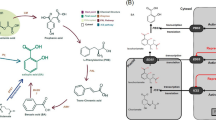
Abstract
Introduction
This study aimed to determine the effects of methyl jasmonate (MJ) combined with putrescine as eco-friendly elicitors on secondary metabolism and gene expression of alkaloid biosynthetic pathway in Catharanthus roseus in vifro-propagated shoots.
Methods
The expression of mitogen-activated protein kinase 3 and the transcription factor, octadecanoid-responsive Catharanthus AP2-domain3, upstream of plant alkaloids’ biosynthetic pathway, and of key genes in the pathway (CrPRXl, STR, DAT, and GS) are investigated as well using qRt-PCR. Antioxidant enzyme (superoxide dismutase, peroxidase, and catalase) activities and non-enzymatic antioxidants (phenolics, flavonoids, and carotenoids) contents have studied to determine the stress levels of the plant by spectrophotometer.
Results
Results showed increased contents of non-enzymatic antioxidants after 4–8 hr and enzymatic antioxidants activities after 24 hr. Alkaloids contents increased mostly after 1 week. The investigated signaling genes upregulated after 8 hr and biosynthetic genes after 24 hr of treatments. Combined treatments had more positive effects on gene expression levels, antioxidant responses, and secondary metabolite production than MJ individually.
Discussion
Increased effects of combined elicitor on genes expression may be due to cross talks between their signaling pathways. Combination of MJ and putrescine can be used as an eco-friendly elicitor for enhancing the production of economically important alkaloids in C. roseus.
Similar content being viewed by others
References
Abdelgawad, Z. A., Khalafaallah, A. A., Abdallah, M. M. (2014) Impact of methyl jasmonate on antioxidant activity and some biochemical aspects of maize plant grown under water stress condition. Agric. Sci. 5, 1077.
Akula, R., Ravishankar, G. A. (2011) Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal. Behav. 6, 1720–1731.
Alkazar, R., Marco, F., Cuevas, J. C., Patron, M., Ferrando, A., Carrasco, P., Tiburcio, A. F., Altabella, T. (2006) Involvement of polyamines in plant response to abiotic stress. Biotechnol Lett. 28, 1867–1876.
Dewanto, V., Wu, X., Adorn, K. K., Liu, R. H. (2002) Thermal processing enhances the nutritional value of tomatoes by increasing total antioxidant activity. J. Agric. Food Chem. 50, 3010–3014.
Gao, F., Su, Q., Fan, Y., Wang, L. (2010) Expression pattern and core region analysis of AtMPK3 promoter in response to environmental stresses. Sci. China Life Sci. 53, 1315–1321.
Ghosh, B. (2000) Polyamines and plant alkaloids. Indian J. Exp. Biol. 38, 1086–1091.
Gill, S. S., Tuteja, N. (2010) Polyamines and abiotic stress tolerance in plants. Plant Signal. Behav. 5, 26–33.
Heath, R. L., Packer, L. (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 125, 189–198.
Hussain, S. S., Ali, M., Ahmad, M., Siddique, K. H. (2011) Polyamines: natural and engineered abiotic and biotic stress tolerance in plants. Biotechnol. Adv. 29, 300–311.
Kubis, J. (2008) Exogenous spermidine differentially alters activities of some scavenging system enzymes, H202 and superoxide radical levels in water-stressed cucumber leaves. J. Plant Physiol. 165, 397–406.
Kumari, P., Reddy, C. R., Jha, B. (2015) Methyl jasmonate-induced lipidomic and biochemical alterations in the intertidal macroalga Gracilaria dura (Gracilariaceae, Rhodophyta). Plant Cell Physiol. 56, 1877–1889.
Kupper, F. C., Gaquerel, E., Cosse, A., Adas, F., Peters, A. F., Muller, D. G., Kloareg, B., Salaun, J. P., Potin, P. (2009) Free fatty acids and methyl jasmonate trigger defense reactions in Laminaria digitata. Plant Cell Physiol. 50, 789–800.
Lichtenthaler, H. K. (1987) Chlorophylls and carotenoids: pigments of photo synthetic biomembranes. Methods Enzymol. 148, 350–382.
Livak, K. J., Schmittgen, T. D. (2001) Analysis of relative gene expression data using real time quantitative PCR and the 2 △△CT method. Methods 25, 402–408.
Mandal, C., Ghosh, N., Adak, M. K., Dey, N. (2013) Interaction of polyamine on oxidative stress induced by exogenously applied hydrogen peroxide in Salvinia nutans Linn. Theor. Exp. Plant Physiol. 25, 223–230.
Minocha, R., Majumdar, R., Minocha, S. C. (2014) Polyamines and abiotic stress in plants: a complex relationship. Front. Plant Sci. 5, 175.
