Abstract
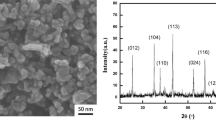
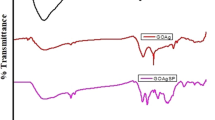
Graphene oxide (GO) is a promising material in improving the corrosion resistance properties of metals. This improvement significantly relies on the microstructure and electrical properties of GO, which nevertheless is rarely studied. Here, multiscale GOs with different flake sizes and oxidation degrees were fabricated and incorporated into waterborne alkyd resin (AR). The physical and chemical structures of GO and AR/GO composites were characterized in detail. Multiscale GOs are successfully prepared, and the corrosion resistance of AR/GO coatings is measured by electrochemical workstation. Electrochemical experiments indicate that GOs with larger flake sizes have excellent barrier properties due to the shielding effect; GOs with appropriate oxidation degrees could effectively improve the dispersion of GO and avoid the conductive path of GO in the matrix, because oxidation degree of GO could influence the dispersion and electrical properties. The corrosion protection efficiency of AR/GO(GO: 120 µm, 1.5 wt%, sp2/sp3 = 2.61) is 98.14%, which is 2.26 times higher than AR. The multiscale effects of GO on the corrosion resistance property of AR coatings are quite general, thus providing guidelines for developing highly efficient corrosion resistant coatings for practical usage.







Similar content being viewed by others
References
N.G. Thompson, M. Yunovich, and D. Dunmire: Cost of corrosion and corrosion maintenance strategies: Corrosion reviews. Corros. Rev. 25, 247 (2007).
C.H. Lee, M. Kato, and A. Usuki: Preparation and properties of bio-based polycarbonate/clay nanocomposites. J. Mater. Chem. 21, 6844 (2011).
U.C. Nwaogu, C. Blawert, N. Scharnagl, W. Dietzel, and K.U. Kainer: Effects of organic acid pickling on the corrosion resistance of magnesium alloy AZ31 sheet. Corros. Sci. 52, 2143 (2010).
A. Usuki, N. Hasegawa, M. Kato, and S. Kobayashi: Polymer–Clay nanocomposites. Adv. Polym. Sci. 179, 1 (2004).
N. Mahato and M.H. Cho: Graphene integrated polyaniline nanostructured composite coating for protecting steels from corrosion: Synthesis, characterization, and protection mechanism of the coating material in acidic environment. Constr. Build. Mater. 115, 618 (2016).
C.H. Chang, T.C. Huang, C.W. Peng, T.C. Yeh, H.I. Lu, W.I. Hung, C.J. Weng, T.I. Yang, and J.M. Yeh: Novel anticorrosion coatings prepared from polyaniline/graphene composites. Carbon 50, 5044 (2012).
Y.H. Yu, Y.Y. Lin, C.H. Lin, C.C. Chan, and Y.C. Huang: High-performance polystyrene/graphene-based nanocomposites with excellent anti-corrosion properties. Polym. Chem. 5, 535 (2013).
K. Krishnamoorthy, K. Jeyasubramanian, M. Premanathan, G. Subbiah, H.S. Shin, and J.K. Sang: Graphene oxide nanopaint. Carbon 72, 328 (2014).
C. Chen, S. Qiu, M. Cui, S. Qin, G. Yan, H. Zhao, L. Wang, and Q. Xue: Achieving high performance corrosion and wear resistant epoxy coatings via incorporation of noncovalent functionalized graphene. Carbon 114, 356 (2017).
S. Ci, P. Cai, Z. Wen, and J. Li: Graphene-based electrode materials for microbial fuel cells. Sci. China Mater. 58, 496 (2015).
U. Mogera, N. Kurra, D. Radhakrishnan, C. Narayana, and G.U. Kulkarni: Low cost, rapid synthesis of graphene on Ni: An efficient barrier for corrosion and thermal oxidation. Carbon 78, 384 (2014).
D.X. He, Y. Qiu, L.L. Li, R. Zhao, and W.D. Xue: Large-scale solvent-thermal synthesis of graphene/magnetite/conductive oligomer ternary composites for microwave absorption. Sci. China Mater. 58, 566 (2015).
