Abstract
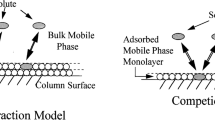
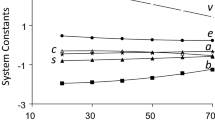
Plots of the retention factor against mobile phase composition were used to organize a varied group of solutes into three categories according to their retention mechanism on an octadecylsilioxane-bonded silica stationary phase, AscentisTM C18, with acetonitrile-water and methanol-water mobile phase compositions containing 10–70% (v/v) organic solvent. The solutes in category 1 could be fit to a general retention model, Eq. (1), and exhibited normal retention behavior for the full composition range. The solutes in category 2 exhibited normal retention behavior at high organic solvent compositions with a discontinuity at low organic solvent mobile phase compositions. The solutes in category 3 exhibited a pronounced step or plateau in the middle region of the retention plots with a retention mechanism similar to category 1 at mobile phase compositions after the discontinuity and a different retention mechanism before the discontinuity. Selecting solutes and appropriate composition ranges from the three categories where a single retention mechanism was operative allowed modeling of the experimental retention factors using the solvation parameter model. These models were then used to predict retention factors for solutes excluded from the models. The overwhelming number of residual values, here defined as the difference between experimental and model predicted retention factors for the excluded solutes, could be explained by contributions from steric repulsion. The latter defined as the inability of solutes to fully insert themselves into the solvated stationary phase because of their size or conformation. Steric repulsion resulted in a systematic reduction in retention compared with predicted values for the fully inserted solute. The bonding density of the stationary phase; the type and composition of the mobile phase; and the size, conformation, type and number of functional groups on the solute are shown to affect the contribution of steric repulsion to the retention mechanism.
Similar content being viewed by others
References
Poole CF, Poole SK (2002) J Chromatogr A 965:263–299
Poole CF (2003) The Essence of Chromatography. Elsevier, Amsterdam
Abraham MH, Roses M, Poole CF, Poole SK (1997) J Chromatogr A 10:358–368
Lazaro E, Rafols C, Roses M (2005) J Chromatogr A 1081:163–173
Wang A, Tan LC, Carr PW (1999) J Chromatogr A 848:21–37
Baczek T, Kaliszan R, Novotna K, Jandera P (2005) J Chromatogr A 1075:109–115
Snyder LR, Dolan JW, Carr PW (2004) J Chromatogr A 1060:77–116
Zhao YH, Abraham MH (2005) J Org Chem 70:2633–2640
Abraham MH, Zhao YH (2005) Phys Chem Chem Phys 7:2418–2422
Sander LC, Pursch M, Wise SA (1999) Anal Chem 71:4821–4830
Poole CF, Poole SK, Gunatilleka AD (2000) Adv Chromatogr 40:159–230
Miyabe K, Guiochon G (2000) J Chromatogr A 903:1–12
Tchapla A, Colin H, Guiochon G (1984) Anal Chem 56:621–625
Tchapla A, Heron S, Lesellier E (1993) J Chromatogr A 656:81–112
Tchapla A, Heron S, Chromatogr J (1994) J Chromatogr 684:175–188
http://www.sigmaaldrich.com/chromatography/Ascentis
Kiridena W, Poole CF, Koziol WW (2004) J Chromatogr A 1060:177–185
Abraham MH, Ibrahim A, Zissimos AM (2004) J Chromatogr A 1037:29–47
Poole CF, Poole SK, Abraham MH (1998) J Chromatogr A 798:207–222
Kiridena W, Poole CF, Koziol WW (2004) J Chromatogr A 1060:177–185
Kiridena W, DeKay C, Villiere ND, Koziol WW, Poole CF (2005) Chromatographia 61:587–593
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Poole, C.F., Ahmed, H., Kiridena, W. et al. Contribution of Steric Repulsion to Retention on an Octadecylsiloxane-Bonded Silica Stationary Phase in Reversed-Phase Liquid Chromatography. Chroma 62, 553–561 (2005). https://doi.org/10.1365/s10337-005-0654-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-005-0654-0