Abstract
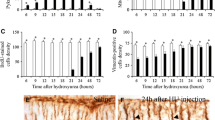
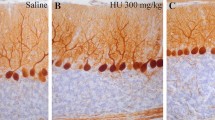
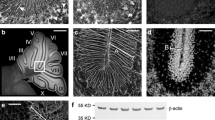
Postnatal development of the cerebellum lasts for weeks in rodents and can be disturbed by systemic 5-bromo-2′-deoxyuridine (BrdU) administration. This thymidine analogue incorporates into the DNA of proliferating cells, and results in more or less serious damage or death of granule cells, the most actively dividing neuronal population in the developing cerebellar cortex. Further consequences of postnatal BrdU administration are the interrupted postnatal migration and integration as well as partial loss of cerebellar Purkinje cells. In the present study, C57B16 mice were administered with 50 μ.g/g body weight BrdU, one sc. injection daily, between P0 and Pll postnatal days, respectively. Large “cavities” appeared in the cytoplasm of a subpopulation of Purkinje cells by P7 in about one-third of administered animals, their number were high at P9 and Pll, especially in vermal lobules VIII, IX, and X. By P13 and P15 the number and size of the cavities (and PCs exhibiting unusual morphology) decreased. EM studies revealed that the unusual Purkinje cells received numerous axonal inputs of unknown origin, first of all on their somatic and dendritic spines. The transitory appearance of a subpopulation of Purkinje cells possessing unusual morphology refers to the influence of other (neuronal, glial, or both) cells on their regular differentiation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Altman, J. Anderson, W. J. (1972) Experimental reorganization of the cerebellar cortex. 1. Morphological effects of elimination of all microneurons with prolonged X-irradiation started at birth. J. Comp. Neurol. 146, 355–306.
Altman, J., Bayer, S. A. (1997) Development of the cerebellar system in relation to its evolution, structure and functions. CRC Press, Boca Raton.
Bannigan, J., Langman, J. (1979) The cellular effect of 5-bromodeoxyuridine on the mammalian embryo. J. Embryol. Exp. Morphol. 50, 123–135.
Bonthius, D.J., Bonthius, N.E., Napper, R.M., Astley, S. J, Clarren, S.K., West, J. R. (1996) Purkinje cell deficits in nonhuman primates following weekly exposure to ethanol during gestation. Teratolog. 53, 230–236.
Caddy, K. W. T., Biscoe, T. J. (1979) Structural and quantitative studies on the normal C3H and Lurcher mutant mouse. Philos. Trans. R. Soe. Lond.. 287, 167–201.
De Barry, J., Gombos, G., Klupp, T., Hamori, J. (1987) Alterations of mouse cerebellar circuits following methylazoxymethanol treatment during development: Immunocytochemical and electron microscopic study. J. Comp. Neurol. 61, 253–265.
De Camilli, P., Miller, P.E., Levitt, P., Walter, U., Greengard, P. (1984) Anatomy of cerebellar Purkinje cells in the rat determined by a specific immunohistochemical marker. Neuroscienc. 11, 761–817.
del Rio, J.A., Soriano, E. (1989) Immunocytochemical detection of 5-bromodeoxyuridine incorporation in the central nervous system of the mouse. BrainRes. 49, 311–317.
Dikraniana, K., Qina, Y. O., Labruyerea, J., Nemmersa, B., Olneya, J. W. (2005) Ethanol-induced neuroapoptosis in the developing rodent cerebellum and related brain stem structures. Dev. BrainRes. 155, 1–13.
Dolbeare, F. (1995) Bromodeoxyuridine: a diagnostic tool in biology and medicine. Part I. Historical perspectives, histochemical methods and cell kinetics. Histochem. J. 27, 339–369.
Dolbeare, F. (1996) Bromodeoxyuridine: a diagnostic tool in biology and medicine. Part III. Proliferation in normal, injured and diseased tissue, growth factors, differentiation, DNA replication sites and in situ hybridization. Histochem. J. 28, 531–575.
Fonnum, F., Lock, E. A. (2000) Cerebellum as a target for toxic substances. Toxicology Letters 112-113, 9–16.
Garcia, R.I., Werner, I., Szabo, G. (1979) Effect of 5-bromo-2’-deoxyuridine on growth and differentiation of cultured embryonic retinal pigment cells. In Vitr. 15, 779–788.
