicova-Jurcakova, Wide electrochemical window of supercapacitors from coffee bean-derived phosphorus-rich carbons. Chemsuschem 6, 2330–2339 (2013). https://doi.org/10.1002/cssc.201300457
Y.S. Yun, M.H. Park, S.J. Hong et al., Hierarchically porous carbon nanosheets from waste coffee grounds for supercapacitors. ACS Appl Mater Interfaces 7, 3684–3690 (2015). https://doi.org/10.1021/am5081919
Y.S. Yun, D.H. Kim, S.J. Hong et al., Microporous carbon nanosheets with redox-active heteroatoms for pseudocapacitive charge storage. Nanoscale 7, 15051–15058 (2015). https://doi.org/10.1039/c5nr04231c
M.H. Park, Y.S. Yun, S.Y. Cho et al., Waste coffee grounds-derived nanoporous carbon nanosheets for supercapacitors. Carbon Lett 19, 66–71 (2016). https://doi.org/10.5714/CL.2016.19.066
M.Y. Song, N.R. Kim, S.Y. Cho et al., Asymmetric energy storage devices based on surface-driven sodium-ion storage. ACS Sustain Chem Eng 5, 616–624 (2017). https://doi.org/10.1021/acssuschemeng.6b01991
M.E. Lee, H.W. Kwak, H.J. Jin, Y.S. Yun, Waste beverage coffee-induced hard carbon granules for sodium-ion batteries. ACS Sustain Chem Eng 7, 12734–12740 (2019). https://doi.org/10.1021/acssuschemeng.9b00971
C.H. Wang, W.C. Wen, H.C. Hsu, B.Y. Yao, High-capacitance KOH-activated nitrogen-containing porous carbon material from waste coffee grounds in supercapacitor. Adv Powder Technol 27, 1387–1395 (2016). https://doi.org/10.1016/j.apt.2016.04.033
X. Ma, L. Zhao, Z. Yu et al., Excellent compatibility of the gravimetric and areal capacitances of an electric-double-layer capacitor configured with S-doped activated carbon. Chemsuschem 11, 3766–3773 (2018). https://doi.org/10.1002/cssc.201801767
J. Hong, H. Kim, J.E. Lee et al., Nitrogen and sulfur dual-doped porous carbon derived from coffee waste and cysteine for electrochemical energy storage. Korean J Chem Eng 37, 1218–1225 (2020). https://doi.org/10.1007/s11814-020-0544-z
S.K. Ramasahayam, A.L. Clark, Z. Hicks, T. Viswanathan, Spent coffee grounds derived P, N co-doped C as electrocatalyst for supercapacitor applications. Electrochim Acta 168, 414–422 (2015). https://doi.org/10.1016/j.electacta.2015.03.193
J. Choi, C. Zequine, S. Bhoyate et al., Waste coffee management: deriving high-performance supercapacitors using nitrogen-doped coffee-derived carbon. C 5, 44 (2019). https://doi.org/10.3390/c5030044
R. Hossain, R.K. Nekouei, I. Mansuri, V. Sahajwalla, In-situ O/N-heteroatom enriched activated carbon by sustainable thermal transformation of waste coffee grounds for supercapacitor material. J Energy Storage 33, 102113 (2021). https://doi.org/10.1016/j.est.2020.102113
S. Phokha, S. Hunpratub, B. Usher et al., Effects of CeO2 nanoparticles on electrochemical properties of carbon/CeO2 composites. Appl Surf Sci 446, 36–46 (2018). https://doi.org/10.1016/j.apsusc.2018.02.209
C.H. Kim, J.H. Kim, S.M. Lee, Transformation of solid waste into nanoporous carbon via carbothermic reduction. ACS Omega 3, 7904–7910 (2018). https://doi.org/10.1021/acsomega.8b01280
W. He, P.-G. Ren, Z. Dai et al., Hierarchical porous carbon composite constructed with 1-D CNT and 2-D GNS anchored on 3-D carbon skeleton from spent coffee grounds for supercapacitor. Appl Surf Sci 558, 149899 (2021). https://doi.org/10.1016/j.apsusc.2021.149899
