Abstract
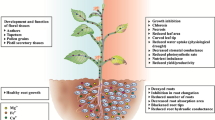
Studies were carried out to investigate the effect of zinc sulfide nanoparticles on growth and anti-oxidant markers in Brassica juncea. Growth of Brassica júncea seedlings was positively affected by zinc sulfide nanoparticle treatment, with a maximum increase of 49% in shoot dry weight, being recorded at 15 ppm concentration. Increased chlorophyll content and maximal sugar accumulation was also recorded at 15 ppm zinc sulfide nanoparticle treatment. The seedlings treated with 15 ppm and higher concentrations of zinc sulfide nanoparticles recorded increased reduced-glutathione leveis as compared to the untreated seedlings. Higher growth indices of the treated seedlings were associated with improved antioxidant marker leveis, recorded in terms of lower proline accumulation, and reduced hydrogen peroxide & lipid peroxidation leveis. No significant change in total ascorbate content was recorded up to 15 ppm, while a 15% increase was recorded at 100 ppm zinc sulfide nanoparticle treatment. The reduced ascorbate content decreased by just 4.44% at 15 ppm zinc sulfide nanoparticle treatment, while the maximum reduced ascorbate level was recorded at 100 ppm. These observations indicate an alteration in the antioxidant status of the treated seedlings, which is responsible for improved growth profile of the seedlings treated with 15 ppm zinc sulfide nanoparticles, as compared to the seedlings treated with 100 ppm zinc sulfide nanoparticles. The results clearly indicate that zinc sulfide nanoparticles can be used to augment the growth of Brassica júncea seedlings, and this growth stimulatory effect is associated with alterations in antioxidant status of the treated seedlings.
Similar content being viewed by others
References
Adhikary B.H., Shrestha J. & Baral B.R. 2010. Effects of micronutrients on growth and productivity of maize in acidic soil. Int. Res. J. Appl. Basic Sci. 1: 8–15.
Alexieva V., Sergiev I., Mapelli S. & Karanov E. 2001. The effect of drought and ultraviolet radiation on growth and stress markers in pea and wheat. Plant Cell Environ. 24: 1337–1344.
Arora S., Sharma P., Kumar S., Nayan R., Khanna P.K. & Zaidi M.G.H. 2012. Gold-nanoparticle induced enhancement in growth and seed yield of Brassica júncea. Plant Growth Regul. 66: 303–310.
Bates L.S., Waldren R.P. & Teare I.D. 1973. Rapid determination of free proline for water-stress studies. Plant Soil 39: 205–207
Calandra P., Longo A. & Turco-Liveri V. 2003. Synthesis of ultrasmall ZnS nanoparticles by solid-solid reaction in the conflned space of AOT reversed micelles. J. Phys. Chem. B. 107: 25–30.
Da Costa M.V.J. & Sharma P.K. 2015. Influence of titanium dioxide nanoparticles on the photosynthetic and biochemical processes in Oryzo, sativa. inter. J. Recent Scient. Res. 6: 2445–2451.
Gao F., Hong F., Liu C., Zheng L., Su M., Wu X., Yang F., Wu C. & Yang P. 2006. Mechanism of nano-anatase Ti02 on promoting photosynthetic carbon reaction of spinach. Biol. Trace Elem. Res. 111 (1-3): 239–53.
Gayou V.L., Salazar-Hernandez B., Delgado Macuil R., Zavala G., Santiago P. & Oliva A. I. 2010. Structural studies of ZnS nanoparticles by high resolution transmission electron microscopy. J. Nano. Res. 9: 125–132
Gerland P., Raftery A.E., Sevcikova H., Li N., Gu D., Spoorenberg T., Alkema L., Fosdick B.K., Chunn J., Lalic N. & Bay G. 2014. World population stabilization unlikely this century. Science 346: 234–237.
Heath R.L. & Packer L. 1968. Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 125: 189–198.
Hiscox A.D. & IsraelstamG. S. 1979. A method for the extraction of chlorophyll from leaf tissue without maceration. Can. J. Bot. 57: 1332–1334.
Law M.Y., Charles S.A. & Halliwell B. 1983. Glutathione and ascorbic acid in spinach (Spinacia oleracea) chloroplasts. The effect of hydrogen peroxide and of Paraquat. Biochem. J. 210: 899–903.
