Abstract
The effect of Cd2+, as one of the most widespread toxic environmental pollutants, was studied on γ-aminobutyric acid (GABA) evoked responses of identified neurons in the central nervous system of the pond snail. Lymnaea stagnalis L. (Gastropoda). In the experiments, the modulation of the action of GABA both on neuronal activity (current clamp recording) and on the a GABA activated membrane Cl−-current (voltage clamp studies) has been shown. It was found that:
-
1.
GABA could evoked three different various types of response in GABA sensitive neurons: i) hyperpolarization with strong inhibition of ongoing spike activity, ii) short depolarization with an increase of spike the activity, iii) biphasic respone with a short excitation followed by a more prolonged long inhibition.
-
2.
In low-Cl− solution the inhibitory action of GABA was reduced or eliminated, but the excitatory one was not or only moderately affected.
-
3.
CdCl2 inhibited the GABA evoked hyperpolarization, but left intact or only slightly reduced the excitation evoked by GABA.
-
4.
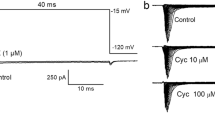
The inward Cl--current evoked by GABA at a −75 mV holding potential was slightly augmented in the presence of 1 μmol/l Cd2+, but was reduced or blocked at higher cadmium concentrations. The effect of Cd2+ was concentration and time dependent.
-
5.
Parallel with reducing the GABA evoked current, cadmium increased both the time to peak and the half inactivation time of the current.
-
6.
CdCl2 alone, in 50 μmol/l concentration, induced a 1–2 nA inward current.
The blocking effect of cadmium on GABA activated inhibitory processes can be an important component of the neurotoxic effects of this heavy metal ion.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Akopian, A., Gabriel, R., Witkovsky, P. (1998) Calcium released from intracellular stores inhibits GABAA-mediated currents in ganglion cells of the turtle retina. J. Neurophysiol. 80, 1105–1115.
Arakawa, O., Nakahiro, M, Narahashi, T. (1991) Mercury modulation of GABA-activated chloride channels and non-specific cation channels in rat dorsal root ganglion neurons. Brain Res. 551, 58–63.
Bokisch, A. J., Walker, R. J. (1986) The ionic mechanism associated with the action of putative transmitters on identified neurons of the snail. Helix aspersa. Comp. Biochem. Physiol. 84C, 231–241.
Büsselberg, D., Michael, D., Platt, B. (1994) Pb2+ reduces voltage- and N-methyl-D-aspartate (NMDA)-activated calcium channel currents. Cell. Mol. Neurobiol. 14, 711–722.
Carpenter, D. O. (1994) The public health significance of metal neurotoxicity. Cell. Mol. Neurobiol. 14, 591–597.
Chang, L. W. (ed.). Toxicology of Metals. CRC Press, Inc., Lewis Publisheres, New York, USA, 1996.
Darlison, M. G., Hutton, M. L., Harvey, R. J. (1993) Molluscan ligand-gated ion-channel receptors. In: Pichon, Y. (ed.). Comparative Molecular Neurobiology. Birkhauser Verlag, Basel, Switzerland, pp. 48–64.
Enz, R., Ross, B. J., Cutting, G. R. (1999) Expression of the voltage-gated chloride channel ClC-2 in rod bipolar cells of the rat retina. J. Neurosci. 19, 9841–9847.
Gupta, A., Wang, Y., Macram, H. (2000) Organizing principles for a diversity of GABAergic inter-neurons and synapses in the neocortex. Science 287, 273–278.
Győri, J., Kiss, T., Shcherbatko, A. D., Belan, P. V., Tepikin, A. V., Osipenko, O. N., Salánki, J. (1991) Effect of Ag+ on membrane permeability of perfuse. Helix pomatia neurons. J. Physiol. (London) 442, 1–13.
Győri, J., Fejtl, M., Carpenter, D. O., Salánki, J. (1994) Effect of HgCl2 on acetylcholine, carbachol, and glutamate currents o. Aplysia neurons. Cell. Mol. Neurobiol. 14, 653–664.
Herlenius, E., Lagercrantz, H. (2001) Neurotransmitter and neuromodulators during early human development. Early Hum. Dev. 65, 21–37.
Hille, B. (1975) The receptor for tetrodotoxin and saxitoxin. A structural hypothesis. Biophys. J. 15, 615–619.
Jungwirth A., Paulmichl, M., Lang F. (1990) Cadmium enhances potassium conductance in cultured renal epitheloid (MDCK) cells. Kidney International 37, 1477–1486.
Kajita, H., Omori, K., Matsuda, H. (2000) The chloride channel ClC-2 contributes to the inwardly rectifying Cl- conductance in cultured porcine choroid plexus epithelial cells. J. Physiol (London) 523, 313–324.
Kaneko, A., Tachibana, M. (1986) Blocking effect of cobalt and related ions on the gamma-aminobu-tyric acid-induced current in turtle retinal cones. J. Physiol. (London) 373, 463–479.
