Abstract
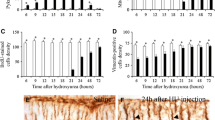
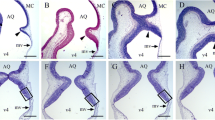
The current paper analyzes the development of the male and female rat cerebellum exposed to hydroxyurea (HU) (300 or 600 mg/kg) as embryo and collected at postnatal day 90. Our study reveals that the administration of this drug compromises neither the cytoarchitecture of the cerebellar cortex nor deep nuclei (DCN). However, in comparison with the saline group, we observed that several cerebellar parameters were lower in the HU injected groups. These parameters included area of the cerebellum, cerebellar cortex length, molecular layer area, Purkinje cell number, granule cell counts, internal granular layer, white matter and cerebellar nuclei areas, and number of deep cerebellar nuclei neurons. These features were larger in the rats injected with saline, smaller in those exposed to 300 mg/kg of HU and smallest in the group receiving 600 mg/kg of this agent. No sex differences in the effect of the HU were observed. In addition, we infer the neurogenetic timetables and the neurogenetic gradients of PCs and DCN neurons in rats exposed to either saline or HU as embryos. For this purpose, 5-bromo-2′-deoxyuridine was injected into pregnant rats previously administered with saline or HU. This thymidine analog was administered following a progressively delayed cumulative labeling method. The data presented here show that systematic differences exist in the pattern of neurogenesis and in the spatial location of cerebellar neurons between rats injected with saline or HU. No sex differences in the effect of the HU were observed. These findings have implications for the administration of this compound to women in gestation as the effects of HU on the development of the cerebellum might persist throughout their offsprings’ life.





Similar content being viewed by others
References
Ahmed OM, Abd El-Tawab SM, Ahmed RG (2010) Effects of experimentally induced maternal hypothyroidism and hyperthyroidism on the development of rat offspring: I. The development of the thyroid hormones-neurotransmitters and adenosinergic system interactions. Int J Dev Neurosci 28:437–454
Altman J, Bayer SA (1997) Development of the cerebellar system: in relation to its evolution, structure and functions. CRC Press, Boca Raton
Banh S, Hales BF (2013) Hydroxyurea exposure triggers tissue-specific activation of p38 mitogen-activated protein kinase signaling and the DNA damage response in organogenesis-stage mouse embryos. Toxicol Sci 133:298–308
Bayer SA, Altman J (1987) Directions in neurogenetic gradients and patterns of anatomical connections in the telencephalon. Prog Neurobiol 29:57–106
Bayer SA, Altman J (1995) Neurogenesis and neuronal migration. In: Patxinos G (ed) The rat nervous system. Academic Press, San Diego, pp 1041–1078
Bayer SA, Altman J, Russo RJ, Zhang X (1993) Timetables of neurogenesis in the human brain based on experimentally determined patterns in the rat. Neurotoxicology 14(1):83–144
Bottone MG, Veronica DB, Piccolini VM, Bottiroli G, De Pascali SA, Fanizzi FP, Bernocchi G (2012) Developmental expression of cellular prion protein and apoptotic molecules in the rat cerebellum: effects of platinum compounds. J Chem Neuroanat 46:19–29
Butts T, Green MJ, Wingate RJT (2014) Development of the cerebellum: simple steps to make a “little brain”. Development 141:4031–4041
Byrd DC, Pitts SR, Alexandre CK (1999) Hydroxyurea in two pregnant women with sickle cell anemia. J Hum Pharmacol Drug Ther 19(12):1459–1462
Campion SN, Davenport SJ, Nowland WS, Cappon GD, Bowman CJ, Hurtt ME (2012) Sensitive windows of skeletal development in rabbits determined by hydroxyurea exposure at different times throughout gestation. Birth Defects Res B 95(3):238–249
DeSesso JM (1979) Cell death and free radicals: a mechanism for hydroxyurea teratogenesis. Med Hypotheses 5:937–951