C. Liu, Z. He, J. Niu et al., Two-dimensional SnO2anchored biomass-derived carbon nanosheet anode for high-performance Li-ion capacitors. RSC Adv 11, 10018–10026 (2021). https://doi.org/10.1039/d1ra00822f
M. Akdemir, T. Avci Hansu, A. Caglar et al., Ruthenium modified defatted spent coffee catalysts for supercapacitor and methanolysis application. Energy Storage 3, 1–10 (2021). https://doi.org/10.1002/est2.243
J.H. Choi, C. Lee, S. Cho et al., High capacitance and energy density supercapacitor based on biomass-derived activated carbons with reduced graphene oxide binder. Carbon N Y 132, 16–24 (2018). https://doi.org/10.1016/j.carbon.2018.01.105
Y.H. Chiu, L.Y. Lin, Effect of activating agents for producing activated carbon using a facile one-step synthesis with waste coffee grounds for symmetric supercapacitors. J Taiwan Inst Chem Eng 101, 177–185 (2019). https://doi.org/10.1016/j.jtice.2019.04.050
N. Ramirez, F. Sardella, C. Deiana et al., Capacitive behavior of activated carbons obtained from coffee husk. RSC Adv 10, 38097–38106 (2020). https://doi.org/10.1039/d0ra06206e
E. Del Ángel-Meraz, O.-F. de Jesús, E.R. Morales et al., The use of activated carbon from coffee endocarp for the manufacture of supercapacitors. J Mater Sci Mater Electron 31, 7547–7554 (2020). https://doi.org/10.1007/s10854-020-03123-1
A. Adan-Mas, L. Alcaraz, P. Arévalo-Cid et al., Coffee-derived activated carbon from second biowaste for supercapacitor applications. Waste Manag 120, 280–289 (2021). https://doi.org/10.1016/j.wasman.2020.11.043
Z. Wang, H. Ogata, S. Morimoto et al., Microwave plasma-induced graphene-sheet fibers from waste coffee grounds. J Mater Chem A 3, 14545–14549 (2015). https://doi.org/10.1039/c5ta03833b
P. Sundriyal, S. Bhattacharya, Polyaniline silver nanoparticle coffee waste extracted porous graphene oxide nanocomposite structures as novel electrode material for rechargeable batteries. Mater Res Express (2017). https://doi.org/10.1088/2053-1591/aa5ece
Y. Nishi, Carbonaceous materials for lithium ion secondary battery anodes. Mol Cryst Liq Cryst Sci Technol Sect A Mol Cryst Liq Cryst 340, 419–424 (2000). https://doi.org/10.1080/10587250008025503
E. Endo, T. Kihira, S. Yamada et al., Surface treatment of carbon electrodes by electron beam irradiation. J Power Sources 93, 215–223 (2001). https://doi.org/10.1016/S0378-7753(00)00581-4
Y.J. Hwang, S.K. Jeong, K.S. Nahm et al., Pyrolytic carbon derived from coffee shells as anode materials for lithium batteries. J Phys Chem Solids 68, 182–188 (2007). https://doi.org/10.1016/j.jpcs.2006.10.007
X.X. Li, J. Hou, X.W. Wang et al., Preparation and electrochemical performance of a honeycomb-like porous anode material. Int J Electrochem Sci 12, 9619–9625 (2017)
J.H. Um, Y. Kim, C.Y. Ahn et al., Biomass waste, coffee grounds-derived carbon for lithium storage. J Electrochem Sci Technol 9, 163–168 (2018). https://doi.org/10.5229/JECST.2018.9.3.163
F. Luna-Lama, D. Rodríguez-Padrón, A.R. Puente-Santiago et al., Non-porous carbonaceous materials derived from coffee waste grounds as highly sustainable anodes for lithium-ion batteries. J Clean Prod 207, 411–417 (2019). https://doi.org/10.1016/j.jclepro.2018.10.024