Y. Dong, Q. Liu, Q. Zhou, Y. Dong, Q. Liu, and Q. Zhou: Corrosion behavior of Cu during graphene growth by CVD. Corros. Sci. 89, 214 (2014).
V. Mišković-Stanković, I. Jevremović, I. Jung, and K.Y. Rhee: Electrochemical study of corrosion behavior of graphene coatings on copper and aluminum in a chloride solution. Carbon 75, 335 (2014).
S. Chen, L. Brown, M. Levendorf, W. Cai, S.Y. Ju, J. Edgeworth, X. Li, C.W. Magnuson, A. Velamakanni, and R.D. Piner: Oxidation resistance of graphene-coated Cu and Cu/Ni alloy. ACS Nano 5, 1321 (2011).
L. Kyhl, S.F. Nielsen, A.G. Čabo, A. Cassidy, J.A. Miwa, and L. Hornekær: Graphene as an anti-corrosion coating layer. Faraday Discuss. 180, 495 (2015).
R.K.S. Raman, P.C. Banerjee, D.E. Lobo, H. Gullapalli, M. Sumandasa, A. Kumar, L. Choudhary, R. Tkacz, P.M. Ajayan, and M. Majumder: Protecting copper from electrochemical degradation by graphene coating. Carbon 50, 4040 (2012).
D. Prasai, J.C. Tuberquia, R.R. Harl, G.K. Jennings, B.R. Rogers, and K.I. Bolotin: Graphene: Corrosion-inhibiting coating. ACS Nano 6, 1102 (2012).
S. Liu, L. Gu, H. Zhao, J. Chen, and H. Yu: Corrosion resistance of graphene-reinforced waterborne epoxy coatings. J. Mater. Sci. Technol. 32, 425 (2016).
B.P. Singh, S. Nayak, K.K. Nanda, B.K. Jena, S. Bhattacharjee, and L. Besra: The production of a corrosion resistant graphene reinforced composite coating on copper by electrophoretic deposition. Carbon 61, 47 (2013).
B. Ramezanzadeh, S. Niroumandrad, A. Ahmadi, M. Mahdavian, and M.H.M. Moghadam: Enhancement of barrier and corrosion protection performance of an epoxy coating through wet transfer of amino functionalized graphene oxide. Corros. Sci. 103, 283 (2016).
B. Ramezanzadeh, A. Ahmadi, and M. Mahdavian: Enhancement of the corrosion protection performance and cathodic delamination resistance of epoxy coating through treatment of steel substrate by a novel nanometric sol-gel based silane composite film filled with functionalized graphene oxide nanosheets. Corros. Sci. 109, 182 (2016).
W.L. Zhang, Y.D. Liu, and H.J. Choi: Fabrication of semiconducting graphene oxide/polyaniline composite particles and their electrorheological response under an applied electric field. Carbon 50, 290 (2012).
Y. Hayatgheib, B. Ramezanzadeh, P. Kardar, and M. Mahdavian: A comparative study on fabrication of a highly effective corrosion protective system based on graphene oxide-polyaniline nanofibers/epoxy composite. Corros. Sci. 133, 358 (2018).
L.C. Tang, Y.J. Wan, D. Yan, Y.B. Pei, L. Zhao, Y.B. Li, L.B. Wu, J.X. Jiang, and G.Q. Lai: The effect of graphene dispersion on the mechanical properties of graphene/epoxy composites. Carbon 60, 16 (2013).
K-C. Chang, M-H. Hsu, H-I. Lu, M-C. Lai, P-J. Liu, C-H. Hsu, W-F. Ji, T-L. Chuang, Y. Wei, and J-M. Yeh: Corrigendum to “Room-temperature cured hydrophobic epoxy/graphene composites as corrosion inhibitor for cold-rolled steel”. Carbon 82, 611 (2015).
J. Zhao, X. Xie, and C. Zhang: Effect of the graphene oxide additive on the corrosion resistance of the plasma electrolytic oxidation coating of the AZ31 magnesium alloy. Corros. Sci. 114, 146 (2016).
W.S. Hummers, Jr. and R.E. Offeman: Preparation of graphitic oxide. J. Am. Chem. Soc. 80, 1339 (1958).