Gores, T.J., Penke, B., Boti, Z., Katarova, Z., Hámori, J. (1993) Immunohistochemical visualization of a metabotropic glutamate receptor. NeuroRepor. 4, 283–286.
Grandes, P., Mateos, J.M., Ruegg, D., Kuhn, R., Knõpfel, T. (1994) Different cellular localization of three splice variants of the mGluRl metabotropic glutamate receptor in rat cerebellum. NeuroRepor. 5, 2249–2252.
Gratzner, H. G. (1982) Monoclonal antibody to 5-bromo- and 5-iododeoxyuridine: a new reagent for detection of DNA replication. Science 218, A1A–A15.
Grimaldi, P., Rossi, F. (2006) Lack of neurogenesis in the adult rat cerebellum after Purkinje cell degeneration and growth factor infusion. Eur. J. Neurosci. 23, 2657–2668.
Hámori, J., Takács, J., Gores, T. J. (1996) Immunocytochemical localization of mGluRl a metabotropic glutamate receptor in inhibitory neurons of the cerebellar cortex. Acta Biol. Hung. 47, 181–194.
Kiss, J., Gores, T.J., Kuhn, R., Knõpfel, T., Csáky, A., Halász, B. (1996) Distribution of metabotropic glutamate receptor 1 a in the rat hypothalamus: an immunocytochemical study using monoclonal and polyclonal antibody. Acta Biol. Hung. 47, 221–237.
Hatten, M.E., Heintz, N. (1995) Mechanisms of neural patterning and specification in the developing cerebellum. Annu. Rev. Neurosci. 18, 385 108
Heckroth, J.A., Goldowitz, D., Eisenman, L. M. (1989) Purkinje cell reduction in the reeler mutant mouse: a quantitative immunohistochemical study. J. Comp. Neurol. 279, 546–555.
Herrup, K., Trenkner, E. (1987) Regional differences in cytoarchitecture of the weaver cerebellum suggest a new model for weaver gene action. Neuroscienc. 23, 871–885.
Hillman, D.E., Chen, S. (1981) Vulnerability of cerebellar development in malnutrition-I. Quantitation of layer volume and neuron numbers. Neuroscienc. 6, 1249–1262.
Hirano, A., Dembitzer, H.M., Jones, J. (1972) An electron microscopic study of cycasin-induced cerebellar alterations. J. Neuropathol. Exp. Neurol. 31, 113–125.
Jande, S.S., Maler, L., Lawson, D. E. M. (1981) Immunohistochemical mapping of vitamin-d-dependent calcium-binding protein in brain. Natur. 294, 765–767’.
Kolb, B., Pedersen, B., Ballermann, M., Gibb, R., Whishaw, I. Q. (1999) Embryonic and postnatal injections of bromodeoxyuridine produce age-dependent morphological and behavioral abnormalities. J. Neurosci. 19, 2337–2346.
Kwok, K.H., Law, K.B., Wong, R.N., Yung, K. K. (1999) Immunolesioning of nerve growth factor p75 receptor-containing neurons in the rat brain by a novel immunotoxin: anti-p75-anti-mouse IgG-trichosanthin conjugates. Brain. Res. 846, 154–163.
Light, K.E., Blecher, S.M., Peirce, D. R. (2002) Time course and manner of Purkinje neuron death following a single ethanol exposure on postnatal day 4 in the developing rat. Neuroscienc. 114, 327.
Martin, L.J., Blackstone, C.D., Huganir, R.L., Price, D. L. (1992) Cellular localization of a metabotropic glutamate receptor in rat brain. Neuro. 9, 259–270.
Miale, I.L., Sidman, R. L. (1961) An autoradiographic analysis of histogenesis in the mouse cerebellum. Exp. Neurol. 4, 277–296.
Moller-Madsen, B., Danscher, G. (1986) Localization of mercury in CNS of the rat. I. Mercuric chloride (HgCl2) per os. Environ. Res. 41, 29–33.
Nagao, T., Kuwagata, M., Saito, Y. (1998) Effects of prenatal exposure to 5-bromo-2’-deoxyuridine on the developing brain and reproductive function in male mouse offspring. Reprod. Toxicol. 12
Ozol, K., Hayden, J.M., Oberdick, J., Hawkes, R. (1999) Transverse zones in the vermis of the mouse cerebellum. J. Comp. Neurol. 412, 95–111.