Kiss, T., Osipenko, O. N. (1994) Toxic effects of heavy metals on ionic channels. Pharmacol. Rev. 46, 245–267.
Leake, L., Walker, R. J. (1980). Invertebrate Neuropharmacology, Blackie, Glasgow and London, U.K.
Ma, J. Y., Narahashi, T. (1993) Differential modulation of GABAA receptor-channel complex by polyvalent cations in rat dorsal root ganglion neurons. Brain Res. 607, 222–232.
Miledi, R., Parker, I., Woodward, R. M. (1989) Membrane currents elicited by divalent cations i. Xenopus oocytes. J. Physiol. (London) 417, 173–195.
Narahashi T., Ma, J. Y., Arakawa, O., Reuveny, E., Nakahiro, M. (1994) GABA receptor-channel complex as a target site of mercury, copper, zink, and lanthanides. Cell. Mol. Neurobiol. 14, 599–621.
Nogawa, K., Kido, T. (1966) Itai-itai disease and health effects of cadmium. In: Chang, L. W. (ed.). Toxicology of Metals. Lewis Publishers, New York, USA, pp. 353–369.
Oortgiesen, M., van Kleef, R. G. D. M., Bajnath, R. B., Vijverberg, H. P. M. (1990) Novel type of ion channel activated by Pb2+, Cd2+ and Al3+ in cultured mouse neuroblastoma cells. J. Membr. Biol. 113, 261–268.
Ropert, N., Guy, N. (1991) Serotonin facilitates GABAergic transmission in the CA1 region of rat hippocampu. in vitro. J. Physiol. (London) 441, 121–136.
Rubakhin, S. S., Győri, J., Carpenter, D. O., Salánki, J. (1995) HgCl2 potentiates GABA activated currents i. Lymnaea stagnalis neurones. Acta Biol. Hung. 46, 431–444.
Rubakhin, S. S., Szűcs, A., S.-Rózsa, K. (1996) Characterization of the GABA response on identified dialyse. Lymnaea neurons. Gen. Pharmac. 27, 731–739.
Salánki, J. (1988) Invertebrates in neuroscience. In: Salánki, J., S.-Rózsa, K. (eds.). Neurobiology of Invertebrates. Transmitters, Modulators and Receptors. Akadémiai Kiadó, Budapest, Hungary, pp. 1–10.
Salánki, J., Győri, J., Carpenter, D. O. (1994) Action of lead on glutamate activated chloride currents i. Helix pomatia L. neurons. Cell. Mol. Neurobiol. 14, 755–768.
S.-Rózsa, K. (2000) Modulation of firing pattern and oscillation in nerve cells o. Lymnaea during network reconstruction. Acta Biol. Hung. 51, 211–230.
S.-Rózsa, K., Salánki, J. (1985) Effects of heavy metals on the chemosensitivity of neuronal somata o. Lymnaea stagnalis L. In: Salánki, J. (ed.). Heavy Metals in Water Organisms. Akadémiai Kiadó, Budapest, Hungary, pp. 387–400.
S.-Rózsa, K., Rubakhin, S. S., Szűcs, A., Stefano, G. B. (1996) Met-enkephalin and morphiceptin modulate a GABA-induced inward current in the CNS o. Lymnaea stagnalis L. Gen. Pharmac. 27, 1337–1345.
S.-Rózsa, K., Salánki, J. (1987) Excitable membranes k]object for evaluating the effect of heavy metal pollution. Acta Biol. Hung. 38, 31–45.
S.-Rózsa, K., Salánki, J., Présing, M. (1988) Use o. Lymnaea stagnalis in monitoring heavy metal pollution. In: Yasuno, M., Whitton, B. A. (eds.). Biological Monitoring of Environmental Pollution. Tokai University Press, Tokyo, Japan, pp. 247–255.
Walker, R. J., Brooks, H. L., Holden-Dye, L. (1996) Evolution and overview of classical transmitter molecules and their receptors. Parasitology 113, S3–S33.
Weinreich, D., Wonderlin, W. F. (1987) Copper activates a unique inward current in molluscan neurones. J. Physiol. (London) 394, 429–443.
Winlow, W., Benjamin, P. R. (1976) Neuronal mapping of the brain of the pond snail. Lymnaea stag-nalis L. In: Salánki, J. (ed.). Neurobiology of Invetebrates. Gastropoda Brain. Akadémiai Kiadó, Budapest, Hungary, pp. 41–59.
Yarowsky, P. J., Carpenter, D. O. (1978) Receptors for gamma-aminobutyric acid (GABA) o. Aplysia neurons. Brain Res. 144, 75–94.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Professor József Hámori on the occasion of his 70th birthday.
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Molnár, G., Győri, J., Salánki, J. et al. Cadmium Ions Modulate Gaba Induced Currents in Molluscan Neurons. BIOLOGIA FUTURA 53, 105–123 (2002). https://doi.org/10.1556/ABiol.53.2002.1-2.12
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/ABiol.53.2002.1-2.12