J. Shen, Y. Hu, M. Shi, X. Lu, C. Qin, C. Li, and M. Ye: Fast and facile preparation of graphene oxide and reduced graphene oxide nanoplatelets. Chem. Mater. 21, 3514 (2009).
S. Stankovich, D.A. Dikin, R.D. Piner, K.A. Kohlhaas, A. Kleinhammes, Y. Jia, Y. Wu, S.B.T. Nguyen, and R.S. Ruoff: Synthesis of graphene-based nanosheets via chemical reduction of exfoliated graphite oxide. Carbon 45, 1558 (2007).
S. Shukla and S. Saxena: Spectroscopic investigation of confinement effects on optical properties of graphene oxide. Appl. Phys. Lett. 98, 183 (2011).
N. Liaros, J. Tucek, K. Dimos, A. Bakandritsos, K.S. Andrikopoulos, D. Gournis, R. Zboril, and S. Couris: The effect of the degree of oxidation on broadband nonlinear absorption and ferromagnetic ordering in graphene oxide. Nanoscale 8, 2908 (2016).
C. Hontoria-Lucas, A.J. López-Peinado, J.D.D. López-González, M.L. Rojas-Cervantes, and R.M. Martín-Aranda: Study of oxygen-containing groups in a series of graphite oxides: Physical and chemical characterization. Carbon 33, 1585 (1995).
G. Venugopal, K. Krishnamoorthy, R. Mohan, and S.J. Kim: An investigation of the electrical transport properties of graphene-oxide thin films. Mater. Chem. Phys. 132, 29 (2011).
T. Bellezze and R. Fratesi: Assessing the efficiency of galvanic cathodic protection inside domestic boilers by means of local probes. Corros. Sci. 52, 3023 (2010).
K. Krishnamoorthy, M. Veerapandian, K. Yun, and S.J. Kim: The chemical and structural analysis of graphene oxide with different degrees of oxidation. Carbon 53, 38 (2013).
W. Sun, L. Wang, T. Wu, Y. Pan, and G. Liu: Synthesis of low-electrical-conductivity graphene/pernigraniline composites and their application in corrosion protection. Carbon 79, 605 (2014).
Z. Chao, H. Shu, W.T. Weng, F. Wei, and T. Liu: Facile preparation of water-dispersible graphene sheets stabilized by acid-treated multi-walled carbon nanotubes and their poly(vinyl alcohol) composites. J. Mater. Chem. 22, 2427 (2012).
K.C. Chang, W.F. Ji, C.W. Li, C.H. Chang, Y.Y. Peng, J.M. Yeh, and W.R. Liu: The effect of varying carboxylic-group content in reduced graphene oxides on the anticorrosive properties of PMMA/reduced graphene oxide composites. Express Polym. Lett. 8, 908 (2014).
J.M. Yeh, K.C. Chang, H.I. Lu, C.H. Chang, C.H. Hsu, W.F. Ji, W.Y. Li, T.L. Chuang, W.R. Liu, and M.H. Tsai: Advanced anticorrosive coatings prepared from electroactive polyimide/graphene nanocomposites with synergistic effects of redox catalytic capability and gas barrier properties. Express Polym. Lett. 8, 243 (2014).
X. Sheng, W. Cai, L. Zhong, D. Xie, and X. Zhang: Synthesis of functionalized graphene/polyaniline nanocomposites with effective synergistic reinforcement on anticorrosion. Ind. Eng. Chem. Res. 55, 8576 (2016).
O.U. Rahman, M. Kashif, and S. Ahmad: Nanoferrite dispersed waterborne epoxy-acrylate: Anticorrosive nanocomposite coatings. Prog. Org. Coat. 80, 77 (2015).
Acknowledgment
The authors would like to acknowledge the financial support by the National Natural Science Foundation of China (Grant No. 51303116).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Li, J., Zhang, H., Sun, F. et al. The multiscale effects of graphene oxide on the corrosion resistance properties of waterborne alkyd resin coatings. Journal of Materials Research 34, 950–958 (2019). https://doi.org/10.1557/jmr.2018.486
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2018.486