Ma C., Chhikara S., Xing B., Musante C., White J.C. & Dhankher O.P. 2013. Physiological and molecular response of Arabidopsis thaliana (L.) to nanoparticle cerium and in-dium oxide exposure. ACS Sustain. Chem. & Engineer. 1 (7): 768–78.
Matysik J., Bhalu B. & Mohanty P. 2002. Molecular mechanisms of quenching of reactive oxygen species by proline under stress in plants. Current Science 82: 525–532.
May M.J., Vernoux T., Leaver C., Van Montagu M. & Inze D. 1998. Glutathione homeostasis in plants: implications for en-vironmental sensing and plant development. J. Exp. Bot. 49: 649–667.
Mishra V., Mishra R.K., Dikshit A. & Pandey A.C. 2014. Inter-actions of nanoparticles with plants: an emerging perspective in the agriculture industry, pp. 159–180. In: Ahmad P. & Rasool S. (eds), Emerging Technologies and Management of Crop Stress Tolerance: Biological Techniques, Elsevier Aca-demic press, USA.
Mittler R. 2002. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 7: 405–10.
Mittler R., Vanderauwera S., Gollery M. & Van Breusegem F. 2004. Reactive oxygen gene network of plants. Trends Plant Sci. 9: 490–498.
Mokrasch L.C. 1954. Analysis of hexose phosphates and sugar mixtures with the anthrone reagent. J. Biol. Chem. 208: 55–59.
Moron M.S., Depierre J. W. & Mannervik B. 1979. Leveis of glutathione, glutathione reduciase and glutathione S-transferase activities in rat lung and liver. Biochim. Biophys. Acta 582: 67–78.
Rao S. & Shekhawat G.S. 2014. Toxicity of ZnO engineered nanoparticles and evaluation of their effect on growth, metabolism and tissue speciflc accumulation in Brassica júncea. J. Environ. Chem. Engineer. 2: 105–114.
Rico C.M., Hong J., Morales M. I., Zhao L., Barrios A. C., Zhang J.Y., Peralta-Videa J.R. & Gardea-Torresdey J.L. 2013. Effect of cerium oxide nanoparticles on rice: a study involving the antioxidant defence system and in vivo fluorescence imaging. Environ. Sci. & Technol 47 (11): 5635–5642.
Sharma P., Bhatt D., Zaidi M.G.H., Saradhi P.P., Khanna P.K. & Arora S. 2012. Silver nanoparticle-mediated enhancement in growth and antioxidant status of Brassica júncea. Appl. Biochem. Biotechnol. 167 (8): 2225–2233.
Shaw A.K. & Hossain Z. 2013. Impact of nano-CuO stress on rice (Oryza sativa L.) seedlings. Chemosphere 93: 906–915.
Tang S.Y. & Cao Y.P. 2003. Effects of spraying different forms of silicon (Si) on growth as stress resistance of rice plants. Sci. Fertil. 2: 16–22.
Thimmaiah S.K. 2009. Standard Methods of Biochemical Analysis. Kalyani publishers, New Delhi, pp. 51–53.
Vitti A., Nuzzaci M., Scopa A., Tataranni G., Tamburrino I. & Sofo A. 2014. Hormonal response and root architecture in Arabidopsis thaliana subjected to heavy metais. inter. J. Plant Biol. 5: 5226–5232.
Xiang C., Werner B.L., E’Lise M.C. & Oliver D.J. 2001. The biological functions of glutathione revisited in Arabidopsis transgenic plants with altered glutathione leveis. Plant Phys-iol. 126: 564–574.
Yang F., Hong F., You W., Liu C., Gao F., Wu C. & Yang P. 2006. Influence of nano-anatase TiO2 on the nitrogen metabolism of growing spinach. Biol. Trace Element Res. 110: 179–190.
Zheng L., Hong F., Lu S. & Liu C. 2005. Effect of nano-Ti02 on strength of naturally aged seeds and growth of spinach. Biol. Trace Element Res. 104: 83–91.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nayan, R., Rawat, M., Negi, B. et al. Zinc sulfide nanoparticle mediated alterations in growth and anti-oxidant status of Brassica juncea. Biologia 71, 896–902 (2016). https://doi.org/10.1515/biolog-2016-0107
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/biolog-2016-0107