Q. Xie, S. Qu, Y. Zhang, P. Zhao, Nitrogen-enriched graphene-like carbon architecture with tunable porosity derived from coffee ground as high performance anodes for lithium ion batteries. Appl Surf Sci 537, 148092 (2021). https://doi.org/10.1016/j.apsusc.2020.148092
S.Y. Tsai, R. Muruganantham, S.H. Tai et al., Coffee grounds-derived carbon as high performance anode materials for energy storage applications. J Taiwan Inst Chem Eng 97, 178–188 (2019). https://doi.org/10.1016/j.jtice.2019.01.020
Z. Zhu, K. Bai, H. Zuo et al., One-pot synthesis of MnO/C N-doped hybrid materials for high performance lithium-ion batteries. J Alloys Compd 805, 692–700 (2019). https://doi.org/10.1016/j.jallcom.2019.07.139
G. Gao, L.Z. Cheong, D. Wang, C. Shen, Pyrolytic carbon derived from spent coffee grounds as anode for sodium-ion batteries. Carbon Resour Convers 1, 104–108 (2018). https://doi.org/10.1016/j.crcon.2018.04.001
P.H. Chiang, S.F. Liu, Y.H. Hung et al., Coffee-ground-derived nanoporous carbon anodes for sodium-ion batteries with high rate performance and cyclic stability. Energy Fuels 34, 7666–7675 (2020). https://doi.org/10.1021/acs.energyfuels.0c01105
P. Zhao, M.H.A. Shiraz, H. Zhu et al., Hierarchically porous carbon from waste coffee grounds for high-performance Li–Se batteries. Electrochim Acta 325, 134931 (2019). https://doi.org/10.1016/j.electacta.2019.134931
B. Kim, J. Park, S. Baik, J.W. Lee, Spent coffee derived hierarchical porous carbon and its application for energy storage. J Porous Mater 27, 451–463 (2020). https://doi.org/10.1007/s10934-019-00826-7
S.M. Kang, M.S. Kim, Y. Jeoun et al., NaCrO2/coffee waste–derived nitrogen-doped carbon composite as high-performance cathode material for sodium ion batteries. Bull Korean Chem Soc 40, 857–862 (2019). https://doi.org/10.1002/bkcs.11834
Q. Guo, S. Li, X. Liu et al., Ultrastable sodium-sulfur batteries without polysulfides formation using slit ultramicropore carbon carrier. Adv Sci 7, 1–12 (2020). https://doi.org/10.1002/advs.201903246
J.L. Gómez-Urbano, G. Moreno-Fernández, M. Arnaiz et al., Graphene-coffee waste derived carbon composites as electrodes for optimized lithium ion capacitors. Carbon N Y 162, 273–282 (2020). https://doi.org/10.1016/j.carbon.2020.02.052
H. Darjazi, A. Staffolani, L. Sbrascini et al., Sustainable anodes for lithium- and sodium-ion batteries based on coffee ground-derived hard carbon and green binders. Energies (2020). https://doi.org/10.3390/en13236216
Z.P. Cano, D. Banham, S. Ye et al., Batteries and fuel cells for emerging electric vehicle markets. Nat Energy 3, 279–289 (2018). https://doi.org/10.1038/s41560-018-0108-1
I. Staffell, D. Scamman, A. Velazquez Abad et al., The role of hydrogen and fuel cells in the global energy system. Energy Environ Sci 12, 463–491 (2019). https://doi.org/10.1039/c8ee01157e
Z.K. Ghouri, S. Al-Meer, N.A.M. Barakat, H.Y. Kim, ZnO@C (core@shell) microspheres derived from spent coffee grounds as applicable non-precious electrode material for DMFCs. Sci Rep 7, 1–8 (2017). https://doi.org/10.1038/s41598-017-01463-3
Y.H. Hung, T.Y. Liu, H.Y. Chen, Renewable coffee waste-derived porous carbons as anode materials for high-performance sustainable microbial fuel cells. ACS Sustain Chem Eng 7, 16991–16999 (2019). https://doi.org/10.1021/acssuschemeng.9b02405