Diav-Citrin O, Hunnisett L, Sher GD, Koren G (1999) Hydroxyurea use during pregnancy: a case report in sickle cell disease and review of the literature. Am J Hematol 60:148–150
Ebels EJ, Peters I, Thijs A (1975) Studies on ectopic granule cells in the cerebellar cortex. III. An investigation into the restoration of the external granular layer after partial destruction. Acta Neuropathol 31:103–107
Hampson DR, Blatt GJ (2015) Autism spectrum disorders and neuropathology of the cerebellum. Front Neurosci 6(9):420. doi:10.3389/fnins.2015.00420
Hervás JP, Martí-Clúa J, Muñoz-García A, Santa-Cruz MC (2002) Proliferative activity in the cerebellar external granular layer evaluated by bromodeoxyuridine labeling. Biotech Histochem 77:27–35
Jacobs B, Johnson NL, Wahl D, Schall M, Maseko BC, Lewandowski A, Raqhanti MA, Wicinski B, Butti C, Hopins WD, Bertelsen MF, Walsh T, Roberts JR, Reep RL, Hof PR, Sherwood CC, Manger PR (2014) Comparative neuronal morphology of the cerebellar cortex in afrotherians, carnivores, cetartiodactyls, and primates. Front Neuroanat 8:24. doi:10.3389/fnana.2014.00024.eCollection2014
Kita Y, Kawakami K, Takahashi Y, Murakami F (2013) Development of cerebellar neurons and glias revealed by in utero electroporation: golgi-like labeling of cerebellar neurons and glias. PLoS ONE 8(7):e70091. doi:10.1371/journal.pone.0070091
Koppel H, Lewis PD, Padel AJ (1983) Cell death in the external granular layer of normal and undernourished rats: further observations, including estimates of rate of cell loss. Cell Tissue Kinet 16:99–106
Lalonde R, Strazielle C (2007) Spontaneous and induced mouse mutations with cerebellar dysfunctions: behavior and neurochemistry. Brain Res 1140:51–74
Lebwohl M, Menter A, Koo J, Feldman SR (2004) Combination therapy to treat moderate to severe psoriasis. J Am Acad Dermatol 50(3):416–430
Leto K, Rolando C, Rossi F (2012) The genesis of cerebellar GABAergic neurons: fate potential and specification mechanisms. Front Neuroanat 20(6):6. doi:10.3389/fnana.2012.00006.eCollection
Manto M (2012) Toxic agents causing cerebellar ataxias. Handb Clin Neurol 103:201–213
Martí J, Wills KV, Ghetti B, Bayer SA (2002) Regional differences in the Purkinje cells settled pattern: a comparative autoradiographic study in control and homozygous weaver mice. Exp Neurol 175:168–181
Martí J, Santa-Cruz MC, Bayer SA, Ghetti B, Hervás JP (2007) Purkinje cell age-distribution in fissures and in foliar crowns: a comparative study in the weaver cerebellum. Brain Struct Funct 212:347–357
Martí J, Santa-Cruz MC, Serra R, Molina O, Hervás JP, Villegas S (2013) Principal component and cluster analysis of morphological variables reveals multiple discrete sub-phenotypes in weaver mouse mutants. Cerebellum 12:406–417
Martí J, Santa-Cruz MC, Serra R, Hervás JP (2015) Systematic differences in time of cerebellar-neuron origin derived from bromodeoxyuridine immunoperoxidase staining protocols and tritiated thymidine autoradiographic: a comparative study. Int J Dev Neurosci 47:216–228
Martinez S, Andreu A, Mecklenburg N, Echevarria D (2013) Cellular and molecular basis of cerebellar development. Front Neuroanat. doi:10.3389/fnana.2013.00018.eCollection2013
Marzban H, Del Bigio MR, Alizadeh J, Ghavami S, Zachariah RM, Rastegar M (2015) Cellular commitment in the developing cerebellum. Front Cell Neurosci 12(8):450. doi:10.3389/fncel.2014.00450.eCollection
Navarra P, Preziosi P (1999) Hydroxyurea: new insights on an old drug. Crit Rev Oncol Hematol 29:249–255
Newton HB (2007) Hydroxyurea chemotherapy in the treatment of meningiomas. Neurosurg Focus 23(4):E11
Oster-Granite ML, Herndon RM (1976) The pathogenesis of parvovirus-induced cerebellar hypoplasia in the Syrian hamster, Mesocricetus auratus. Fluorescent antibody, foliation, cytoarchitecture, Golgi and electron microscopic studies. J Comp Neurol 169:481–521
Patxinos G, Watson C (1998) The rat brain in stereotaxic coordinates, 4th edn. Academic Press, San Diego