Ozol, K., Hawkes, R. (1997) Calbindin organization in the newborn mouse cerebellum: genealogical analysis of Purkinje cell compartments. Soc. Neurosci. Abstr. 23, 19.
Sekerkova, G., Ilijic, E., Mugnaini, E. (2004) Bromodeoxyuridine administered during neurogenesis of the projection neurons causes cerebellar defects in rat. J. Comp. Neurol. 470, 221–239.
Sotelo, C., Rio, J. P. (1988) Cerebellar malformation obtained in rats by early postnatal treatment with 6-aminonicotinamide. Role of neuron-glia interactions in cerebellar development. Neuroscienc. 5, 1737–1759.
Takács, J., Gombos, G., Gores, T., Becker, T., de Barry, J., Hámori, J. (1977) Distribution of metabotropic glutamate receptor type la in the Purkinje cell dendritic spines is independent of the presence of presynaptic parallel fibers. J. Neurosci. Res. 50, 433–442.
Takács, J., Hámori, J., Gores, T. J. (1994) MGluRla immunoreactivity in Purkinje cells of the developing rat cerebellar cortex. Eur. J. Neurosci. Suppl. 7, 51.
Takacs, J., Borostyankoi, Z.A., Veisenberger, E., Vastagh, C., Vig, J., Gores, T.J., Hamori, J. (2000) Postnatal development of unipolar brush cells in the cerebellar cortex of cat. J. Neurosci. Res. 61, 107–115.
Takacs, J., Vig, J., Vastagh, Cs., Hamori, J. (2003) Effect of 5-bromo-2’-deoxyuridine (BrdU) on the postnatal development of cerebellum. In: Sixth IBRO World Congress of Neuroscience, July 10–15, Prague, Abstracts, p. 144.
Takacs, J., Vig, J., Hamori, J. (2006) Unusual morphology of Purkinje cells following postnatal BrdU administration in the cerebellar cortex of developing mouse. Clinical Neuroscience. Supplemen. 1, pp. 64.
Takács, J., Vig, J., Hámori, J. (2006) Vascular damage following postnatal BrdU administration in the cerebellar cortex of mouse. FENS 5th Forum, Vienna, July 8-12, 2006. Abstract book, pp. 229.
Taupin, P. (2007) BrdU immunohistochemistry for studying adult neurogenesis: paradigms, pitfalls, limitations, and validation. Brain Res. Rev. 53, 198–214.
Vig, J., Takács, J., Vastagh, C., Baldauf Z., Veisenberger, E., Hámori, J. (2003) Distribution of mGluRla and SMI 311 immunoreactive Lugaro cells in the kitten cerebellum. J. Neurocytol. 32, 217–227.
Wassef M., Zanetta, J.P., Brehier, A., Sotelo, C. (1985) Transient biochemical compartmentalization of Purkinje-cells during early cerebellar development. Dev. Biol. Ill, 129-137.
Wassef, M., Angaut, P., Arsenio-Nunes, L., Bourrat, F., Sotelo, C. (1992) Purkinje cell heterogenity: its role in organizing the topography of the cerebellar cortex connections. In: Llinas, R., Sotelo, C. (eds) The cerebellum revisited Springer-Verlag, New York-Heidelberg-Berlin, pp. 5–21.
Wright, D.D., Blackstone, C.D., Huganir, R.L., Ryugo, D. K. (1996) Immunocytochemical localization of the mGluRla metabotropic glutamate receptor in the dorsal cochlear nucleus. J. Comp. Neurol. 364, 729–745.
Yu, W. H. A. (1975) The effect of 5-bromodeoxyurudine on the postnatal development of the rat cerebellum. Anat. Rec. 181, 516–517.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Professor József Hámori on the occasion of his 80th birthday
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Takács, J. Unusual Morphological Damage of Purkinje Cells Following Postnatal BrdU Administration in the Cerebellar Cortex of Mouse. BIOLOGIA FUTURA 63 (Suppl 1), 19–37 (2012). https://doi.org/10.1556/ABiol.63.2012.Suppl.1.4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/ABiol.63.2012.Suppl.1.4