Miranda-Ham, L., Islas-Flores, I. (2007) Accumulation of mono-terpenoid indole alkaloids in periwinkle seedlings (Cathar-anthus roseus) as a model for the study of plant-environment interactions. Biochem. Mol. Biol. Educ. 35, 206–210.
Ncube, B., Van Staden, J. (2015) Tilting plant metabolism for improved metabolite biosynthesis and enhanced human benefit. Molecules 20, 12698–12731.
Ozturk, B., Yildiz, K., Ozkan, Y. (2015). Effects of pre-harvest methyl jasmonate treatments on bioactive compounds and peel color development of “Fuji” apples. Int. J. Food Prop. 18, 954–962.
Pan, Q., Mustafa, N. R., Tang, K., Choi, Y. H., Verpoorte, R. (2016) Monoterpenoid indole alkaloids biosynthesis and its regulation in Catharanthus roseus: a literature review from genes to metabolites. Phytochem. Rev. 15, 221–250.
Poonam, S., Kaur, H., Geetika, S. (2013). Effect of jasmonic acid on photosynthetic pigments and stress markers in Cajanus cajan (L.) Millsp. seedlings under copper stress. Am. J. Plant Sci. 4, 817.
Qu, Y., Easson, M. E., Simionescu, R., Hajicek, J., Thamm, A. M., Salim, V., De Luca, V. (2018) Solution of the multistep pathway for assembly of corynanthean, strychnos, iboga, and aspidosperma monoterpenoid indole alkaloids from 19E-geissoschizine. Proc. Natl. Acad. Sci. U. S. A. 115, 3180–3185.
Raina, S. K., Wankhede, D. P., Jaggi, M., Singh, P., Jalmi, S. K., Raghuram, B., Sinha, A. K. (2012) CrMPK3, a mitogen activated protein kinase from Catharanthus roseus and its possible role in stress induced biosynthesis of monoterpenoid indole alkaloids. BMC Plant Biol. 12, 134.
Sanchez-Rojo, S., Cerda-Garcia-Rojas, C. M., Esparza-Garcia, F., Plasencia, J., Poggi-Varaldo, H. M., Ponce-Noyola, T., Ramos-Valdivi, A. C. (2015) Long-term response on growth, antioxidant enzymes, and secondary metabolites in salicylic acid pre-treated Uncaria tomentosa microplants. Biotechnol. Lett. 37, 2489–2496.
Tadolini, B. (1988) Polyamine inhibition of lipoperoxidation. The influence of polyamines on iron oxidation in the presence of compounds mimicking phospholipid polar heads. Biochemic. J. 249, 33–36.
Talaat, I. M., Bekheta, M. A., Mahgoub, M. H. (2005) Physiological response of periwinkle plants (Catharanthus roseus L.) to tryptophan and putrescine. Int. J. Agric. Biol. 7, 210–213.
Van Moerkercke, A., Steensma, P., Schweizer, F., Pollier, J., Gariboldi, I., Payne, R., Bossche, R. V., Miettinen, K., Espoz, J., Purnama, P. C., Kellner, F. (2015) The bHLH transcription factor BIS1 controls the iridoid branch of the monoterpenoid indole alkaloid pathway in Catharanthus roseus. Proc. Natl. Acad. Sci. U. S A. 112, 8130–8135.
Verma, S., Mishra, S. N. (2005) Putrescine alleviation of growth in salt stressed Brassica juncea by inducing antioxidative defense system. J. Plant Physiol. 162, 66977’.
Walters, D. R. (2003) Polyamines and plant disease. Phytochem-istry 64, 97–107.
Wojciak-Kosior, M., Sowa, I., Blicharski, T., Strzemski, M., Dresler, S., Szymczak, G., Wnorowski, A., Kocjan, R., Swieboda, R. (2016) The stimulatory effect of strontium ions on phytoestrogens content in Glycine max (L.) Merr. Molecules 21, 1–9.
Zhang, H., Hedhili, S., Montiel, G., Zhang, Y., Chatel, G., Pre, M., Gantet, P., Memelink, J. (2011) The basic helix-loop-helix transcription factor CrMYC2 controls the jasmonate-responsive expression of the ORCA genes that regulate alkaloid biosynthesis in Catharanthus roseus. Plant J. 67, 61–71.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Khataee, E., Karimi, F. & Razavi, K. Alkaloids production and antioxidant properties in Catharanthus roseus (L.) G. Don. shoots and study of alkaloid biosynthesis-related gene expression levels in response to methyl jasmonate and putrescine treatments as eco-friendly elicitors. BIOLOGIA FUTURA 70, 38–46 (2019). https://doi.org/10.1556/019.70.2019.05
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/019.70.2019.05