M. Sahoo, S. Ramaprabhu, Nitrogen and sulfur co-doped porous carbon: is an efficient electrocatalyst as platinum or a hoax for oxygen reduction reaction in acidic environment PEM fuel cell? Energy 119, 1075–1083 (2017). https://doi.org/10.1016/j.energy.2016.11.066
M. del Cueto, P. Ocón, J.M.L. Poyato, Comparative study of oxygen reduction reaction mechanism on nitrogen-, phosphorus-, and boron-doped graphene surfaces for fuel cell applications. J Phys Chem C 119, 2004–2009 (2015). https://doi.org/10.1021/jp512588r
R. Li, Z. Wei, X. Gou, Nitrogen and phosphorus dual-doped graphene/carbon nanosheets as bifunctional electrocatalysts for oxygen reduction and evolution. ACS Catal 5, 4133–4142 (2015). https://doi.org/10.1021/acscatal.5b00601
S.K. Ramasahayam, S. Azam, T. Viswanathan, Phosphorous, nitrogen co-doped carbon from spent coffee grounds for fuel cell applications. J Appl Polym Sci 132, 1–8 (2015). https://doi.org/10.1002/app.41948
S.M. Unni, L. George, S.N. Bhange et al., Valorization of coffee bean waste: a coffee bean waste derived multifunctional catalyst for photocatalytic hydrogen production and electrocatalytic oxygen reduction reactions. RSC Adv 6, 82103–82111 (2016). https://doi.org/10.1039/c6ra14907c
D.Y. Chung, Y.J. Son, J.M. Yoo et al., Coffee waste-derived hierarchical porous carbon as a highly active and durable electrocatalyst for electrochemical energy applications. ACS Appl Mater Interfaces 9, 41303–41313 (2017). https://doi.org/10.1021/acsami.7b13799
A. Srinu, S.G. Peera, V. Parthiban et al., Heteroatom engineering and Co–doping of N and P to porous carbon derived from spent coffee grounds as an efficient electrocatalyst for oxygen reduction reactions in alkaline medium. ChemistrySelect 3, 690–702 (2018). https://doi.org/10.1002/slct.201702042
S. Akula, A.K. Sahu, Heteroatoms co-doping (N, F) to the porous carbon derived from spent coffee grounds as an effective catalyst for oxygen reduction reaction in polymer electrolyte fuel cells. J Electrochem Soc 166, F93–F101 (2019). https://doi.org/10.1149/2.0441902jes
T.E. Rufford, D. Hulicova-Jurcakova, Z. Zhu, G.Q. Lu, Empirical analysis of the contributions of mesopores and micropores to the double-layer capacitance of carbons. J Phys Chem C 113, 19335–19343 (2009). https://doi.org/10.1021/jp905975q
S. Saini, P. Chand, A. Joshi, Biomass derived carbon for supercapacitor applications: review. J Ener Stor 39, 102646–102667 (2021). https://doi.org/10.1016/j.est.2021.102646
Y. Wang, J. Wang, X. Zhang, D. Bhattacharyya, E.M. Sabolsky, Quantifying environmental and economic impacts of highly porous activated carbon from lignocellulosic biomass for high-performance supercapacitors. Energies 15, 351–370 (2022). https://doi.org/10.3390/en15010351
Acknowledgments
This work was funded by the project Mission Innovation POA 2021-2023 Italian Energy Materials Acceleration Platform—IEMAP.
Funding
Open access funding provided by Consiglio Nazionale Delle Ricerche (CNR) within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Stufano, P., Perrotta, A., Labarile, R. et al. The second life of coffee can be even more energizing: Circularity of materials for bio-based electrochemical energy storage devices. MRS Energy & Sustainability 9, 443–460 (2022). https://doi.org/10.1557/s43581-022-00036-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/s43581-022-00036-w