Phillips JR, Hewedi DH, Eissa AM, Moustafa AA (2015) The cerebellum and psychiatric disorders. Front Public Health 3:66. doi:10.3389/fpubh.2015.00066.eCollection2015
Preziosi P, Parente L, Navarra P (1992) Cytokines and eicosanoids in cancer drug toxicity. Trends Pharmacol Sci 13:226–229
Saban N, Bujak M (2009) Hydroxyurea and hydroxamic acid derivatives as antitumor drugs. Cancer Chemother Pharmacol 64:213–221
Sampson M, Archibong AE, Powell A, Strange B, Roberson S, Hills ER, Bourne P (2010) Perturbation of the developmental potential of preimplantation mouse embryos by hydroxyurea. Int J Environ Res Public Health 7:2033–2044
Schlisser AE, Hales BF (2013) Deprenyl enhances the teratogenicity of hydroxyurea in organogenesis stage mouse embryos. Toxicol Sci 134:391–399
Sekerkova G, Ilijic E, Mugnaini E (2004) Time of origin of unipolar brush cells in the rat cerebellum as observed by prenatal bromodeoxyuridine labeling. Neuroscience 127:845–858
Shakiba A (2014) The role of the cerebellum in neurobiology of psychiatric disorders. Neurol Clin 32:1105–1115
Shao J, Zhou B, Chu B, Yen Y (2006) Ribonucleotide reductase inhibitors and future drug design. Curr Cancer Drug Targets 6:409–431
Sillitoe RV, Joyner AL (2007) Morphology, molecular codes, and circuitry produce the three-dimensional complexity of the cerebellum. Annu Rev Cell Dev Biol 23:549–577
Sotelo C, Rossi F (2013) Purkinje cell migration and differentiation. In: Manto M, Gruol DL, Schmahmann J, Koibuchi N, Rossi F (eds) Handbook of the cerebellum and cerebellar disorders. Springer, The Netherlands, pp 147–178
Stoodley CJ (2016) The cerebellum and neurodevelopmental disorders. Cerebellum 15(1):34–37
Sultan F, Glickstein M (2007) The cerebellum: comparative and animal studies. Cerebellum 6:168–176
Thauvin-Robinet C, Maingueneau C, Robert E, Elefant E, Guy H, Caillot D, Casasnovas RO, Douvier S, Nivelon-Chevallier A (2001) Exposure to hydroxyurea during pregnancy: a case series. Leukemia 15(8):1309–1311
Ware RE, Aygun B (2009) Advances in the use of hydroxyurea. Am Soc Hematol Educ Progr. doi:10.1182/asheducation-2009.1.62
Ware RE, Despotovic JM, Mortier NA, Flanagan JM, He J, Smeltzer MP, Kimble AC, Aygun B, Wu S, Howard T, Sparreboom A (2011) Pharmacokinetics, pharmacodynamics, and pharmacogenetics of hydroxyurea treatment for children with sickle cell anemia. Blood 118:4985–4991
Wong TE, Brandow AM, Lim W, Lottenberg R (2014) Update on the use of hydroxyurea therapy in sickle disease. Blood 124(26):3850–3857
Woo GH, Katayama K, Jung JY, Uetsuka K, Bak EJ, Nakayama H, Doi K (2003) Hydroxyurea (HU)-induced apoptosis in the mouse fetal tissues. Histol Histopathol 18:387–392
Woo GH, Katayama K, Bak EJ, Ueno H, Tamauchi H, Uetsuka K, Nakayama H, Doi K (2004) Effects of prenatal hydroxyurea-treatment on mouse offspring. Exp Toxicol Pathol 56(1-2):1–7
Woo GH, Bak EJ, Nakayama H, Doi K (2005) Hydroxyurea (HU)-induced apoptosis in the mouse fetal lung. Exp Mol Pathology 79:59–67
Woo GH, Bak EJ, Katayama K, Doi K (2006) Molecular mechanisms of hydroxyurea (HU)-induced apoptosis in the mouse fetal brain. Neurotocol Teratol 28:125–134
Wullimann MF, Mueller T, Distel M, Babaryka A, Grothe B, Köster RW (2011) The long adventurous journey of rhombic lip in jawed vertebrates: a comparative developmental analysis. Front Neuroanat 21(5):27. doi:10.3389/fnana.2011.00027.eCollection
Zala C, Rouleau D, Montaner JS (2000) Role of hydroxyurea in treatment of disease due to human immunodeficiency virus infection. Clin Infect Dis 30:S143–S150
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Martí, J., Santa-Cruz, M.C., Serra, R. et al. Hydroxyurea Treatment and Development of the Rat Cerebellum: Effects on the Neurogenetic Profiles and Settled Patterns of Purkinje Cells and Deep Cerebellar Nuclei Neurons. Neurotox Res 30, 563–580 (2016). https://doi.org/10.1007/s12640-016-9649-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-016-9649-x